Correlation of the estimated glomerular filtration rate (eGFR) and the antiretroviral therapy of HIV-1 patients at a rural hospital in South Western Uganda
Ritah Kiconco*1,2,3
1Department of Medical Laboratory Science, School of Allied Health Sciences, Kampala International University-Western Campus, Ishaka-Bushenyi, Uganda.
2Department of Medical Laboratory Science, Faculty of Medicine, Mbarara University of Science and Technology, Mbarara, Uganda.
3Department of Pathology and Diagnostics, Kampala International University Teaching Hospital, Ishaka-Bushenyi, Uganda
Received Date: 18/09/2020; Published Date: 21/10/2020
*Corresponding author: Ritah Kiconco, Department of Medical Laboratory Science, School of Allied Health Sciences, Kampala International University-Western Campus, Ishaka-Bushenyi, Department of Medical Laboratory Science, Faculty of Medicine, Mbarara University of Science and Technology, Mbarara, Department of Pathology and Diagnostics, Kampala International University Teaching Hospital, Ishaka-Bushenyi, Uganda. rkiconco@must.ac.ug
Abstract
Background: Patients infected with Human Immunodeficiency Virus (HIV) live longer while receiving combination therapies of Highly Active Anti-Retroviral Therapy (HAART). However, kidney diseases have emerged as significant causes of morbidity and mortality among these patients. If patients are not monitored, renal complications may develop and a poor prognosis could result into severe kidney damage. Thisvstudy therefore, investigated the effect of the various combination therapies given to the patients’ vis a vie their estimated glomerular filtration rate. Methodology: This cross-sectional study recruited 200 study participants who were receiving HIV healthcare services from St Daniel Comboni Hospital Kyamuhunga in Bushenyi district. Blood samples were obtained for the analysis of creatinine levels done on a Reflotron® chemistry analyser. Estimated Glomerular Filtration Rate (eGFR) was computed using an online calculator that followed the MDRD (modification of Diet in Renal Disease) equation. Other socio demographic data were obtained from participants along with their drug combination therapies. Results: Of the 200 study participants, 109 were female. Most of them were between the ages of 19-49 years. There were 4 drug combination therapies that were investigated in this study; (AZT+3TC+EFV- 12.5%, AZT+3TC+NVP-21.5%, TDF+3TC+EFV-43.5%, TDF+3TC+NVP-22.5%) and upon linear regression analysis, none of them had a statistically significant relationship with the eGFR although they seemed to have both a positive and negative correlation with the latter. Conclusion: Combination therapies given to HIV/AIDS patients are safe to use and do not pose as such a significant health risk to kidney damage unless they are poorly prescribed and the patients are not routinely monitored.
Introduction
As patients infected with HIV/AIDS live longer while receiving antiretroviral therapy, [1,2] kidney diseases have emerged among them as significant causes of morbidity and mortality. [3] All over the world, most of the antiretroviral first-line drugs used in the treatment of HIV patients includes regimen using a Nucleoside / Nucleotide Reverse Transcriptase Inhibitors (NRTI) with a protease inhibitor or a Non-Nucleoside Reverse Transcriptase Inhibitor (NNRTI) [4]. Uganda’s health ministry has implemented the guidelines for administering treatment to naïve HIV patients using first-line drug combinations containing Tenofovir (TDF), Zidovudine (AZT), Lamivudine (3TC), Efavirenz (EFV), and emicitabine (FTC) as guided by the World Health Organization (WHO). Although these drugs usually undergo several clinical trials before they are declared safe, several reports have indicated that they could possess nephrotoxic properties [4].
The kidney plays a major role in the excretion of antiretroviral drugs and this makes it vulnerable to various types of injuries from some of these agents including Acute Kidney Injury (AKI), tubulopathies, Chronic Kidney Disease (CKD) and end stage renal disease requiring renal replacement therapy [5]. Antiretroviral therapy can contribute to renal dysfunction directly by inducing acute tubular necrosis, acute interstitial nephritis, crystal nephropathy, and renal tubular disorders or indirectly via drug interactions. [5] Chronic kidney disease should be diagnosed in its early stages through routine screening and careful attention to changes in glomerular filtration rate or creatinine clearance [6]. It is also important that patients with Chronic Kidney Disease (CKD) are not denied the best combinations, especially since most drugs can be adjusted based on the estimated glomerular filtration rate [7]. With the increase in HAART use, clinicians must screen patients for the development of kidney disease especially if the regimen employed increases risk of kidney injury.
One of the metabolites which is exclusively filtered by the kidney is creatinine. Most of the creatinine is found in the muscle tissue where it is present as creatine phosphate and serves as a high energy storage reservoir for conversion to Adenosine Tri Phosphate (ATP). Creatinine, the waste product of this reaction, is transported to the kidneys and eliminated [8]. Independent of the diet, serum creatinine concentration depends almost entirely upon its excretion rate by the kidneys. For this reason, its elevation is highly specific for kidney disease and in this study, it was used to calculate the eGFR together with other factors such as the age, race and gender of the study participant.
Materials and Methods
Study area
The study was carried out at the HIV clinic of St Daniel Comboni Hospital Kyamuhunga in Bushenyi district. This private not for profit health facility has a bed capacity of 100 and provides minimum health care which is comprehensive of curative, preventive, promotive and rehabilitative care to the rural community. It is located approximately 300km from Kampala, the capital city of Uganda.
Study design and population
This was an observational study that recruited known HIV/AIDS patients who had been receiving antiretroviral therapy for at least 6 months and had no debilitating illness that could otherwise impede them from participation. Only Adults who were of either sex were enrolled to participate in this study during the months of January to March 2013. The participants were recruited following the systematic random sampling technique. Those who met the study criteria were asked to consent to participation before they were subjected to the study questions on the data collection form. Our study was approved by a select committee from the School of Allied Health Sciences and the Department of Medical Laboratory Sciences at Kampala International University. Permission was sought from the management of St Daniels Comboni hospital before commencement of the data collection activities. Written informed consent was obtained from participants prior to being recruited into the study. To ensure confidentiality, study identification numbers were used rather than participants’ names. No names were used on data collection documents except consent forms. All documents in this study were safely locked up and accessed by only the study team. Data collection tools were stored separately from consent forms. Those Study participants who wished not to participate in the study had a right to opt out at any point during the study and this was not to affect their routine healthcare at the clinic.
Sample Size determination
The Sample size was calculated from Fischer’s formula [9] indicated below:
n = Z2P (1-P)/W2
Where,
n = minimum sample size, Z=1.96 (for 95% confidence interval), P = 15% estimated population with HIV/AIDS and are on antiretroviral therapy and W is the margin of error which is 5%. Therefore, 200 participants were recruited into this study.
Laboratory analysis of creatinine
Approximately 4 milliliters of whole blood were collected from each participant into an Ethylene Diamine Tetra Acetic acid (EDTA) coated vacutainer following standard operating procedures for venepuncture. The specimen was further processed on a Reflotron® chemistry analyser using system compatible reagent kits. During analysis, the instrument displayed the results of creatine levels after 1.5 minutes in mg/dl. The results were subsequently recorded and used to compute the estimated glomerular filtration rate.
Computation of the estimated Glomerular Filtration Rate
This was done using an online calculator which follows the MDRD equation for the computation of the estimated Glomerular Filtration Rate (eGFR) found at https://www.mdcalc.com/mdrd-gfr-equatin#next-steps This equation utilizes information about the study participants age, race, gender and creatinine levels in either µmol/l or mg/dl. After obtaining the eGFR, the value was interpreted on a scale found at the same website. This scale was modified to suite the study sample size and two categories of pre-clinical and clinical cases with kidney disease were developed.
Data Analysis
The generated data was analyzed using Epi info 7, a data analysis software program developed by the Centers for Disease Control and Prevention available via the link https://www.cdc.gov/epiinfo/index.html Descriptive statistics of the study population such as the age, sex and combination therapy were analyzed using frequencies and percentages. Means and Standard deviation were used to describe the continuous variables such as the creatinine levels and the estimated glomerular filtration rate. Through cross tabulation, Fischer’s exact test was used to check for significance among the different combination therapies with the age and gender of the study participants. Linear regression was done to ascertain any significant relationship between the glomerular filtration rate and the combination therapies.
Results and Discussion
Table 1: Characteristics of the study population, N=200
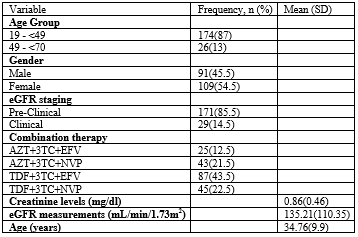
Abbreviations: eGFR: estimated Gleomerular Filtration Rate; TDF: Tenofovir; AZT: Zidovudine; 3TC: Lamivudine; EFV: Efavirenz; NVP: Nevirapine; mL: milliliter; min: minute; m2: meters squared
Table 2: Crosstabulation of Age group, Sex and eGFR staging with different combination therapies.
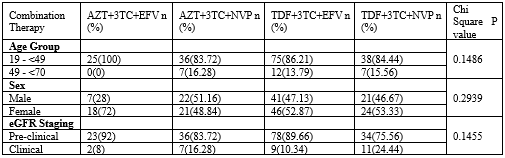
P>0.05 was statistically significant.
Abbreviations: eGFR: estimated Glomerular Filtration Rate; TDF: Tenofovir; AZT: Zidovudine; 3TC: Lamivudine; EFV: Efavirenz; NVP: Nevirapine
Table 3: Linear regression of the eGFR measurements against the age, sex, and combination therapies.
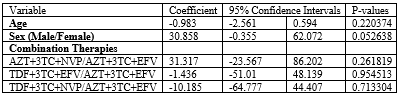
P>0.05 was statistically significant,
Abbreviations: eGFR: estimated Glomerular Filtration Rate; TDF: Tenofovir; AZT: Zidovudine; 3TC: Lamivudine; EFV: Efavirenz; NVP: Nevirapine
In this study, we investigated the effect of combined therapies on the estimated glomerular filtration rate of HIV patients who were taking these drugs. In our study according to table 1, the population was mainly composed of youths who were averagely aged 34 years and majority were women (54.5%). It was reported in the Uganda AIDS national indicator survey of 2011 that 12 to 13 percent of young women and men say they had sex before age 15 and that by age 18, sixty percent of the young women and forty-seven percent of the young men have initiated sexual activity. In this same survey, most of the youth had agreed to not have used protection methods. This therefore explains the noticeable number of youths and women we had in our study. Also, most of the participants were taking a combination therapy containing Tenofovir Disoproxil Fumarate (TDF). TDF containing combinations have been promoted as first-line drug therapy due to less viral resistance developing among those taking them [10,11] and thus the wide usage among the HIV-1 infected patients in developing countries like Uganda.
Studies on the renal impairment among patients on antiretroviral therapy have reported conflicting results especially in Africa. According to the eGFR staging used in our study, most of the participants were in the pre-clinical or normal staging implying that most of them had their eGFR greater than or equal to 90 mL/min/1.73m2 and only 14.5% (Table 1) had abnormal eGFR thus categorizing them as clinical. The prevalence of an abnormal eGFR in this study was quite higher than the 4% observed in a similar study conducted in Malawi that was looking at the renal outcomes of patients initiated on TDF combination therapy [12]. We believe that the discrepancies in this prevalence, firstly could have been contributed by the difference in the formulas used to determine the estimated Glomerular Filtration Rate (eGFR). The study in Malawi used the Cockcroft-Gault formula while we used the Modification of Diet in Renal Disease (MDRD) formula. Secondly, the same study derived its prevalence from only those participants who were taking a combination therapy containing TDF and yet in our study, we included even those who were taking other combination therapy exclusive of TDF. Thirdly, the patients in the Malawi study were new recruits to the antiretroviral therapy while those in our study had been on this treatment for at least 6 months. It can be hypothesized that the duration on treatment can also affect the integrity of the kidney status and thus the observed low and high prevalence in these studies respectively. In the same vein, another study conducted in Ethiopia on the effect of antiretroviral therapy on the renal and liver function of HIV patients [13] obtained an 11.6% prevalence which is quite similar to our study’s finding. This study just like ours used the MDRD equation to estimate the eGFR. Also, the prevalence was obtained from those patients that had been on antiretroviral therapy for some time.
There was no significant relationship between the combination therapies and the different age groups, sex and eGFR staging (Table 2). This finding is in agreement with two cohort studies conducted in eastern and southwestern Uganda [14] who found that there were no differences in renal function even when different durations on antiretroviral therapy were compared. The study in Malawi however, found that kidney injury predominately occurred during the first six months of combined ART and increasing age was a predisposing factor [12]. Contrary to this, a similar study assessing the severe renal dysfunction associated with renal impairment among patients initiated on ART [6]. Found that younger age and female sex were associated with an increase in eGFR with small but statistically significant differences between regimen.
In our study the estimated Glomerular Filtration Rate (eGFR) measurements across the age, gender and combination therapies had no statistically significant relationship despite that fact that those who took a combination containing Tenofovir Disoproxil Fumarate (TDF) had a negative correlation with the eGFR meaning that taking a combination therapy containing TDF decreases the eGFR. Those who took a combination therapy containing Zidovudine (AZT) had a positive correlation with the eGFR implying that a combination therapy with AZT increases the eGFR thus having a good renal outcome (Table 3). In a study conducted to ascertain the extent to which renal impairment due to TDF therapy occurs, [15] it was found that the eGFR decreased during the first 3 months of TDF exposure and it increased upon discontinuation of the antiretroviral therapy. Another similar but controlled study among Asian population could not agree more on the effect of the TDF therapy and the observed decline in eGFR [16].
We acknowledge that our study has limitations that might have affected the study findings such as the lack of a control group and not thoroughly evaluating the kidney injury using other biochemical parameters such as assessment of albuminuria.
Conclusion
Combination therapies given to HIV/AIDS patients are safe to use and do not pose a significant health risk to kidney damage unless they are poorly prescribed and there is no individual patient monitoring especially for those taking TDF combination therapy. Assessment of the eGFR is a good indicator of deteriorating renal function among HIV-1 patients and routine assessment especially from prolonged usage of antiretroviral therapy is still encouraged as nephrotoxic drugs can have devastating effects on the kidney.
Data Availability
The data sets generated and/or analysed during this study are available from the corresponding author on request.
Conflicts of Interest
The author declares no conflict of interest.
Funding Statement
The study was self-funded.
Acknowledgment
Great appreciation is extended to the laboratory staff at St Daniel Comboni Hospital who aided in the laboratory determination of the creatinine levels at the hospital’s clinical laboratory unit.
References:
- Wandeler G, Johnson LF, Egger M. Trends in life expectancy of HIV-positive adults on ART across the globe: comparisons with general population. Current Opinion in HIV and AIDS. 2016; 11(5):492.
- Katz IT, Maughan-Brown B. Improved life expectancy of people living with HIV: who is left behind? The lancet HIV. 2017; 4(8): e324-e326.
- Mayor AM, Dworkin M, Quesada L, Rios-Olivares E, Hunter-Mellado RF. The morbidity and mortality associated with kidney disease in an HIV infected cohort in Puerto Rico. Ethnicity & disease. 2010; 20(101): S1.
- Kalyesubula R, Perazella MA. Nephrotoxicity of HAART. AIDS research and treatment. 2011; 2011.
- Alfano G, Cappelli G, Fontana F, Di Lullo L, Di Iorio B, Bellasi A, et al. Kidney disease in HIV infection. Journal of Clinical Medicine. 2019; 8(8):1254.
- Reid A, Stöhr W, Walker AS, Williams IG, Kityo C, Hughes P, et al. Severe Renal Dysfunction and Risk Factors Associated with Renal Impairment in HIV-Infected Adults in Africa Initiating Antiretroviral Therapy. Clinical infectious diseases. 2008; 46(8): 1271-81.
- Gianotti N, Galli L, Poli A, Salpietro S, Nozza S, Carbone A, et al. Estimated glomerular filtration rate trajectories in HIV-infected subjects treated with different ritonavir-boosted protease inhibitors and tenofovir disoproxil fumarate or abacavir. Medicine. 2016; 95(22).
- Erickson-Viitanen S, Geiger P, Yang W, Bessman SP. The creatine-creatine phosphate shuttle for energy transport—compartmentation of creatine phosphokinase in muscle. Regulation of Phosphate and Mineral Metabolism: Springer; 1982. p. 115-125.
- Kish L. Survey Sampling. New York: (1965). John Wiley & Sons.
- Etiebet M-AA, Shepherd J, Nowak RG, Charurat M, Chang H, Ajayi S, et al. Tenofovir Based Regimens Associated with Less Drug Resistance in HIV-1 Infected Nigerians Failing First-Line Antiretroviral Therapy. AIDS (London, England). 2013; 27(4): 553.
- Snow‐Lampart A, Chappell B, Curtis M, Zhu Y, Myrick F, Schawalder J, et al. No resistance to tenofovir disoproxil fumarate detected after up to 144 weeks of therapy in patients monoinfected with chronic hepatitis B virus. Hepatology. 2011; 53(3): 763-773.
- Chikwapulo B, Ngwira B, Sagno JB, Evans R. Renal outcomes in patients initiated on tenofovir disoproxil fumarate-based antiretroviral therapy at a community health centre in Malawi. International journal of STD & AIDS. 2018; 29(7): 650-657.
- Wondifraw Baynes H, Tegene B, Gebremichael M, Birhane G, Kedir W, Biadgo B. Assessment of the effect of antiretroviral therapy on renal and liver functions among HIV-infected patients: a retrospective study. HIV/AIDS (Auckland). 2017; 9: 1-7.
- Salome T, Kasamba I, Mayanja BN, Kazooba P, Were J, Kaleebu P, et al. The effect of Tenofovir on renal function among Ugandan adults on long-term antiretroviral therapy: a cross-sectional enrolment analysis. AIDS research and therapy. 2016; 13(1): 28.
- Jose S, Hamzah L, Campbell LJ, Hill T, Fisher M, Leen C, et al. Incomplete Reversibility of Estimated Glomerular Filtration Rate Decline Following Tenofovir Disoproxil Fumarate Exposure. The Journal of infectious diseases. 2014; 210(3): 363-373.
- Suzuki S, Nishijima T, Kawasaki Y, Kurosawa T, Mutoh Y, Kikuchi Y, et al. Effect of Tenofovir Disoproxil Fumarate on Incidence of Chronic Kidney Disease and Rate of Estimated Glomerular Filtration Rate Decrement in HIV-1–Infected Treatment-Naïve Asian Patients: Results from 12-Year Observational Cohort. AIDS patient care and STDs. 2017; 31(3):105-112.

