Microbial Quality Assessment of Selected Lebanese Dairy Products and the Antimicrobial Resistance of Bacterial Isolates
Imtithal S1*†, Samer S1†, Nadia A1, Hallak2, Ali A1 and Khatib2
1Department of Biological and Chemical Sciences, School of Arts and Sciences, Lebanese International University, Beirut, Lebanon
2Department of Nutrition and Food Sciences, School of Arts and Sciences, Lebanese International University, Beirut, Lebanon
†Both authors significantly contributed to the content of this manuscript.
Received Date: 01/07/2020; Published Date: 23/07/2020
*Corresponding author: Imtithal Sheet, Department of Biological and Chemical Sciences, School of Arts and Sciences, Lebanese International University, Beirut, 146404, Lebanon. E-mail: imtithal.sheet@liu.edu.lb
Abstract
The microbial quality of dairy products is of major concern to the Lebanese consumers due to the possibility of microbial contamination during the production process. The purpose of this study was to assess the microbial load of packaged and unpackaged samples of Labneh and Halloumi cheese and to determine the prevalence of antimicrobial resistance among the isolated bacteria. A total of 30 samples (15 Labneh & 15 Halloumi cheese samples) were collected in Beirut district, Lebanon. This study showed that the unpackaged samples of Labneh and Halloumi cheese were 9.7 folds more contaminated (in term of total bacterial count) compared to the packaged samples. The mean counts of S.aureus and total coliforms showed a higher significant level in Halloumi compared to Labneh samples without exceeding the normal respective ranges of Lebanese standards. Our findings showed no significant difference in the count of S. aureus, coliforms, yeast and molds in the packaged samples of Labneh compared to the unpackaged ones. Significant differences in terms of APC were found between the packaged and unpackaged Halloumi samples with the latest being more contaminated, though within the normal Lebanese standards. Finally, isolated S.aureus and E.coli revealed resistance to some antibiotics.
Keywords: Antibiotic Resistance; Food Safety; Halloumi Cheese; Labneh; Microbial Assessment
Introduction
Dairy products play an important role in maintaining body integrity and health well-being as well as reducing risks of many nutritional deficiency diseases through their composition of minerals, fats, proteins, carbohydrates and vitamins [1]. The high protein and lipid content in dairy products is considered to be an ideal environment for the growth of wide range of microorganisms with resultant spoilage of the product as well as serious health risks to consumers [2].
The potential hazards and biological risks in the dairy sector in Lebanon have also been assessed by the United Nation Industrial Development Organization (UNIDO) scientific committee and the identified risks were also alarming [3]. Currently, the microbial quality of dairy products is of major concern to the Lebanese consumers due to the major problems in the safety and quality control in many dairy industries. Bacterial contamination of dairy products can originate from different sources such as industrial water, poor hygiene, transport and milk collection conditions, and inferior quality raw milk [2]. Most contaminants of dairy products are coliforms and psychrotrophic group of microorganisms [4]. However, the predominant human bacterial pathogens that can potentially be transferred to milk include mainly Listeria monocytogenes, Salmonella sp., Staphylococcus aureus, and pathogenic Escherichia coli [5].
Labneh (strained yogurt) is a traditional dairy product in the Middle East, known as strained yogurt in Greece and the rest of Europe. It is a semi-solid dairy product obtained from yogurt after removal of part of its whey and water soluble compounds. Concentrated yogurt is produced either from cow, goat or sheep milk using either traditional or mechanical methods [6].
Additionally, brined cheeses are the most important family of cheeses for East-Mediterranean and neighboring countries [7]. The very well-known cheese and widely consumed in Lebanon and neighboring countries is Halloumi cheese. It is a member of the white brined cheeses family mainly made from sheep milk, goat milk or cow milk, and is classified as a semi hard to hard cheese, elastic without holes, and easily sliced [8].
Halloumi cheese is generally considered a safe and nutritious food, but foodborne illnesses linked to cheese consumption have been occurred in many countries [9].The factors that define the spoilage rate of cheese products are water activity, pH, storage temperature, characteristics of the lactic starter culture, types and viability of contaminating microorganisms, and the characteristics and quantities of residual enzymes [10].
The presence of microbes in dairy products might be attributed to several reasons including the quality and safety of raw milk, improper thermal treatment (pasteurization) of raw milk, and poor hygienic practices during production process of dairy products, utilization of non-potable water, improper handling of products or due to inadequate packaging and storage conditions [11].
Another major concern about bacterial pathogens of dairy products is that antimicrobials are frequently used for prevention and treatment of bovine infectious diseases such as mastitis [12]. Currently, the major worldwide public concern and health hazards associated with the use of antibiotics in livestock productions is the development of the antimicrobial resistant bacterial strains of animal origin and its consequent effect on human health [13]. It is generally accepted that antimicrobial resistant bacteria are produced, maintained and disseminated as a result of selection pressure induced by the use of antimicrobial drugs [14].
The outspread use of antibiotics, more specifically their unrestricted and indiscriminate use in agriculture, is threatened by the emergence of high level of antibiotic resistance of important pathogens [15]. The classes of antimicrobial drugs that are widely used in agriculture at the global level, which are of growing scientific concern with regards to their potential adverse effects and risk management steps, include the tetracyclines, aminoglycosides and β-lactams antibiotics [16].
Therefore, the principal aim of the current study is to assess the microbial load of different samples of the widely consumed Lebanese dairy products as Labneh and Halloumi cheese (packaged and unpackaged) collected from different retail markets in Beirut district. Another objective is to determine the prevalence of antimicrobial resistance among the pathogenic bacteria isolated from the collected dairy products.
Materials and Methods
Sampling
A total of 30 samples of two types of dairy products (Labneh and Halloumi cheese) were collected from different supermarkets and retail markets at Beirut district. The samples were packaged Labneh (8 samples), unpackaged Labneh (7 samples), packaged Halloumi cheese (8 samples) and unpackaged Halloumi cheese (7 samples). Cheese samples were collected with the brine used in the storage processes. All samples were transported directly to the laboratory in a cool box. Microbial analysis of these samples was conducted immediately and all analyses were performed in duplicate.
25 g of each collected dairy sample were transferred and placed aseptically in a sterile plastic bag containing 225 ml of sterile saline solution for homogenization. Homogenization was carried out using stomacher machine (Model, 1605 BL Smart) for 3 minutes. Following homogenization, ten-fold serial dilutions up to 103 were prepared and inoculated on appropriate media [17]. The pH values were measured in Labneh and Halloumi cheese samples using pH meter.
Microbial Analysis
Evaluation of microbiological criteria in dairy products is a vital step to ensure efficiency of any food safety plan. The level of coliforms is a crucial criterion to monitor processing and hygienic conditions. Furthermore, the aerobic plate count is one of the most common tests applied to indicate the microbial quality of food (El-Ziney, 2018).
Aerobic plate counts (APC), total coliforms, Staphylococcus aureus, and yeasts/molds counts were determined for each sample, as well as the presence or absence of different types of coliforms such as Escherichia coli, Enterobacter sp., Citrobacter sp. and Shigella.
Determination of Aerobic Plate Count (APC)
The most common procedure for the enumeration of bacteria is the aerobic plate count. It’s one of the microbial indicators for food quality [18]. 1 ml of each dilution was inoculated and spread onto plate count agar (PCA) (Himedia). The sample was left to dry on agar plate and then incubated at 35±1°C for 48±2 hours. Colony counts were made using colony counter and recorded as CFU/g [18,19].
Detection and Isolation of Total Coliform
For total coliform count, 1 ml of each dilution was spread evenly on a dried surface of MacConkey agar (Himedia). All inoculated plates were incubated at 35±1°C for 24±2 hours. Finally, colony count was made using colony counter and recorded as CFU/g. Typical isolated colonies were picked and cultured on nutrient agar slants and incubated at 35±1°C for 24±2hours for further identification using biochemical confirmatory tests as catalase test, citrate, indole, methyl red, Voges-Proskauer test and urease production [19].
Detection and Isolation of Staphylococcus aureus
The isolation, enumeration and identification of S. aureus were carried out using standard methods of USFDA [20]. S. aureus was detected and enumerated via surface plating 1 ml of each dilution on Baired Parker media agar (BPA) (Himedia) and incubating plates at 35±1°C for 48 ±2 hours. Colonies with typical S. aureus morphology appeared as black, convex and with or without light halo. The number of colonies was enumerated and recorded as CFU/g [19].
Isolation of Yeast and Molds
Yeast and molds were isolated and counted from the different dairy products using Sabouraud dextrose agar (SDA) (Himedia). 1 ml of each diluted sample was spread evenly on a dried surface of SDA and plates were incubated at 25±1°C for 5 days. The number of colonies was enumerated and recorded as CFU/g [21]. This procedure is based on the Food and Drug Administration of the United States (USFDA) [22].
Antibiotic Susceptibility Test
The isolates were revived by sub-culturing on nutrient agar at 35±1°C for 24±2 hours before being used for antibiotic susceptibility testing. The isolates were tested for their susceptibility to 4 different antimicrobial drugs namely Tetracycline (TCE 30μg), Penicillin (pcn 10μg), Ampicillin (amp 25μg), and Erythromycin (ERY 15μg) using the disk-diffusion method as set by the Clinical and Laboratory Standards Institute [22]. The antibiotic disks (Oxoid) were gently pressed to ensure their contact with the inoculated Mueller-Hinton agar surface, and the plates were incubated at 35±1°C for 24±2 hours. After the incubation period, the zones of inhibition were measured to the nearest millimeter. The interpretive breakpoints for resistance were determined according to the standard table as recommended by National Committee for The Clinical Laboratory Standard (NCCLS) interpretative methods [23]. The isolates were reported as susceptible, intermediate, or resistant.
Statistical Analysis
The data was analyzed using analysis of variance (ANOVA) complete randomized design, and computed via the IBM-SPSS 21 software. Differences among means of the treatments were analyzed using Duncan’s multiple range test. Significant differences are determined when p≤0.05.
Results
The study of the pH for the collected dairy samples was the first step of the current study. Our results indicated that the range of measured pH in the packaged Labneh sample was between 4.00 and 4.38, and between 4.02 and 4.99 in the unpackaged Labneh samples. The pH of Labneh samples should not exceed 4.5 as set by Lebanese standards. The range of pH values in the packaged and unpackaged Halloumi cheese samples was 5.70-6.36, and 6.01-6.68, respectively.
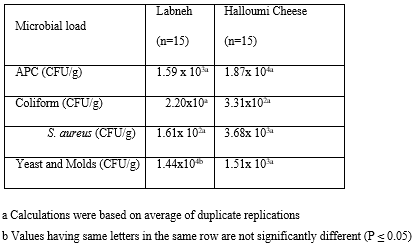
Bacterial loads in Labneh samples compared to Halloumi samples were determined in the current study as shown in Table 1. Average count of total aerobic bacteria in the 15 Labneh samples was 1.59 x 103 CFU/g compared to 1.87 x 104 CFU/g in the 15 Halloumi cheese samples. The mean count of Staphylococcus aureus and total coliform bacteria showed a significant higher level of these bacteria in Halloumi cheese samples (3.68 x 103 and 3.31 x 102 respectively) compared to Labneh samples (1.61 x 102 and 2.20 x 10 respectively). According to our results, the level of contamination of Labneh by S. aureus and coliform bacteria did not exceed the limit set by Lebanese standards (25x102 CFU/g), while Halloumi cheese samples exceeded it. Halloumi and Labneh samples were also tested for yeast and molds contamination. The data in the current study showed a similar pattern of molds and yeasts contamination with 1.51 x 103 and 1.44 x 103 CFU/g for the Labneh and Halloumi cheese samples, respectively. These were within the accepted range of the Lebanese standards that should not exceed 25x103 CFU/g. The differences in bacterial count in Labneh compared to Halloumi cheese samples were significant at p<0.05. (Table 1)
Table 1: Microbial Load of selected dairy samples (Labneh and Halloumi Cheese).
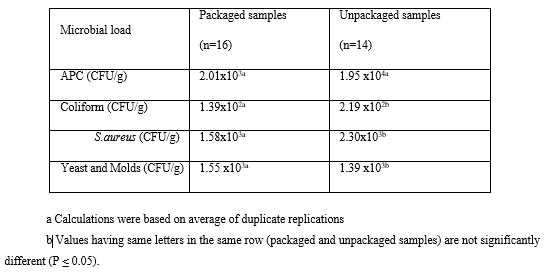
Microbial load in both packaged dairy samples (Labneh and Halloumi cheese) compared to the unpackaged dairy samples is shown in Table 2. The average counts of total aerobic bacteria in the packaged samples were 2.01 x103 CFU/g compared to 1.95 x104 CFU/g in unpackaged samples. The counts of isolated bacteria in the packaged and unpackaged samples showed significant difference. As shown in this table, the unpackaged samples were 9.7 folds more contaminated (in term of total bacterial count) compared to the packaged samples which are unacceptable values in accordance with the Lebanese standards (25x102 CFU/g). In the unpackaged and packaged samples, the counts of S. aureus were 2.30 x103 and 1.58x103 CFU/g respectively; whereas the counts of coliform bacteria were 2.19x102 and 1.39x102 CFU/g respectively, and the counts of yeast and molds were 1.39x103 and 1.55x103 CFU/g respectively. These results (Table 2) showed the absence of significant difference in term of total S. aureus, total coliform bacteria, yeast and molds count in the packaged and unpackaged samples. (Table 2)
Table 2: Average microbial count of packaged and unpackaged dairy samples.
When addressing the importance of packaging of Labneh, our finding showed, as indicated in Table 3, no significant difference in the count of S. aureus, coliform bacteria, yeast and molds in the packaged samples of Labneh compared to the unpackaged ones. In the packaged Labneh samples, the counts of S. aureus, coliform bacteria, yeast and molds were respectively 2.30x103, 8.13x100 and 1.23x103 CFU/g respectively compared to 8.20x10, 3.9x10 and 1.67x103 CFU/g respectively in the unpackaged samples. These counts are within the Lebanese standards. The difference in means of microbial load in packaged and unpackaged Labneh was not significant. The APC of packaged Labneh was 3.70x102 compared to 2.99x103 obtained from the unpackaged samples of Labneh samples. (Table 3)
Table 3: Average microbial count of packaged and unpackaged Labneh and Halloumi cheese.
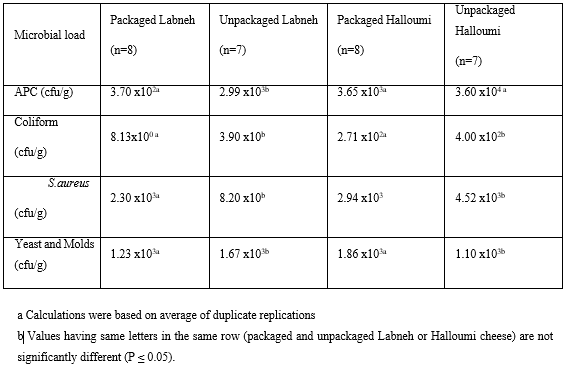
The means, represented in Table 3, of the APC of packaged and unpackaged Halloumi cheese were 3.65x103 and 3.60x104 respectively. These results translate the significant difference in terms of APC between the two dairy samples. Table 3 also shows the means of S. aureus, total coliform bacteria, yeast and molds counts in the packaged Halloumi being respectively 2.94x103, 2.71x102 and 1.86x103 CFU/g compared to counts in the unpackaged Halloumi samples that are respectively 4.52x103, 4.00x102 and 1.10x103 CFU/g. These differences in the microbial counts of the packaged and unpackaged tested Halloumi samples were not significant. These counts are within the Lebanese standards
The distribution of isolated bacteria, yeasts and molds in packaged and unpackaged Labneh and in packaged and unpackaged Halloumi cheese is shown in the Table 4. Our study showed that in unpackaged Halloumi samples (n=7), S. aureus was found in 85.71% of the tested samples, coliform bacteria were isolated from 71.42% of the samples, whereas yeast and molds were present in 100% of the samples. As for the 8 packaged samples of Halloumi cheese, S. aureus, total coliform bacteria, yeast and molds were found in 100%, 37.50% and 100% of these samples respectively. (Table 4)
Table 4: Incidence and percentage (%) of isolated microbes from the packaged and unpackaged Labneh and Halloumi Cheese.
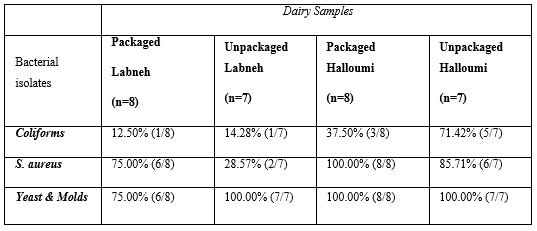
In Labneh tested samples, S. aureus was found in 75.00% of the 8 packaged tested samples, whereas it was isolated from 28.57% of the 7 unpackaged samples. Coliform bacteria were present in 12.50% and in 14.28% of packaged and unpackaged Labneh samples, respectively. Yeast and molds were detected in 75% and in 100% of packaged and unpackaged Labneh samples, respectively. However, 12.50% of the packaged Labneh samples contained no germs.
The distribution of isolated bacterial coliform species in packaged and unpackaged Labneh and packaged and unpackaged Halloumi cheese is represented in the Table 5. As shown in this table, Enterobacter sp. was the most frequently isolated species in the packaged Labneh (12.50% of the 8 samples) and Shigella sp. was predominant in the unpackaged Labneh samples (14.28% of the 7 samples). The present study showed that among the coliform bacteria, E.coli is the most frequently isolated species, found in 100% of the contaminated Halloumi packaged cheese samples (37.50% of the 8 samples). Citrobacter sp. was the predominant species in the contaminated unpackaged Halloumi cheese samples with percentage of 60.00%. (Table 5)
Table 5: Incidence and percentage (%) of isolated bacterial coliform species from the packaged and unpackaged Labneh and Halloumi Cheese.
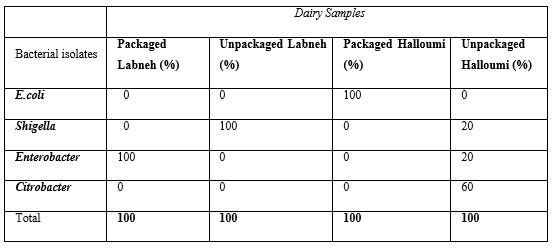
The remaining bacterial species Escherichia coli, Citrobacter and Shigella sp. were not identified in any of the examined packaged Labneh samples (n=8). However, E.coli, Citrobacter and Enterobacter sp. were not identified in any of the contaminated unpackaged Labneh samples. The distribution of the remaining species as Shigella and Enterobacter sp. in the contaminated unpackaged Halloumi cheese was 20.00% for both. However, neither Shigella nor Enterobacter nor Citrobacter sp. were identified in the collected unpackaged Halloumi samples.
When animals are processed for food, the bacteria from the animal can contaminate milk and other dairy products such as Labneh and Halloumi cheese. Manipulating food without following hygiene standards could also cause contamination of Labneh and Halloumi cheese with bacteria including the species that are resistant to antibiotics. 33% of the packaged and unpackaged Halloumi samples contaminated with E. coli, have shown resistance against tetracycline, while other contaminating coliform species isolated from these samples were sensitive to this antibiotic.
When the packaged Halloumi samples were examined, as shown in (Table 6), S. aureus isolates from the contaminated samples were screened for antibiotic resistance against 4 antibiotics: tetracycline, penicillin, ampicillin and erythromycin. In the packaged Halloumi cheese, 75% of S. aureus isolates were resistant to tetracycline and to penicillin whereas 25% were resistant to ampicillin. 87.5% of S. aureus isolates from the packaged Halloumi were resistant to erythromycin. The S. aureus isolates identified in the unpackaged Halloumi samples (100%) were all sensitive to tetracycline and resistant to penicillin and ampicillin. 50% of S. aureus isolates were resistant erythromycin. (Table 6)
Table 6: Antibiotic resistance profile of S. aureus isolated from dairy products against different types of antibiotics.
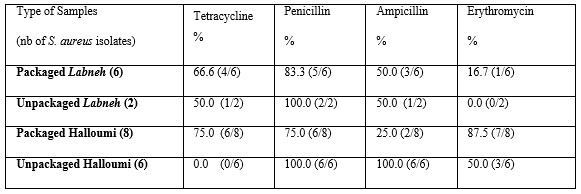
When determining the resistance developed by the S. aureus isolated from packaged Labneh, against the selected 4 antibiotics, summarized in Table 6, it turned out that 66.6% were resistant to tetracycline, 83.3%, 50% and 16.7% were resistant to penicillin, ampicillin and erythromycin, respectively. 50% of the S. aureus isolates obtained from packaged Labneh were found resistant to tetracycline and ampicillin. 100% of the isolates showed resistant to penicillin. All the S. aureus isolates were sensitive to erythromycin.
Discussion
Food safety is an increasing concern worldwide. Dairy products are susceptible to microbial contamination during the production or/and packaging. Halloumi cheese and Labneh are dairy products widely consumed in Lebanon and other Middle East, African and European countries. They are good source of proteins and provide important nutritional value in diet. Microbial quality of Halloumi cheese and Labneh, in Lebanon was, in this study, evaluated in the collected samples, packaged or not. The limitation of this study is the sample size. 15 halloumi samples and 15 Labneh samples were analyzed, 8 packaged and 7 unpackaged each. This should be considered in the perspectives of the current study.
Our findings showed that the presence of different species of microorganisms in these dairy products though the microbial contamination is within the normal ranges as determined by Lebanese standards, underlying general microbiological quality and good hygienic handling during the production process of these products and their packaging steps. The microbial contamination can be caused by environmental sources such as water and air. The lack of aseptic handling and good hygienic practices in the production line and/ or packaging processes is also a source of microbial contamination.
In the present study, S. aureus was found in 85.71% of unpackaged Halloumi samples, in 100% of packaged Halloumi samples, in 28.57% of unpackaged Labneh samples and in 75% of packaged Labneh sample. This bacterium, to cause food born disease, requires high dose contamination (105- 106 CFU/ ml) as established by the International Dairy Federation, whereas the average of S. aureus load in the tested samples did not exceed these standards [24]. However, [25] found that the unacceptable levels of S. aureus were found on average in 1% of Labneh samples collected from Ramallah and AL-Bireh district in Palestine during 2001 and 2004 [25]. Furthermore, et al. [26] found that 50% of the 15 dairy samples collected from Cairo, Giza were S. aureus positive with a mean value of 89×103 CFU/g [26] which is higher than the count of S. aureus observed in the current study. Tested 312 cheese samples from different farms in Sweden and found that 88 of those samples were positive for S. aureus [27]. However, Stated that S. aureus was not detected in any of cheese samples collected from markets in Cairo, Egypt [28].
The assessment of food contamination by coliform bacterial species is an indicator of the hygiene level while manipulating food. The highest count of the tested dairy samples was 1.96x103 CFU/g translating effective hygiene during the production and packaging of these samples. The findings of the current study were in agreement with who examined 19 Labneh samples from local market in Jordan and revealed that the mean values of the coliforms in Labneh were less than 10 CFU/g [29]. Another study, who tested Labneh samples collected from Dairy Day farms in Kfarshima in Lebanon, found that the coliforms were not detected in Labneh samples which indicates adequate heat treatment of milk and high hygienic standards of processing and packaging steps [30]. However, [6] reported that the average of Coliform count in branded and unbranded Labneh samples, collected from Jordan, varied between 6.00 to 10.00 and 3.00×102 to 8.00×102 CFU/g, respectively [6]. The average count of coliforms were 9.70×10 CFU/g and 1.95 ×102 CFU/g, respectively in two different studies of Labneh samples in Syria by [31,32], while the counts of the E.coli were 10.00 CFU/g and 1.05×10 CFU/g respectively [30,31], which is higher than the total coliform counts observed in the current study. In contrast, tested 722 samples of dairy products that were collected from different sources in Ramallah and AL-Bireh district and found that the proportion of samples with unacceptable level of coliforms increased from 2001 to 2004 [25]. Furthermore, reported that Enterobacteriaceae was detected in cultured dairy product with high rate amounted to 19.23% (5/26) [33]. Moreover, showed that 9 (30%) of the Labneh samples were positive for Coliforms with a mean value of 1.6×104 CFU [26], which is higher than the total coliform counts observed in the current study. As for the Halloumi cheese samples, the findings of the current study were in agreement with who tested 273 raw and pasteurized cheese from the U.S. and other countries and found that 75 of those samples were positive for Coliforms in concentration above 10 CFU/g [34]. Moreover, tested cheese samples from different farms in Sweden and detected E. coli in 34% of raw milk cheeses and 3% of cheeses made with pasteurized milk [27].
This study has shown that, Enterobacteriacae was identified in 37.5% of packaged Halloumi samples, and in 71.42% of the unpackaged Halloumi samples. Reported that the presence of Enterobacteriaceae was high in soft cheese at rate of 10 out 21 samples (47.62%) compared to other dairy products [33]. E. coli was found in soft cheese at rate of 14.30% (3/21) and this result could be due to high moisture content that enhances the microbial survival and reproduction rate. It’s clearly inferred that the manual production and handling of Labneh will allow wide range of microorganisms contaminate the product [33]. However, reported that the total coliform and E.coli were detected in all samples of fresh cheese and this might be due to the usage of raw milk without heat treatment, on the other hand, the total coliforms were not detected in all samples during the cold storage period [28]. This may be due to increase of the salt in cheese, storage in brine solution, and the role of lactic acid bacteria in the preservation of products and their metabolites on the growth of coliforms [28]. Recently, The Lebanese Minister of Agriculture assured the importance of applying strictly the Legislative decree No. 97/83 which stated that all milk and dairy products should be packaged and delivered to the consumer in tightly closed containers.
The critical limit for yeast and molds in dairy products is 25x103 CFU/g. Molds can produce mycotoxin and at high concentration can deteriorate food and cause food borne illnesses. Their counts in all the tested samples were found below the critical limit set by Lebanese standards. However, [6] reported that the average of yeast and molds count in branded and unbranded Labneh samples varied between 5.00×102 to 10.00×102 and 1.00×106 to 12.00×106 CFU/g, respectively [6]. Furthermore, found that the mean values of yeast and mold count were 1.1 ×107CFU/g in the tested Labneh samples in Jordan [29]. Moreover, determined the mean value of the yeast and mold in traditional turkey Labneh ranged between 1.4 ×106 and 1.3 ×107 CFU/g [35]. All these findings were higher than the yeast and molds counts observed in the current study. However, showed that the average rate of Labneh samples with unacceptable levels of yeast and molds was 2.3% [25]. Furthermore, examined Labneh samples collected from Dairy Day (Kfarshima) in Lebanon and they found that yeast and molds were not detected in fresh Labneh samples due to the hygienic conditions of processing that prevent the recontamination of product by such microorganisms [30]. Investigated that the yeast was present in 70% of the samples with a mean value of 4.2×108 CFU/g [26], which was higher than the yeast and molds counts observed in the current study. Concerning Halloumi cheese samples, reported that molds and yeasts were absent in all fresh cheese samples. However after 3 months, the molds and yeasts were detected in cheese during storage [28]. Moreover, examined 24 samples of Halloumi cheese made from bovine and ovine milk collected from different retail markets in Cyprus and they found that the mean counts of yeast isolated from mature bovine and ovine cheeses were 2.8×105 and 2.3×105 CFU/g, respectively [36]. This finding is higher than the counts of yeast and molds observed in the current study.
The Federation of Veterinarians of Europe (FVE) emphasized that the prudent use of antibiotics is an integral part of good veterinary practice (European Medicine Agency). A big part of responsibility in transferring resistant bacteria also lies in the hands of companies and individuals that deal with food. Educating workers who work in production, processing, manufacturing, preparation and trade with foodstuffs is of the utmost importance. In our study, we tested the antibiotics resistance of 22 S. aureus isolated from the contaminated samples. We found that 31.8% (7 out of 22) were resistant to one or more antibiotics, 68.18% were multidrug resistant (MDR), which means they were resistant to at least 3 groups of antibiotics. In packaged Halloumi cheese, S. aureus isolates were highly resistant to tetracycline and penicillin however in unpackaged Halloumi cheese were highly resistant to penicillin and ampicillin. Studied the resistance of S. aureus to various antibiotics and found that S. aureus was resistant to penicillin, intermediate to erythromycin and sensitive to tetracycline [37]. In contrast with Akindolire et al. (2015) who examined the resistance of S. aureus to various antibiotics isolates from dairy products in North-West province of South Africa found that S. aureus isolates were resistant to penicillin, ampicillin and erythromycin [38]. However, examined 50 dairy products and tested the resistance of 5 isolates of S. aureus to various antibiotics and found that 3 samples out of 5 were susceptible to tetracycline and one isolate was resistant to tetracycline and the remaining one isolate showed intermediate response to tetracycline [39]. In the present study, S. aureus isolates were resistant to multiple classes of antibiotics (penicillin, erythromycin and ampicillin), which can cause serious health problems.
Human pathogen can accumulate antibiotic resistance genes because of the use of antibiotics in Agriculture which impact the treatment of infectious diseases [40]. Indeed, tetracyclines and beta-lactams antibiotics administrated to treat infected humans are administrated to farm animals for disease treatment and prevention. In addition, antibiotics are used in many non-EU countries in sub-therapeutic doses to stimulate animal growth [40]. Kyselková and collaborators have shown in 2015 that animals, in conventional dairy farms, harbor tetracycline resistant genes (TC-r genes) from their early age. These TC-resistant bacteria colonizing the intestine have been acquired from both their mothers and the farm environment [40].
In general, S. aureus exhibits some special features which are not found in other relevant bacteria. This bacterium is capable of expressing a variety of virulence factors and thus considered medically relevant when encountered in dairy products. S. aureus continues to demonstrate the ability to develop and expand resistance to a wide range of antimicrobial drugs. Susceptible populations of bacteria may become resistant to antimicrobial agents through mutation and selection or by the acquisition of new genetic material from other resistant organisms through transformation, transduction and conjugation [39].
Moreover, the high percentage of resistance to these drugs may be attributed to their frequent use in the treatment of various human diseases in Lebanon. In addition, another reason could be excessive or indiscriminate use of these antimicrobial drugs in veterinary medicine. However, the high susceptibility of isolate to these drugs may be due to the fact that these antimicrobials are not used anymore as either therapeutic antimicrobials in veterinary medicine or as growth promoters in conventional animal fattening [36].
Conclusion
In general, the findings of the current study revealed that the microbial load for the collected dairy samples were considerably within the range of Lebanese standards. The unpackaged samples of Labneh and Halloumi cheese were 9.7 folds more contaminated (in term of total bacterial count) compared to the packaged samples. Limitations in the current study include the small sample size due to limited resources. Currently, more samples are being assessed for microbial contamination in order to provide more accurate results.
The antibiotic resistance of S. aureus and E. coli, in the analyzed samples, was studied revealing that the isolated E.coli showed resistance against tetracycline whereas S. aureus were resistant to penicillin, ampicillin, tetracycline and erythromycin, and in some samples it showed multi drugs resistance. On the basis of data obtained in the present study, conclusion may be drawn that the microbial load and antibiotic resistance in dairy products in Lebanon is increasing. There is a clear need to reduce the contamination of dairy products, and to restrict the use of antibiotics by local farmers in order to avoid the antibiotic resistant crisis. The presence of antibiotic resistant bacteria in dairy products is an alarming concern because it causes serious dangers to public health. Thus, it’s crucial to increase the situational awareness on the importance of uncontrolled use of antimicrobials in order to limit the emergence of drug-resistant bacteria.
Acknowledgments
This study was financially supported by the Lebanese International University.
Authors’ Contributions
Imtithal Sheet and Samer Sakr designed this study, analyzed and interpreted the data and drafted the manuscript with equal contributions at all levels. Nadia Al Hallak did the Laboratory experimental work and data analysis. Ali Al-Khatib supervised this work, analyzed and interpreted the data and revised the manuscript. All authors approved the final version of the manuscript.
References
-
-
- Rodrigues LA, Ortolani MBT and Nero LA. Microbiological quality of yogurt commercialized in Viçosa, Minas Gerais, Brazil. African Journal of Microbiology Research. 2010;4(3):210-213.
- Oliver SP, Jayarao BM and Almedia RA. Food borne pathogens in milk and the dairy environment food safety and public health. Foodborne Pathogens and Disease. 2005;2(2):115-29.
- Dib, H. Qualitative Risk assessment of food sector in Lebanon. Qualeb, Ministry of Trade and Economy, Lebanon. 2008.
- Agu KC, Archibong EJ, Anekwe DC, Ago CA, Okafor AC, and Awah NS. Assessment of bacteria present in yoghurt sold on Awka Metropolis. Journal of Applied Medical Sciences. 2014;2(6):3071-3075.
- Juan LA, Eva R, Susana L, José María L and Margarita M. Antimicrobial Activity of Lactic Acid Bacteria in Dairy Products and Gut: Effect on Pathogens. BioMed Research International. 2015:1-9. https://doi.org/10.1155/2015/584183
- Gharaibeh A. A Comparative study of the microbial, physiochemical and sensory properties of samples of Labneh produced at large (Industrial) scale and small scale. Food Science and Quality Management. 2017;63:1-6.
- Efstathios A and Anna P. Characteristics of major traditional regional cheese varieties of East-Mediterranean countries: a review. Dairy Science Technology. 2008;88(4-5):495-510.
- Papademas P and Robinson RK. Halloumi cheese: The product and its characteristics. International Journal of Dairy Technology. 2007;51(3):98-103.
- Kyoung-Hee C, Heeyoung L, Soomin L, Sejeong Kand Yohan Y. Cheese microbial risk assessments — A Review. Asian-Australasian Journal of Animal Sciences. 2018;29(3):307-314.
- Ledenbach LH & Marshall RT. Microbiological spoilage of dairy products. Compendium of the Microbiological Spoilage of Foods and Beverages. 2009;41-67. doi:10.1007/978-1-4419-0826-1_2.
- Arau´jo V, Pagliares V, Queiroz M, and Freitas-Almeida A. Occurrence of Staphylococcus and enteropathogens in soft cheese commercialized in the city of Rio de Janeiro, Brazil. Journal of Applied Microbiology. 2002;92(6):1172-1177.
- Srinivasan V, Gillespie B, Lewis M, Nguyen L, Headrick S, Schukken Y, et al. Phenotypic and genotypic antimicrobial resistance patterns of Escherichia coli isolated from dairy cows with mastitis. Veterinary Microbiology. 2007;124(3):319-328.
- Haftay T, Netsenet G, Kidane W, Hagos H, Seyfe G, Abrha B, et al. Antimicrobial resistance profile of E. coli isolated from raw cow milk and fresh fruit juice in Mekelle, Tigray, Ethiopia. Veterinary Medicine International. 2018(1):1-7.
- Miranda JM, Mondragón A, Vázquez BI, Fente CA, Cepeda A, & Franco Microbiological quality and antimicrobial resistance of Escherichia coli and Staphylococcus aureus isolated from conventional and organic “Arzúa-Ulloa” cheese. CyTA – Journal of Food. 2009;7(2):103-110.
- Dhruba JS, Irani M, Najam S, Virendra R, Parshant K. and Sanjit, D. Antibiotics in agriculture: Use and impact. International Journal of Environmental Protection and Policy. 2018;4 (1):4-19.
- Manyi-Loh, C, Mamphweli S, Meyer E, Okoh A. Antibiotic use in agriculture and its consequential resistance in environmental sources: Potential public health implications. Molecules. 2018;23(4):795.
- Saleh I, Zouhairi O, Alwan N, Hawi A, Barbour E and Harakeh S. Antimicrobial resistance and pathogenicity of Escherichia coli isolated from common dairy products in the Lebanon. Annals of Tropical Medicine and Parasitology. 2009;103(1):39-52.
- Maturin L, Peeler JT, Jackson GJ, Merker R, Bandler R. FDA Bacteriological Analytical Manual, 8th U.S. Food and Drug Administration. 1998. http://www.cfsan.fda.gov/∼ebam/bam-3.html.
- Garbaj AM, Awad EM and Eldaghayes IM. Enterohemorrhagic Escherichia coli O157 in milk and dairy products from Libya: Isolation and molecular identification by partial sequencing of 16S rDNA. Veterinary World. 2016;9(11):1184-1189.
- El-Ansary MA. Assessment of microbiological quality of yoghurt sold in El-Behera Governorate. Alexandria Journal of Veterinary Sciences. 2014;43(1):52-57.
- Matthew A. Performance standards for antimicrobial disk susceptibility tests. Document M2-A9 (10th Edition). Clinical and Laboratory Standards Institute (CLSI). 2008.
- Revision A, Arlington VA. Association of Official Analytical Chemists, Official Method of Analysis, 15th ed., AOAC. Food and Drug Administration, FDA. Bacteriological Analytical Manual. 1990.
- Harakeh S, Yassine H, Gharios M, Barbour E, Hajjar S, El-Fadel M, et al. Isolation, molecular characterization and antimicrobial resistance patterns of Salmonella and Escherichia coli isolates from meat-based fast food in Lebanon. The Science of the Total Environment. 2005;341(1-3):33-44.
- International Dairy Federation (IDF). Recommendations for the hygienic manufacture of milk and milk based products, appendix A. In: Spoilage and Pathogenic Bacteria in Milk Based Products. International Dairy Federation, Belgium. 1994.
- Al-Khatib I, and Al-Mitwalli S. Microbiological quality and sample collection policy for dairy products in Ramallah and AL-Birehdistrict, Palestine. Eastern Mediterranean Health Journal. 2009;15(3):709-716.
- Ahmed L, Morgan S, Hafez R and Abdel-All A. Hygienic quality of some fermented milk products. International Journal of Dairy Science. 2014;9(3):63-73.
- Rosengren Å, Fabricius A, Guss B, Sylvén S and Lindqvist R. Occurrence of foodborne pathogens and characterization of Staphylococcus aureus in cheese produced on farm-dairies. International Journal of Food Microbiology. 2010;144(2):263-269.
- Elgaml N, Moussa M and Saleh A. Comparison of the properties of Halloumi cheese made from goat milk, cow milk and their mixture. Journal of Sustainable Agricultural Sciences. 2017;43(2):77-87.
- Yamani MI and Abu-Jaber MM. Yeast flora of Labneh produced by in-bag straining of cow milk set yogurt. Journal of Dairy Science. 1994;77:3558-3564.
- Al-Kadamany E, Khattar M, Haddad T and Toufeili I. Estimation of shelf-life of concentrated yogurt by monitoring selected microbiological and physiochemical changes during storage. LWT - Food Science and Technology. 2003;36(4):407-414.
- Alshehabi M, Hamed F and Sayah A. Effect of the type of the starter and inoculation rate on the characteristics Labneh made in a direct method compared with the traditional method, Syrian Journal of Agriculture Research. 2015;2(2):55-67.
- Harfoush M. A study of some chemical and microbial properties of labneh (concentrated yogurt) made in the Syrian Coastal Region. Tishreen University Journal for Research and Scientific Studies- Biological Sciences Series. 2011;33(3):54-70.
- El-Ziney M. Evaluation of microbiological quality and safety of milk and dairy products with reference to European and Gulf standards. Food and Public Health. 2018;8(2):47-56.
- Trmčić A, Chauhan K, Kent DJ, Ralyea RD, Martin NH, Boor KJ, et al. Coliform detection in cheese is associated with specific cheese characteristics, but no association was found with pathogen detection. Journal of Dairy Science. 2016;99(8):6105-6120.
- Somer VF and Gulden BK. Microbiological, physiological properties and biogenic amine contents of the strained yogurts from Turkish local markets. African Journal of Biotechnology. 2012;11(78):14338-14343.
- Papademas P, Robinson R. A comparison of the chemical, microbiological and sensory characteristics of bovine and ovine Halloumi cheese. International Dairy Journal. 2000;10(11):761-768.
- Pant R, Nirwal S and Rai N. Prevalence of antibiotic resistant bacteria and analysis of microbial quality of raw milk samples collected from different regions of Dehradun. International Journal of PharmTech Research. 2013;5(2):804-810.
- Akindolire MA, Babalola OO and Ateba CN. Detection of antibiotic resistant Staphylococcus aureus from milk: A public health implication. International Journal of Environmental Research and Public Health. 2015;12(9):10254-10275.
- Sasidharan S, Prema B and Latha Y. Antimicrobial drug resistance of Staphylococcus aureus in dairy products. Asian Pacific Journal of Tropical Biomedicine. 2011;1(1):130-132.
- Kyselková M, Jirout J, Vrchotová N, Schmitt H, Elhottová D. Spread of tetracycline resistance genes at a conventional dairy farm. Frontiers in Microbiology. 2015;6:536
-

