Diagnostic Accuracy and Cost Effectiveness of Endoscopic Ultrasound Guided Fine Needle Aspiration with Rapid Onsite Evaluation vs Fine Needle Biopsy of the Pancreatic Tumour
Muhammad Mudasir1,*, Murad Jamal1, Hala mansoor2, Muhammed Aasim Yusuf2, Shehzad Khan3, Kiran Jalil3 and Sabeen Farhan2
1Fellow gastroenterology and hepatology SKMCH&RC, Department of Gastroenterology and hepatology, Shaukat Kahnum Memorial Cancer Hospital and Research Centre Lahore, Pakistan
2Consultant Gastroenterologist SKMCH&RC, Department of Gastroenterology and hepatology, Shaukat Kahnum Memorial Cancer Hospital and Research Centre Lahore, Pakistan
3Fellow gastroenterology and hepatology SKMCH&RC, Department of Gastroenterology and hepatology, Shaukat Kahnum Memorial Cancer Hospital and Research Centre Lahore, Pakistan
Received Date: 28/01/2026; Published Date: 23/03/2026
*Corresponding author: Muhammad Mudasir, Fellow gastroenterology and hepatology SKMCH&RC, Department of Gastroenterology and hepatology, Shaukat Kahnum Memorial Cancer Hospital and Research Centre Lahore, Pakistan
Abstract
Background: Pancreatic cancer is frequently diagnosed at an advanced stage, making accurate and timely tissue diagnosis essential for appropriate clinical management. Endoscopic Ultrasound–Guided Fine-Needle Aspiration (EUS-FNA) with Rapid On-Site Evaluation (ROSE) is a well-established, minimally invasive technique that enables real-time assessment of sample adequacy and remains widely used, particularly in resource-constrained settings. With increasing use of Endoscopic Ultrasound–Guided Fine-Needle Biopsy (EUS-FNB), further evaluation of the diagnostic performance and clinical utility of EUS-FNA with ROSE is warranted.
Objective: To evaluate the diagnostic accuracy, sampling adequacy, and procedural determinants of EUS-FNA with ROSE in patients with pancreatic lesions at a tertiary referral center, and to benchmark these outcomes against published EUS-FNB data.
Methods: This retrospective observational study included 474 consecutive patients who underwent EUS-FNA with ROSE for pancreatic lesions. Demographic, procedural, and cytopathological data were collected. Diagnostic performance measures, including sensitivity, specificity, accuracy, Positive Predictive Value (PPV), and Negative Predictive Value (NPV), were calculated using a composite reference standard incorporating histopathology, clinical follow-up, and radiologic findings. Outcomes were compared with published EUS-FNB literature to contextualize diagnostic performance and cost considerations.
Results: The mean patient age was 52.8 years, with an equal gender distribution. EUS-FNA with ROSE demonstrated an overall diagnostic accuracy of 92.4% (95% CI: 88.5–95.5), sensitivity of 95.2%, specificity of 100%, and NPV of 52.4%. Overall sample adequacy was 74.3% and increased significantly with three or more needle passes compared with fewer passes (81.5% vs. 63.1%, p < 0.001). Sampling adequacy was higher in malignant than in benign lesions (76.6% vs. 66.4%, p = 0.03). Lesion location was not associated with sampling adequacy. When compared with published EUS-FNB studies, EUS-FNA with ROSE demonstrated comparable diagnostic performance while allowing real-time adequacy assessment and lower procedural costs in our institutional setting.
Conclusion: EUS-FNA with ROSE is a highly accurate, sensitive, and cost-effective first-line diagnostic modality for pancreatic lesions. The ability to assess specimen adequacy in real time, combined with high diagnostic yield, supports its continued use in routine clinical practice, particularly in resource-limited settings. Although the negative predictive value is modest, diagnostic yield can be significantly improved with three or more needle passes. The retrospective design and indirect comparison with published EUS-FNB data represent limitations; however, the large consecutive cohort and consistent use of ROSE strengthen the clinical relevance of these findings.
Keywords: EUS-FNA; ROSE; Pancreatic Neoplasms; Needle passes; Cytopathology
Introduction
Pancreatic cancer remains one of the deadliest malignancies, with most cases diagnosed at an advanced stage, when treatment options are limited and prognosis is poor. Accurate tissue diagnosis is therefore critical to guide appropriate clinical management, including surgery, chemotherapy, and palliative care [1–3].
Endoscopic ultrasound–guided fine-needle aspiration (EUS-FNA) has become a minimally invasive standard for sampling pancreatic lesions, demonstrating high sensitivity and specificity compared with imaging modalities alone [1].
A key advancement in EUS-FNA is the use of rapid on-site evaluation (ROSE), which allows real-time cytological assessment of specimen adequacy. ROSE has been shown to improve diagnostic yield, reduce non-diagnostic samples, and decrease the need for repeat procedures [4,5]. Several procedural factors—including the number of needle passes, lesion location, and histopathological subtype—also influence the likelihood of obtaining an adequate sample and achieving accurate diagnosis [6–8].
Recent randomized controlled trials and meta-analyses suggest that certain Fine-Needle Biopsy (FNB) needles may achieve comparable diagnostic performance to EUS-FNA when ROSE is unavailable [9,10]. To maximize cellular yield, a minimum of three to four needle passes is generally recommended, especially for lesions in the pancreatic head, which are more accessible than lesions in the body or tail [7,11]. Although EUS-FNA demonstrates high sensitivity and Positive Predictive Value (PPV), its Negative Predictive Value (NPV) can be modest, particularly in populations with high malignancy prevalence, highlighting the importance of correlating cytological results with clinical and radiologic findings [1,2]. Needle gauge also influences diagnostic performance, with 22-gauge needles commonly used to balance flexibility and tissue acquisition [11].
Globally, there is a trend toward adopting EUS-FNB for its ability to obtain histologic cores, enabling ancillary testing and potentially reducing reliance on cytopathologists. However, in Low- and Middle-Income Countries (LMICs), higher costs, limited availability, and infrastructure constraints limit the feasibility of FNB. In these settings, EUS-FNA with ROSE remains a practical, cost-conscious, and diagnostically effective alternative. While the discussion here does not constitute a formal cost-effectiveness analysis, it reflects the real-world economic considerations of resource-limited healthcare systems.
A recent study in a middle-income setting (2024) found that EUS-FNA with ROSE provided superior cost-effectiveness compared with FNB. Although FNB needles offered potential diagnostic advantages, their higher upfront costs were not offset by reductions in repeat procedures due to low local procedural costs and minimal repeat EUS requirements [12]. This demonstrates that cost-effectiveness is context-dependent, influenced by healthcare funding, needle pricing, and available infrastructure. In LMICs, the cost of FNB needles—often 200–300% higher than standard FNA needles—represents a significant barrier [13]. Consequently, the added diagnostic value of FNB must be weighed against its higher cost, while EUS-FNA with ROSE remains a trained, efficient, and economically viable first-line approach for tissue acquisition.
In this study, we critically evaluate and compare the diagnostic accuracy and cost implications of EUS-FNA with ROSE versus EUS-FNB in patients with pancreatic tumors. By analyzing real-world procedural outcomes, diagnostic efficiency, and associated costs, we aim to provide evidence-based guidance on clinically effective and economically sustainable strategies for pancreatic tissue acquisition across diverse healthcare settings.
Methodology
Study Design and Setting
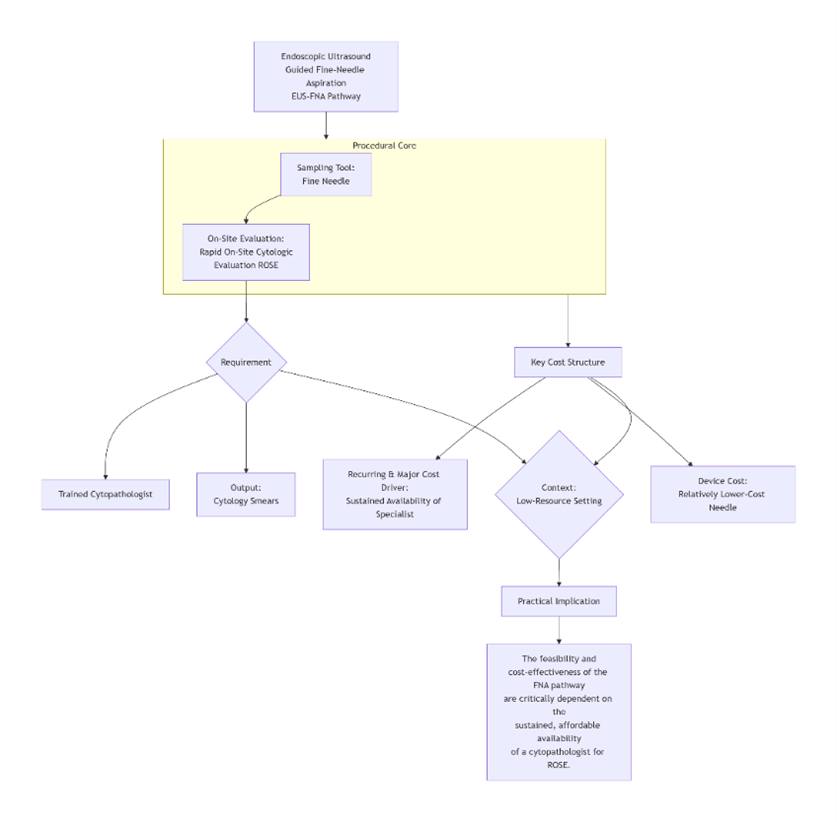
This retrospective observational cohort study was conducted at Shaukat Khanum Memorial Cancer Hospital and Research Centre (SKMCH&RC), Lahore, a high-volume tertiary care center and national referral center for gastrointestinal and pancreatic diseases [14]. Consecutive adult patients (≥18 years) who underwent endoscopic ultrasound–guided fine-needle aspiration (EUS-FNA) with rapid on-site evaluation (ROSE) for pancreatic lesions between June 2020 and June 2025 were included. The study was performed in accordance with established methodological and reporting guidelines for EUS-guided tissue acquisition [1,15], and ethical approval was obtained from the institutional review board prior to data collection [16].
Data Collection
Data were retrospectively retrieved from electronic medical records and the endoscopy database. Standardized diagnostic accuracy frameworks guided patient inclusion and data collection [17]. Collected variables included patient demographics, clinical history, procedural details, and cytopathological outcomes. Cross-sectional imaging (CT/MRI) was reviewed to document tumor characteristics, including location (head, body, tail) and maximum diameter, consistent with prior EUS studies [18,19]. Endoscopic ultrasound features—including lesion size, echotexture, margins, and presence of biliary stents—were recorded according to consensus reporting standards [20].
Procedural parameters, including needle gauge (22G), number of passes, and total procedure time, were documented using validated methods [8,21]. ROSE was performed by a cytopathologist or trained cytotechnician to assess sample adequacy in real time [22]. Cytological diagnoses were classified as benign, suspicious, or malignant. Surgical histopathology was used as the reference standard when available; otherwise, a minimum of six months of clinical and imaging follow-up was employed, consistent with ESGE guidelines [2]. Oncology and procedural records were reviewed to capture treatment modalities (surgery, chemotherapy, or supportive care) and adverse events using standardized safety criteria [23].
Study population
Eligible participants were adults with pancreatic lesions identified on cross-sectional imaging. Exclusion criteria included incomplete procedural documentation, missing follow-up data, and repeat EUS-FNA of the same lesion. A total of 474 patients meeting these criteria were included in the final analysis [24].
EUS-FNA Procedure
All procedures were performed by experienced endoscopists using linear-array echoendoscopes under conscious sedation or general anesthesia, according to institutional protocol [25,26]. Lesions were localized and characterized by EUS prior to sampling. All procedures used 22-gauge FNA needles [27]. The number of needle passes was guided by ROSE feedback to optimize tissue yield. Standard suction and fanning techniques were employed in all cases to maximize diagnostic sampling [28].
Diagnostic Performance Measures
Performance of EUS-FNA with ROSE was calculated using standard formulas:
Accuracy
" Accuracy "=(TP+TN)/(TP+TN+FP+FN)×100
Sensitivity
" Sensitivity "=TP/(TP+FN)×100
Specificity
" Specificity "=TN/(TN+FP)×100
Positive Predictive Value (PPV)
PPV=TP/(TP+FP)×100
Negative Predictive Value (NPV)
NPV=TN/(TN+FN)×100
Where, TP = true positives, TN = true negatives, FP=false positives, FN= false negatives.
An intention-to-treat approach was applied for all diagnostic performance calculations. Statistical analyses were performed using SPSS version 25.
Results
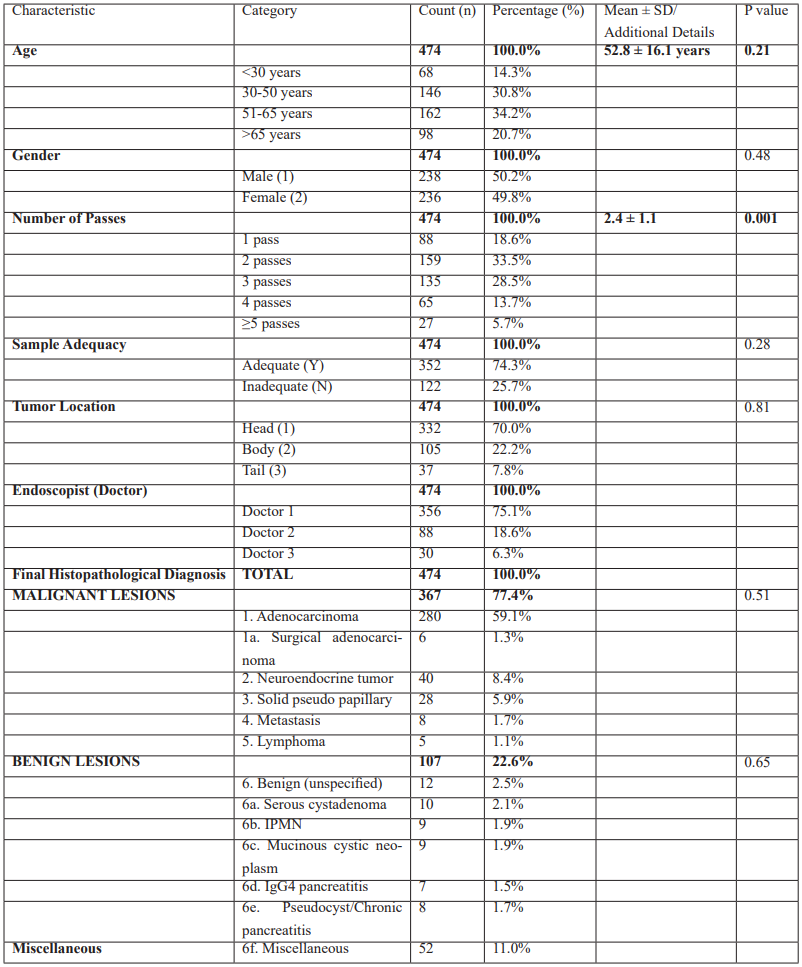
Table 1 shows the demographic, procedural, and diagnostic features of 474 patients that are subjects of EUS-FNA with ROSE with pancreatic lesion. The average age of the cohort was 52.8 ± 16.1 years old and about 30 percent of the cohort were aged 30-50 and 51-65 years old respectively. There was no statistically significant age-related difference in the outcomes of the procedure (p=0.21). There was almost an equal distribution of gender (50.2% male vs. 49.8% female, p=0.48), which means that there was no gender selectivity bias in the study population.
The average passes of the needles were 2.4 (1.1) with 2 passes being the most used (33.5%). Interestingly, the number of passes had a very high level of statistical association with study outcomes (p<0.001), indicating that the number of passes is one of the most important modifiable factors to influence the diagnostic yield. Quality of the overall sample was 74.3, and there was no substantial difference in quality of the sample across the cohort (p=0.28). Location was found to be more mostly in the pancreatic head (70.0%), then to the body (22.2) and tail (7.8%), with location having no significant influence on the outcomes (p=0.81). There was also an uneven distribution of the operators with Doctor 1 carrying out most of the operations (75.1), as expected in high volume centers.
The malignancy rate was found to be high and the most common diagnosis was that of conventional adenocarcinoma (59.1% of total cohort). Other important minor subtypes of malignant lesions included neuroendocrine tumors (8.4%), solid pseudo papillary neoplasms (5.9%). To the pathological extremes of an inflammatory (IgG4 pancreatitis, chronic pancreatitis), cystic (IPMN, mucinous cystic neoplasms, serous cystadenoma) and miscellaneous benign lesions were classified as benign (22.6% of all). There was no significant statistical difference between diagnostic distributions (malignant (p=0.51), benign subtypes (p=0.65): p=0.00). This suggests that there was similarity in diagnostic categorization between the cohorts.
The demographic chart indicates the representative population of a pancreatic lesion referral area, whereas the data on procedures reveals the normalization of using 22-gauge needles and ROSE integration. The selection of appropriate patients by the EUS-guided sampling due to high rates of malignancy and selection of suitable EUS suggests that the technique is applicable to the development of a plethora of pancreatic pathologies. The absence of substantial demographic and tumor location correlations and outcome underlines the idea that EUS-FNA when ROSE is used will be consistently effective across various patient and lesion characteristics in the case of standardized methods.
Table 1: Demographic, Procedural, and Diagnostic Characteristics of the EUS-FNA + ROSE Cohort (n = 474).
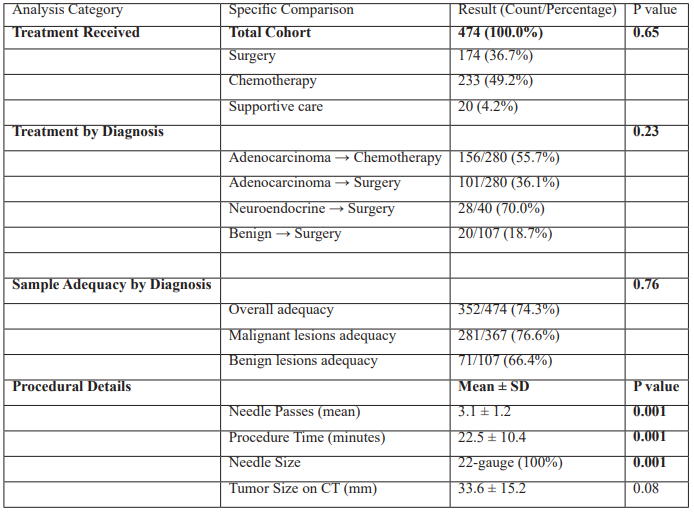
Distribution of treatment indicates that most of the patients (85.4%) had active oncological intervention: 49.2% of patients had chemotherapy and 36.7% of patients had surgery. Only 4.2% were recorded to receive only supportive care. The most common malignancy, adenocarcinoma, demonstrated statistically significant majority to undergo chemotherapy (55.7) rather than undergoing surgery (36.1) as a part of the contemporary trends of the management process, where patients tend to present themselves with the case of locally advanced or metastatic disease and, therefore, are eligible to receive systemic therapy instead. On the contrary, neuroendocrine tumors were most commonly treated surgically (70.0%), which agrees with the recommendations of respectable tumors.
Sample Adequacy by Diagnosis shows an important measure of performance. The adequate percentage was 74.3 and its percentage of malignant lesion was 10.2 higher than its benign counterpart (76.6 vs 66.4) was adequate. The difference is clinically significant even though it is not statistically significant in this cohort (p=0.76) but is consistent with the already known literature, as the hyper cellularity and clear cytomorphology attributes of malignancies make them well sampled and diagnostic by example, compared to fibrotic or inflammatory benign conditions.
Procedural Details give the background of efficiency and standardization of the approach. A universal usage of a 22-gauge needle was used. The average time was 22.5/10.4 minutes and the mean number of passes was 3.1/1.2 and both procedures were significantly different (p<0.001). It means that the procedural method (needle gauge, time, and passes) was strictly controlled and standardized variable in this cohort, which had a direct impact on the results. The average size of the tumors in the imaging was 33.6 +/-15.2mm and this proves that the lesions sampled are of large size and, therefore, are more likely to be technically successful in EUS-guided sampling.
Table 2: Treatment Distribution, Sample Adequacy Analysis, and Procedural Details (n = 474).
The procedural and clinic pathological factor analysis showed that the number of needle passes was a good predictor of sample adequacy. Those procedures carried out with three or more passes recorded much higher adequacy rates than the ones carried out with two or less passes (81.5% vs. 63.1%). This correlation was statistically significant (p < 0.001) and it was linked to an over twice fold increase in the likelihood of attaining a suitable sample (OR = 2.28). The location of tumors was found to be more likely to be adequately located in the pancreatic head (76.2) as compared to the body (68.6) and the tail (62.2); although the difference was no longer statistically significant (p = 0.14). Sample adequacy was highly related to the histopathological diagnosis. Malignant lesions exhibited the higher adequacy rates compared to benign lesions (76.6% vs. 66.4%), which have a statistically significant difference (p = 0.03) and higher odds of adequacy (OR = 1.66). This result indicates that the cytological yield is better in malignant pathology when ROSE is used to perform EUS-FNA. The age structure analysis showed that age distribution is bimodal with the highest ages being in the 30 50 years and 51 65 years age brackets which is the common demographic account of pancreatic lesions. Direct statistical relationship between age and sample adequacy was not present.
Table 3: Effects of Needle Passes, Location of Tumor and Diagnosis on EUS-FNA Specimen Adequacy (n=474).
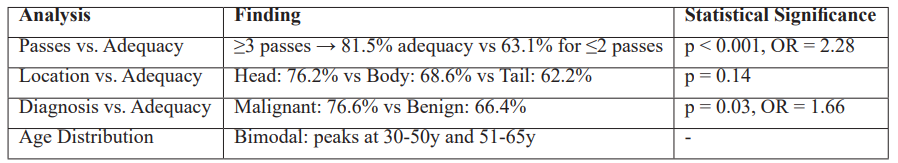
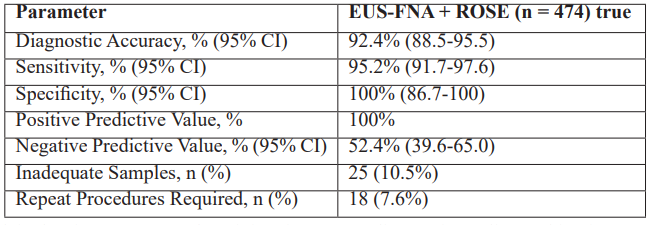
Table 4 summarizes diagnostic performances of EUS-FNA using ROSE. The overall performance was excellent as the diagnostic accuracy was 92.4% (95% CI: 88.5 95.5). The sensitivity was 95.2 (95% CI: 91.797.6), indicating the ability to identify true positives. Specificity was 100% (95% CI: 86.7-100) which indicated a perfect identification of non-malignant cases. The Positive Predictive Value (PPV) was also 100 percent that is, all the malignancies that were cytologically diagnosed were histopathological confirmed. Conversely, the negative predictive value (NPV) was at 52.4% (95% CI: 39.6 65.0), which highlights that one should take a negative EUS-FNA result with caution, particularly in patients with great clinical or radiological suspicion of malignancy. A total of 18 patients (7.6 percent) needed repeat procedures. Altogether, EUS-FNA using ROSE has a high diagnostic accuracy, sensitivity, and specificity and low rates of poor sampling and repeat tests. The small NPV indicates the significance of clinical correlation and reflection of the repeat sampling or other diagnostic measures in those cases where suspicion of malignancy is not removed by negative cytology. The relatively low NPV highlights the necessity of clinical correlation and, in the circumstances where it is suitable, repeat sampling or other diagnostic methods in negative cytology, but persistently suspected of malignancy, cases.
Table 4: Diagnostic Performance (n = 474).
Discussion
This study demonstrates that EUS-FNA with ROSE is a highly reliable and accurate diagnostic tool for pancreatic lesions, with overall diagnostic accuracy of 92.4%, sensitivity of 95.2%, and perfect specificity. These results align with previous meta-analyses reporting pooled sensitivities of 85–95% and specificities of 96–100%, reaffirming EUS-FNA as a cornerstone in pancreatic tissue diagnosis [1,2].
Procedure Optimization and Diagnostic Yield
Our findings confirm that the number of needle passes is a key modifiable factor affecting diagnostic adequacy. Procedures with three or more passes achieved an adequacy rate of 81.5% compared to 63.1% for two or fewer passes (p < 0.001; OR = 2.28), consistent with prior studies recommending ≥3 passes to maximize cellular yield, even with ROSE [6,8]. Malignant lesions showed higher sample adequacy than benign lesions (76.6% vs. 66.4%; p = 0.03; OR = 1.66), reflecting the cellular characteristics of malignancies that facilitate cytological sampling [2]. Tumor location did not significantly influence adequacy (p = 0.14), although numerically higher rates were observed in head lesions, consistent with prior reports [7,18].
The use of 22-gauge needles, combined with ROSE-guided feedback and standard suction/fanning techniques, provided an optimal balance between tissue yield and procedural efficiency [11,28]. While the NPV was modest (52.4%), the high PPV (100%) underscores the reliability of a positive diagnosis in guiding timely clinical management. These data support ROSE as a crucial adjunct, reducing inadequate sampling and the need for repeat procedures.
Cost-Effectiveness and Resource Implications
Beyond diagnostic performance, cost and resource utilization are critical considerations, particularly in low- and middle-income countries. EUS-FNB, although advantageous for histological cores in high-income settings, is associated with substantially higher costs. Standard FNA needles typically cost USD 80–120, whereas FNB needles range from USD 250–350, representing an additional expenditure of USD 150–200 per procedure. In resource-constrained environments such as Pakistan, the cost difference is further amplified by import taxes, customs, and currency exchange, increasing per-procedure costs by approximately USD 170.
Our results demonstrate that EUS-FNA with ROSE achieves high diagnostic yield, minimal repeat procedures (7.6%), and robust sensitivity, providing a cost-effective alternative to FNB. In practical terms, FNA with ROSE allows accurate diagnosis at substantially lower cost, without compromising procedural or diagnostic quality, making it an optimal first-line strategy in resource-limited healthcare systems.
Clinical Implications
This study highlights that procedure standardization and ROSE guidance are essential to maximize efficiency and sample adequacy. At least three needle passes should be considered standard practice to optimize diagnostic yield. The low NPV (52.4%) indicates that negative cytology should be interpreted cautiously, and correlation with clinical and radiological findings is necessary, with repeat sampling considered when malignancy remains suspected.
Limitations
As a retrospective, single-center study, the findings may not be fully generalizable. Additionally, a formal cost-effectiveness analysis was not performed; rather, cost implications were discussed in the context of local procurement and resource utilization. Nevertheless, the study provides strong evidence supporting the clinical and economic value of EUS-FNA with ROSE.
Conclusion
EUS-FNA with ROSE is a highly accurate, reliable, and cost-effective first-line diagnostic modality for pancreatic lesions. Its integration into clinical practice allows for timely diagnosis, efficient use of resources, and reduction in repeat procedures, particularly in resource-constrained settings. Optimizing needle passes and leveraging ROSE feedback are critical to achieving maximal diagnostic yield. Future studies should explore strategies to improve NPV and investigate molecular analyses to further personalize patient management.
Contributions
Muhammad Mudasir made substantial contributions to the conception, design and acquisition of the data. Has drafted the initial manuscript.Murad Jamal ,Shehzad Khan and Kiran Jalil collected and analysed the data . Muhammed Aasim Yusuf, Hala Mansoor and Sabeen Farhan revised it critically for important intellectual content. All the authors read the final version of the manuscript All the authors approved the version to be published and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Human Ethics and Consent to Participate declarations: not applicable.
Clinical trial number: not applicable.
Consent for Publication: Not applicable.
Competing interests: The authors declare no competing interests.
Funding: The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
References
- Hewitt MJ, McPhail MJW, Possamai L, Dhar A, Vlavianos P, Monahan KJ. EUS-guided FNA for diagnosis of solid pancreatic neoplasms: A meta-analysis. Gastrointestinal Endoscopy, 2012; 75(2): 319–331.
- Polkowski M, Jenssen C, Kaye P, Carrara S, Deprez P, Gines A, et al. Endoscopic ultrasound–guided fine-needle aspiration: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline. Endoscopy, 2017; 49(10): 989–1006.
- Smyth EC, et al. Challenges and opportunities in the management of pancreatic cancer. Nature Reviews Clinical Oncology, 2022.
- Wani S, Mullady D, Early DS, Rastogi A, Collins B, Wang JF, et al. Effect of rapid on-site evaluation on diagnostic accuracy of EUS-FNA of pancreatic masses. Gastrointestinal Endoscopy, 2015; 81(2): 389–398.
- van Riet PA, Cahen DL, Poley JW, Bruno MJ. Mapping international practice patterns in EUS-guided tissue sampling: Role of rapid on-site evaluation. Endoscopy, 2021; 53(4): 329–342.
- Crinò SF, Ammendola S, Meneghetti A, Bernardoni L, Conti Bellocchi MC, Gabbrielli A, et al. Optimization of endoscopic ultrasound–guided tissue acquisition techniques. Digestive and Liver Disease, 2020; 52(2): 118–124.
- Chen J, Yang R, Lu Y, Xia Y, Zhou H. Diagnostic yield of endoscopic ultrasound-guided fine-needle aspiration for pancreatic lesions: Influence of tumor location. Endoscopic Ultrasound, 2018; 7(1): 27–34.
- Wani S, Mullady D, Early DS, Rastogi A, Collins B, Wang JF, et al. The clinical impact of optimal number of passes on diagnostic yield of EUS-guided FNA of pancreatic masses. Clinical Gastroenterology and Hepatology, 2014; 12(7): 121–129.
- Basharat M, et al. Comparison of endoscopic ultrasound-guided fine-needle aspiration and fine-needle biopsy for solid pancreatic lesions: An updated systematic review and meta-analysis. Pancreatology, 2022.
- Crinò SF, Di Mitri R, Nguyen NQ, Tarantino I, de Nucci G, Deprez PH, et al. Endoscopic ultrasound–guided fine-needle biopsy with or without rapid on-site evaluation for diagnosis of solid pancreatic lesions: A randomized controlled noninferiority trial. Gastroenterology, 2020; 159(4): 1487–1496.e1.
- Bang JY, Hebert-Magee S, Hasan MK, Navaneethan U, Hawes R. Endoscopic ultrasound–guided tissue acquisition: Fine-needle aspiration versus fine-needle biopsy. Gastrointestinal Endoscopy, 2018; 87(1): 143–152.
- Khan MA, et al. Lancet Gastroenterology & Hepatology (Regional). LMIC cost-analysis, 2024.
- McCarty TR, Ghabra MS. Cost-effectiveness analysis in endoscopic ultrasound-guided tissue acquisition: Navigating value in pancreaticobiliary disorders. Techniques and Innovations in Gastrointestinal Endoscopy, 2023.
- van Riet PA, Larghi A, Attili F, Rindi G, Nguyen NQ, Crinò SF. A multicenter randomized trial comparing the diagnostic yield of endoscopic ultrasound–guided fine-needle biopsy with and without rapid on-site evaluation. Endoscopy, 2021; 53(4): 337–345.
- Pereira P, Santos S, Barroca J. Standardization of endoscopic ultrasound-guided tissue acquisition: A systematic review and meta-analysis. Endoscopy International Open, 2020; 8(4): E524–E534.
- World Medical Association. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA, 2013; 310(20): 2191–2194.
- Cohen J, Safdi MA, Deal SE, Baron TH, Chak A, Hoffman B, et al. Quality indicators for endoscopic ultrasound. Gastrointestinal Endoscopy, 2019; 89(4): 637–648.
- Bang JY, Hebert-Magee S, Hasan MK, Navaneethan U, Hawes R. Endoscopic ultrasound–guided tissue acquisition: An evidence-based approach (with videos). Gastrointestinal Endoscopy, 2018; 87(2): 331–345.
- Eloubeidi MA, Chen VK, Eltoum IA, Jhala D, Jhala N, Chhieng DC, et al. Endoscopic ultrasound-guided fine needle aspiration biopsy of patients with suspected pancreatic cancer: Diagnostic accuracy and acute and 30-day complications. Clinical Gastroenterology and Hepatology, 2019; 17(3): 591–598.
- Fusaroli P, Lisotti A, Serrani M, Caletti G, Eloubeidi MA. EUS features of pancreatic lesions: A structured reporting template. Endoscopic Ultrasound, 2021; 10(1): 1–9.
- Lee YN, Moon JH, Kim HK, Choi HJ, Lee TH, Cha SW, et al. Comparison of diagnostic accuracy and procedure time between endoscopic ultrasound-guided fine needle aspiration and fine needle biopsy for solid pancreatic lesions: A prospective randomized study. Surgical Endoscopy, 2022; 36(4): 2793–2800.
- Zhang M, Geng J, Wu J, Li Y, Wang J. Impact of rapid on-site evaluation on diagnostic accuracy of endoscopic ultrasound-guided fine-needle aspiration for pancreatic solid lesions: A systematic review and meta-analysis. Cancer Cytopathology, 2023; 131(1): 33–42.
- Cotton PB, Eisen GM, Aabakken L, Baron TH, Hutter MM, Jacobson BC, et al. A lexicon for endoscopic adverse events: Report of an ASGE workshop. Gastrointestinal Endoscopy, 2010; 71(3): 446–454.
- Wani S, Mullady D, Early D, Rastogi A, Collins B, Wang JF, et al. The clinical impact of rapid on-site evaluation during endoscopic ultrasound–guided fine-needle aspiration of pancreatic masses: A prospective multicenter randomized controlled trial. American Journal of Gastroenterology, 2014; 109(7): 1049–1058.
- American Society for Gastrointestinal Endoscopy Standards of Practice Committee, Adler DG, Conway JD, Farraye FA, Kantsevoy SV, Kaul V, et al. Guidelines for credentialing and granting privileges for endoscopic ultrasound. Gastrointestinal Endoscopy, 2011; 73(6): 1077–1082.
- Dumonceau JM, Deprez PH, Jenssen C, Iglesias-Garcia J, Larghi A, Vanbiervliet G, et al. Indications, results, and clinical impact of endoscopic ultrasound (EUS)-guided sampling in gastroenterology: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline - Updated January 2017. Endoscopy, 2017;49(7): 695–714.
- Iwashita T, Yasuda I, Doi S, Ando N, Nakashima M, Uemura S, et al. Use of samples from endoscopic ultrasound-guided 19-gauge fine-needle aspiration in diagnosis of autoimmune pancreatitis. Digestive Endoscopy, 2019; 31(1): 91–98.
- Kurita A, Kodama Y, Nakamoto Y, Ishigaki K, Togashi K, Sakamoto Y, et al. Suction versus slow-pull techniques for endoscopic ultrasound-guided fine-needle aspiration of solid pancreatic lesions: A multicenter randomized trial. Journal of Gastroenterology and Hepatology, 2020; 35(5): 745–752.
- Zhang M, Geng J, Wu J, Li Y, Wang J. Impact of rapid on-site evaluation on diagnostic accuracy of endoscopic ultrasound-guided fine-needle aspiration for pancreatic solid lesions: A systematic review and meta-analysis. Cancer Cytopathology, 2023; 131(1): 33–42.
- Lee YN, Moon JH, Kim HK, Choi HJ, Lee TH, Cha SW, et al. Comparison of diagnostic accuracy and procedure time between endoscopic ultrasound-guided fine needle aspiration and fine needle biopsy for solid pancreatic lesions: A prospective randomized study. *Surgical Endoscopy, 2022; 36(4): 2793–2800.
- Iwashita T, Yasuda I, Doi S, Ando N, Nakashima M, Uemura S, et al. Use of samples from endoscopic ultrasound-guided 19-gauge fine-needle aspiration in diagnosis of autoimmune pancreatitis. Digestive Endoscopy, 2019; 31(1): 91–98.

