Evaluation of Rational and Irrational Antibiotics Prescribed for Children Getting Treatment in the OPD of a Tertiary Care Hospital, Kharian, Pakistan: Results and Implications
Huma Zareen1, Usama idress1, Javeria Zainab1, Aqna Malik1,2,*, Nimra Zahoor1, Izza Tahir Cheema1 and Sadia Munir1
1Department of Pharmacy, University of Lahore, Lahore
2Department of Pharmacy, University of Chenab, Gujrat
Received Date: 10/10/2025; Published Date: 30/10/2025
*Corresponding author: Dr. Aqna Malik, Department of Pharmacy, University of Chenab, Gujrat; Department of Pharmacy, University of Lahore, Lahore
https://orcid.org/0000-0003-3875-9621
Abstract
Background: The global misuse of antibiotics is leading to the emergence of antibiotic resistance (AMR), reducing the effectiveness of these vital medications. The World Health Organization (WHO) is working to strengthen antimicrobial stewardship programs, improve monitoring of antimicrobial resistance, and decrease unnecessary antibiotic use through the Global Action Plan on AMR. The AWaRe classification of antibiotics was created by WHO to facilitate tools for antibiotic stewardship at local, national, and global levels, aiming to lower the risk of antimicrobial resistance. The AWaRe list categorizes antibiotics into three groups: Access, Watch, and Reserve, based on their clinical importance, with overuse or misuse potentially contributing to antimicrobial resistance.
Aim: This study aimed to determine the percentage of pediatric patients in different hospitals in Gujrat presenting with common infections and to generate statistical data highlighting the ratio of rational to irrational antibiotic prescribing.
Objectives:
- To assess the proportion of rational versus irrational antibiotic prescriptions among pediatric patients visiting the pediatricians at the OPD of the hospital.
- To evaluate the relevance of the AWaRe list of antibiotics in the OPD at the hospital.
Materials and Methodology: A cross-sectional study was conducted from October 2023 to March 2024 in a tertiary care hospital in Kharian district, Pakistan. Prescriptions of pediatric patients of both genders aged 1-17 years who visited the OPD of the tertiary care hospital for minor ailments during the study period, and who were prescribed antibiotics for various bacterial infections, were included. Data collected from prescriptions was compared against major antibiotic guidelines (AWaRe list) recommended by WHO and the American Academy of Pediatrics (AAP), and cross-referenced with Cochrane studies.
Results: Among 200 prescriptions for children aged 1-17 years, data on the total number of infections across various categories, including UTIs, URTIs, and dermatological infections, showed that the highest prevalence was in boys for both genders, especially among toddlers aged 1-3 years. GIT infections made up nearly 50% of the total data. Of 26 prescriptions for fluoroquinolones (either alone or in combination), 23.08% were considered inappropriate. Similarly, 42.03% of cephalosporin prescriptions (either alone or in combination) were deemed inappropriate, based on a total of 69 prescriptions. Additionally, 40.82% of 49 prescriptions containing penicillin antibiotics were classified as irrational. Comparison of prescribed antibiotics with the standard AWaRe list revealed that (0.27±0.4671) antibiotics were in the Access group, which is less than 0.73±0.46 antibiotics in the Watch group.
Keywords: Antibiotics; Irrational use; AWaRe list; Inappropriate
Background
Sir Alexander Fleming introduced the modern era of antibiotics with his 1928 discovery of penicillin. Since then, antibiotics have completely changed how we treat life-threatening infections. During World War II, soldiers effectively managed bacterial illnesses with penicillin. It is now common practice to treat pediatric infections with antibiotics because newborns and children are among the most vulnerable groups to illness. When it comes to medical care for children, antibiotics are vital because infectious diseases are the leading cause of death in countries with limited resources [1]. Acute respiratory tract infections ARTIs, viral fevers, and watery diarrhea make up most pediatric outpatient visits AMR, a result of antibiotic misuse, is one of the greatest dangers to public health on a global scale. Among the most frequent ailments experienced by children, Upper Respiratory Tract Infections (URTIs) rank high. The majority of URTI prescriptions include antibiotics [2], almost 40%. While 10% of all antibiotic prescriptions given for outpatients are for urinary tract infections (UTIs), making them the 2nd most prevalent reason of prescribing antibiotics are prescribed each year. However, the emergence of Antibiotic Resistance (AMR) has become an inevitable reality [3]. Global standards for responsible drug use are often violated through improper prescription, dispensing, and the number of patients who do not follow their prescribed dosage [4]. This improper dosing is especially risky for infectious diseases and chronic conditions affecting children, as it increases morbidity and mortality. Worldwide, irrational antibiotic use remains a major issue, with studies showing that 75% of antibiotic prescriptions are filled incorrectly, compromising safety and effectiveness [5]. Overuse of antibiotics is a major contributor to the rise of resistant bacteria. Pharmaceutical marketing, diagnostic uncertainty, medical personnel's lack of training and experience, and political and economic considerations all contribute to antibiotic overuse [6]. Educational programmes can raise patient and physician awareness and reduce misuse of prescribing [7]. WHO defines rational medicine as giving patients the right medications for the right indications at the right doses for their needs for an adequate amount of time at the lowest possible cost to the patient and society, all while having accurate information? If any of these conditions are not met, pharmacological treatment is unnecessary [8]. AMR in common pathogens is more prevalent in nations that use antibiotics more often, according to 80% of antibiotics are used by the general public, whereas 20% are reserved for hospital use [9,10]. Comprehensive community monitoring of antibiotic usage is one way to guide and control antibiotic overuse and misuse [11]. Antimicrobial Resistance (AMR) and antibiotic usage patterns are being studied by numerous developed nations through large-scale surveillance projects [12]. Since rigorous monitoring is not possible in settings with limited resources, the problem of Antimicrobial Resistance (AMR) has sadly not gotten the same level of attention in developing nations [12]. As a result, there is a dearth of community-based databases that track antibiotic usage and antimicrobial resistance in poor countries. It was important to conduct a study to determine antibiotic use and the association between infectious disease diagnosis and antibiotic prescription because there is currently very little research on AMR in children in Pakistan. Irrational antibiotic prescribing to children under the age of 17 in both the public and private sectors was the primary aim of this investigation. According to Chakraborty, in comparison to WHO guidelines, 98.9% of prescriptions deviated from the recommended standard of care, among which 99.6% of the prescriptions with deviations were written by the junior doctors [13]. Similarly, in his study, Alili reports that among pediatric outpatients with pneumonia, there was a 90% of irrational antibiotic prescribing [14].
Methods
A prospective observational study was carried out in the OPD of a district-level tertiary care hospital (Kharian) from October 2023 to February 2024. Prescriptions of pediatric patients of both genders aged 1-17 years such as Toddlers (1-3), Preschooler (3-5), Child (5-9), Preteen (9-12), teenager (12-17), who have visited the pediatric OPD of the tertiary care hospital of Kharian district for minor ailments during the study duration and were prescribed antibiotics for different bacterial infections were included in the study. The data collected for respiratory infections (URTIs, LRTIs), urinary tract infections (UTIs), dermatological infections, Gastrointestinal Infections (GIT), and enteric fever. 200 prescriptions were collected and assessed and were prescribed with antibiotics. prescriptions were compared against major antibiotic guidelines (Aware list) recommended by the World Health Organization (WHO) and the American Academy of Pediatrics (AAP), and cross-referenced against Cochrane studies. The sample size was calculated by using the Cochran formula for an unknown population.
Data collection was done in the presence and assistance of 2 experienced, clinically oriented pharmacists and one pediatrician. The data is collected from patients’ medical records. The data collected included demographics of children, physician-assigned diagnosis, numbers of medications prescribed, and all the details regarding the prescribed antibiotics (e.g., name, dose, duration, route of administration, frequency).
Statistical analysis: The analysis was performed using Microsoft Excel, GraphPad 10.6.1. Frequency, percentage, mean, and standard deviation were computed as descriptive statistics.
Results and Discussion
Among 200 children of both age groups who were prescribed antibiotics, (35% were girls and 65% were boys) belong to different age groups, with maximum prescriptions were noted in Toddlers (1-3), 36.5%, Preschoolers (3-5), 21.5%, Children (6-9), 20.5%, Preteens (9-12), 12%, teenagers (12-17), 9.5%. 34%.
Table 1: Number of infections spread among different age groups of pediatric patients.
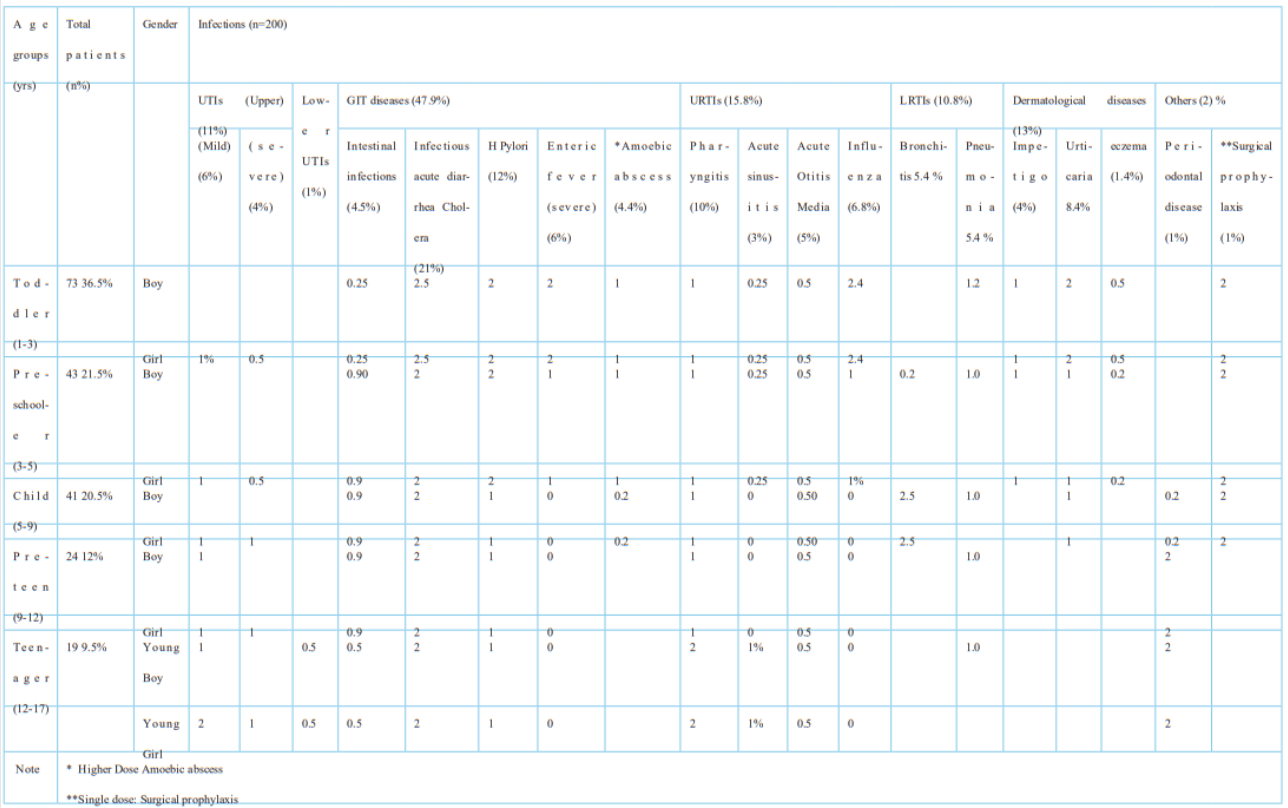
Total number of Infections according to prescription data:
Data collected about the total number of infections of different categories like UTIs, URTIs and dermatological infections, among participants revealed that the highest number of infections were seen in boys among both genders and Toddlers aged 1-3 years. Overall, GIT infections were nearly half of all data and were observed in all age groups of pediatric patients.
Table 2: Gender wise comparison of different age groups of pediatrics with incidence of rational and irrational prescriptions in the outpatient setting.
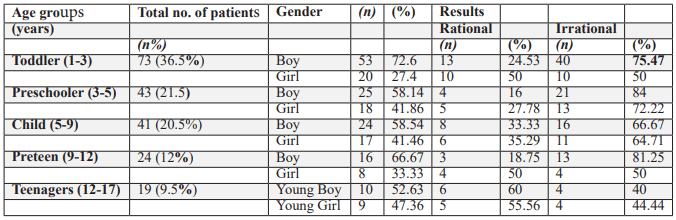
Gender wise comparison of different age groups of pediatrics with frequency of rational and irrational prescribing among partcipants in the OPD explained in Table:1. With maximum irrational prescribing was noted among toddler boys followed by
AMR is mainly driven by OPD prescribing, linked with key factors such as overall antibiotic use, the types or classes of antibiotics, dosage and frequency, and public behavior. The irrational and inappropriate use of antibiotics was observed in 5 different pediatric age groups: Toddler (1-3), Preschooler (3-5), Child (5-9), Preteen (9-12), and Teenager (12-17) in the current study. Toddlers were more prone to irrational antibiotic prescribing. The frequency and incidence were noticeably higher in children aged 1 to 5 years, mainly in boys (65%), and the prevalence rate decreased as children grew older.
Table 3: Comparison of different irrational prescribing trends among irrational prescriptions of pediatric patients in the outpatient setting.
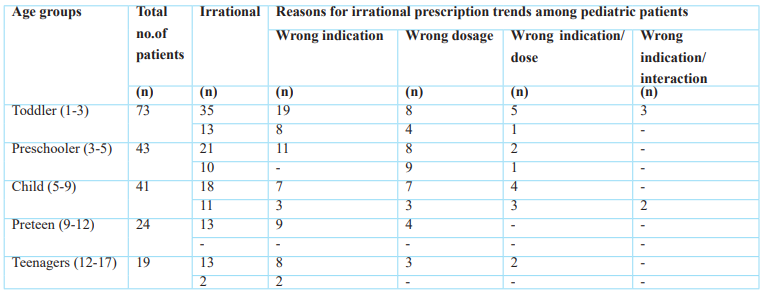
Table 3a: Prescribed antibiotics groups (alone or combination) and their status of appropriate or inappropriate prescribing.
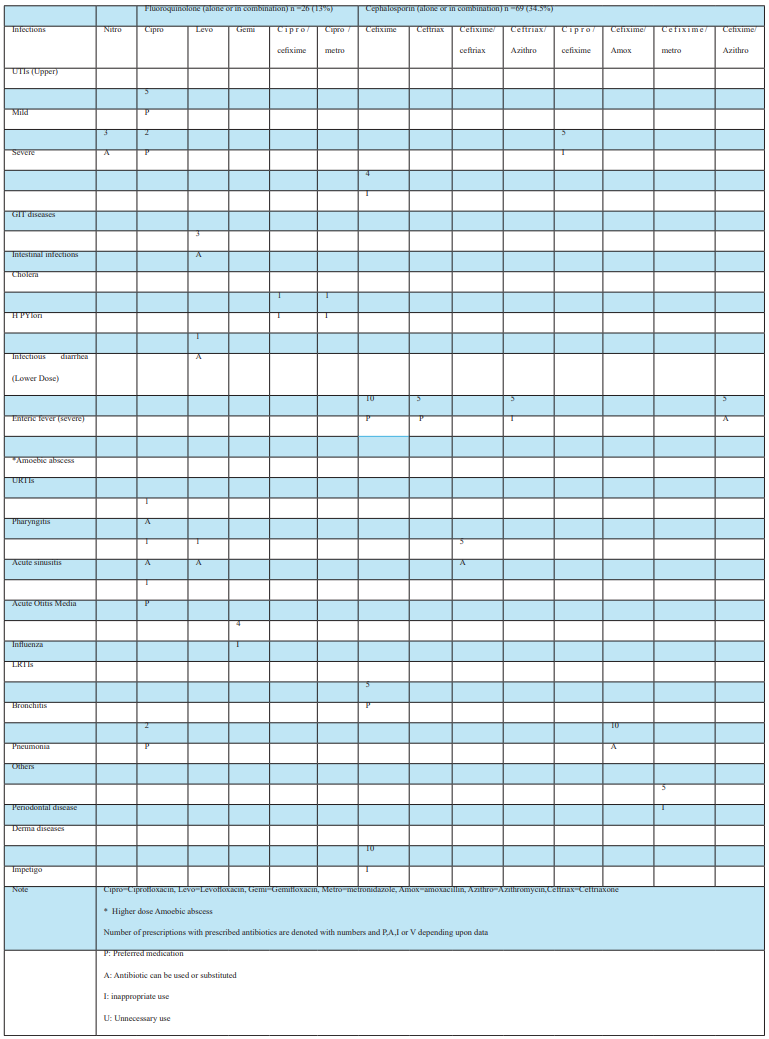
Table 3b: Prescribed antibiotics groups (alone or combination) and their status of appropriate or inappropriate prescribing.

Table 3c: Statistical Analysis of appropriate or inappropriate prescribing of antibiotic groups (alone or in combination).
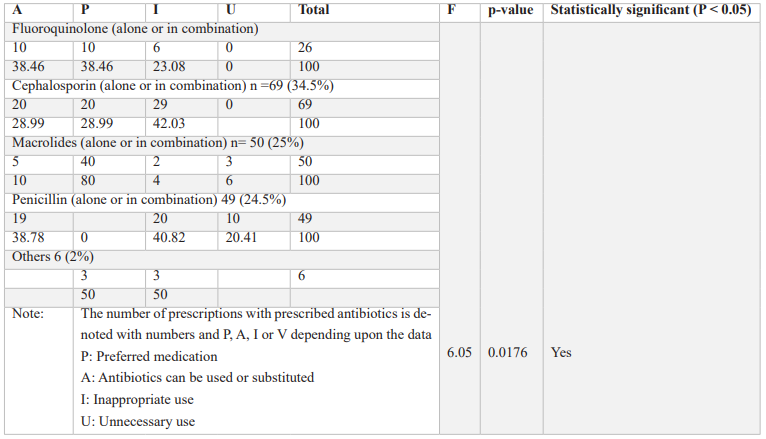
Different antibiotics (either alone or in combination), which were prescribed for various bacterial infections, are listed in Table 3 (a) and Table (b). After complete observation of prescriptions obtained from participants, Antibiotics in different groups were categorized as P: Preferred medication, A: Antibiotics can be used or substituted, I: Inappropriate use, U: Unnecessary use. Among Fluoroquinolones (alone or in combination), which were prescribed to 26 out of 200 pediatric patients, 10 (38.46%) belonged to group A, while similar numbers of patients were prescribed antibiotics that were according to WHO guidelines, and 6 were prescribed inappropriate antibiotics that were against WHO guidelines. Other antibiotic groups (alone or in combination) were also categorized, and One-way ANOVA was applied. A significant p-value determines a significant difference among different antibiotic groups, and the highest variation was observed in the penicillin antibiotic group due to highly resistant antibiotics in this group.
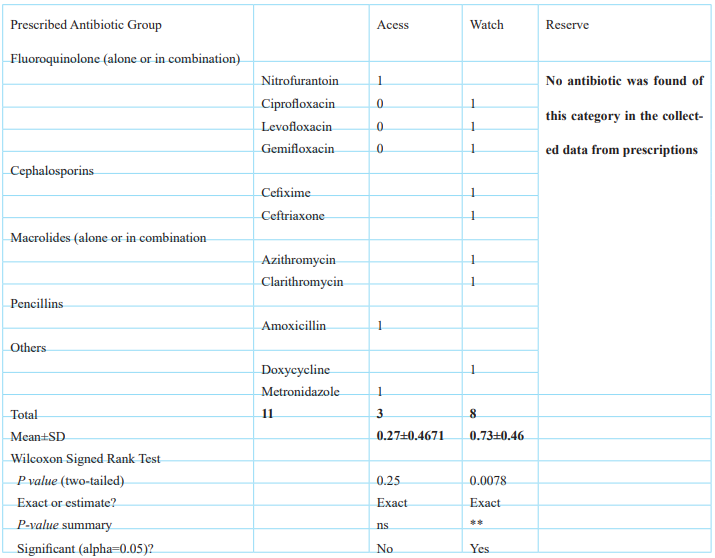
All prescribed antibiotics were cross-checked against the AWARE List of Antibiotics and analyzed for the percentage of antibiotics prescribed irrationally.
In Table 4(a), the prescribed antibiotics for n = 200 patients were categorized according to the WHO AwaRe List. Among the prescribed antibiotics, which were solely given for the infections mentioned earlier, 3 out of 11 (0.27±0.4671) were from the access list, and 8 out of 11 (0.73±0.46) were from the Watch list.
In Table 4(b): Prescribed antibiotics to n=200 patients were categorized as monotherapy, combination therapy, recommended by EML 2023, or not recommended in EML 2023.
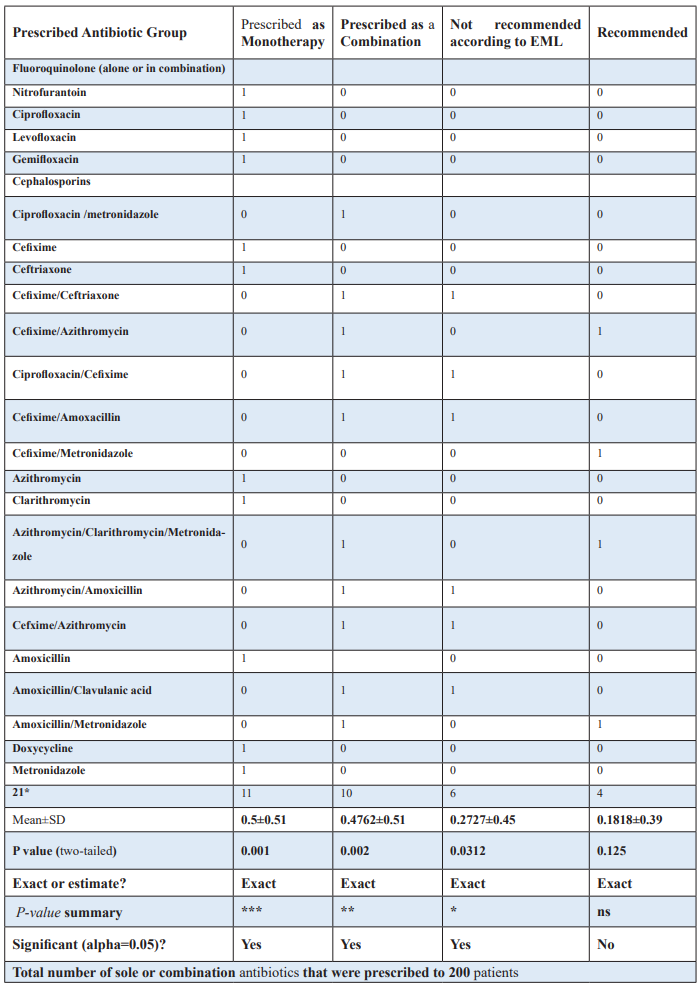
One-sample Wilcoxon test was applied to compare Non-Normal Data regarding prescribed antibiotics, rejecting the hypothesis of rational prescribing in OPD settings of a Tertiary care hospital.
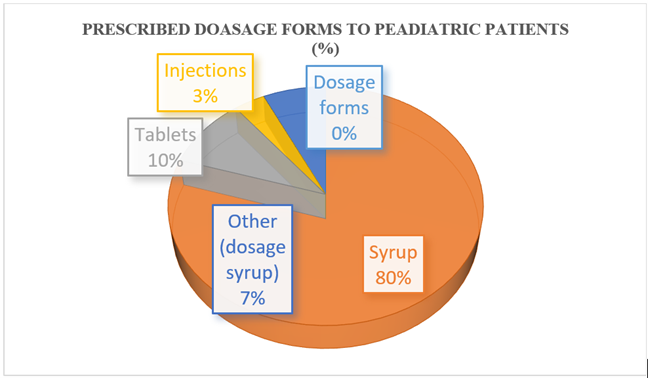
Percentage of prescribed dosage forms of antibiotics:
Among 200 patients, 158 (79%) were prescribed syrups, which are suitable for children.
Adjuvant Medication Prescriptions among Paediatric Patients (n=200):
Among 200 patients, 23 (11.5%) were prescribed solo adjuvant medication, while 154 (77%) were prescribed a combination of adjuvant medications, including anti-histamines, etc. All these medications may prolong antibiotic absorption due to drug-drug interactions.
Discussion
Over 200 paediatric antibiotic prescriptions were reviewed. Irrational prescribing was identified based on trends such as incorrect indication, dosage, interactions, or combinations [15]. Toddlers had the highest rates of inappropriate prescribing, with cephalosporins at 42.03% and penicillin at 40.82% [16]. Incorrect drug choices were observed in RTIs, including pharyngitis (0.5%) and bronchitis (2.5%) [18], often because children did not need antibiotics in these cases. Many paediatric patients were prescribed antibiotics for the first time without a definitive diagnosis, suggesting that clinicians prioritize controlling life-threatening infections while delaying concerns about antibiotic side effects and antimicrobial resistance (AMR) [18]. Moorthi et al. (2011) concluded that clinicians should avoid prescribing antibiotics in less severe cases to reduce resistance [19]. The main reasons for prescribing antibiotics were diarrhea (43%) and acute upper respiratory tract infections (URTIs) (21%) [20]. Since most cases of diarrhea and URTIs are viral and self-limiting, irrational antibiotic use leads to residues, resistant microbes, and resistance genes that harm humans and the environment. These findings align study results of [21]. The most commonly prescribed antibiotic groups were cephalosporins and penicillins, consistent with findings [22]. These antibiotics are fundamental for treating infections [23] and their higher prescription rates may be due to their broad spectrum, effectiveness, and tolerability in children [24]. Regarding antibiotic combinations, recommendations from EML 2023 should be followed [25] to avoid potential side effects. For example, cefixime and ceftriaxone are both in the same class of antibiotics and may increase the risk or severity of nephrotoxicity when combined with cefuroxime, yet this combination was prescribed to 5 (2.5%) patients [26]. The ceftriaxone/azithromycin combination was deemed safe; azithromycin combined with ceftriaxone can reduce systemic inflammation and offer survival benefits in a murine model of polymicrobial sepsis [27]. However, incorrect instructions of azithromycin administration post-meals were observed in 5 (2,5%) prescriptions, which should be taken on an empty stomach [28]. Cefixime and amoxicillin were prescribed together to 5 (2.5%) children with enteric fever, but this combination is not recommended because of the high risk of promoting AMR; one of the drugs could be substituted or removed. No drug-drug interactions were identified between cefixime and metronidazole. Additionally, the combination of azithromycin with amoxicillin is not recommended and should not be prescribed [29]. The combination of clarithromycin, amoxicillin, and metronidazole was inappropriately prescribed to 1 (0.5%) paediatric patient [30].
Recommendations: Antibiotic Resistance should be documented in every hospital.
Limitations: The appropriateness of the suggested drugs was evaluated devoid of any qualitative considerations. The prescribed drug was unwarranted based on the specific diagnosis or the test findings. So, further study needs to be done to fill these gaps.
References
- Le Doare K, Barker CI, Irwin A, Sharland M. Improving antibiotic prescribing for children in the resource-poor setting. Br J Clin Pharmacol, 2015; 79(3): 446-455. https://doi.org/10.1111/bcp.12320.
- Zhao SR, Griffin MR, Patterson BL, Mace RL, Wyatt D, Zhu Y, et al. Risk Factors for Outpatient Use of Antibiotics in Children with Acute Respiratory Illnesses. South Med J, 2017; 110(3): 172-180. https://doi.org/10.14423/smj.0000000000000622.
- Chardavoyne PC, Kasmire KE. Appropriateness of Antibiotic Prescriptions for Urinary Tract Infections. West J Emerg Med, 21(3): 633-639. https://doi.org/10.5811/westjem.2020.1.45944.
- Sun S, White BD. Pharmacist and prescriber responsibilities for avoiding Prescription Drug Misuse. AMA Journal of Ethics, 2021; 23(6): 471-479.
- Hossain MJ, Jabin N, Ahmmed F, Sultana A, Abdur Rahman SM, Islam MR. Irrational use of antibiotics and factors associated with antibiotic resistance: Findings from a cross-sectional study in Bangladesh. Health Sci Rep, 2023; 6(8): e1465. https://doi.org/10.1002/hsr2.1465
- Adebisi YA. Balancing the risks and benefits of antibiotic use in a globalized world: the ethics of antimicrobial resistance. Globalization and Health, 2023; 19(1): 27 DOI:10.1186/s12992-023-00930-z.
- Soumerai SB, Majumdar S, Lipton HL. Evaluating and improving physician prescribing. Pharmacoepidemiology, 2000; 483-503.
- Sarkar S, Srivastava V, Samajhdar SS, Pattanayak C, Tripathi S. Rational use of antibiotics: An area of concern. Journal of Young Pharmacists, 2022; 14(2): 165.
- Salam MA, Al-Amin MY, Salam MT, Pawar JS, Akhter N, Rabaan AA, et al. Antimicrobial resistance: a growing serious threat for global public health. Healthcare, 2023.
- Kardas P, Devine S, Golembesky A, Roberts C. A systematic review and meta-analysis of misuse of antibiotic therapies in the community. International journal of antimicrobial agents, 2025; 26(2): 106-113.
- Manik M, Rozario E, Hossain S, Ahmed MK, Islam M, Bhuiyan M. The Role of Big Data in Combatting Antibiotic Resistance Predictive Models for Global Surveillance. Nanotechnology Perceptions, 2020; 16(3): 361-378.
- Sulis G, Sayood S, Gandra S. Antimicrobial resistance in low-and middle-income countries: current status and future directions. Expert review of anti-infective therapy, 2022; 20(2): 147-160.
- Chakraborty D, Debnath F, Kanungo S, Mukhopadhyay S, Chakraborty N, Basu R, et al. Rationality of prescriptions by rational use of medicine consensus approach in common respiratory and gastrointestinal infections: an outpatient department based cross-sectional study from India. Tropical Medicine and Infectious Disease, 2023; 8(2): 88.
- Alili-Idrizi E, Dauti M, Malaj L. Irrational prescribing of antibiotics in pediatric outpatients: a need for change. Journal of Pediatric Sciences, 2015; 7.
- Fathima F. The Prescribing Trends and Safety Profile in Paediatrics Rajiv Gandhi University of Health Sciences (India), 2017.
- Pasha SS. A Study on Assessing the Prescribing Pattern of Antibiotics Among Pediatric Patients Rajiv Gandhi University of Health Sciences (India), 2011.
- Kamin W, Seifert G, Zwiauer K, Bonhoeffer J, De Ketelaere V, D'Avino A, et al. Phytotherapy for acute respiratory tract infections in children: a systematically conducted, comprehensive review. Frontiers in Pediatrics, 2025; 13:
- Karageorgos S, Hibberd O, Mullally PJW, Segura-Retana R, Soyer S, Hall D. Antibiotic Use for Common Infections in Pediatric Emergency Departments: A Narrative Review. Antibiotics (Basel), 2023; 12(7). https://doi.org/10.3390/antibiotics12071092.
- Hussein M, Barclay J, Baker M, Wu Y, Thombare VJ, Patil N, et al. A Comparative Review of the Pharmacology of Dalbavancin and Oritavancin for Gram-Positive Infections: Birds of a Feather or Apples and Oranges? Infectious Diseases and Therapy, 2025; 1-26.
- Álvarez-Zapata M, García-Soto M, Salazar-Martínez A, Soria-Guerra R, Portales-Pérez D, Leija-Cuevas C, et al. Synbiotic Effect of Saccharomyces boulardii and Fructans from Agave salmiana on the Modulation of Intestinal Microbiota in Children Under Antibiotics. Probiotics and Antimicrobial Proteins, 1-14.
- Yosef HL, Yohannes M, Diriba R, Legese MH. Multidrug-resistant bacterial profiles of inanimate objects at Zewditu Memorial Hospital, Addis Ababa, Ethiopia. PloS one, 2025; 20(10): e0333361.
- Macy E, McCormick TA, Adams JL, Crawford WW, Nguyen MT, Hoang L, et al. Association between removal of a warning against cephalosporin use in patients with penicillin allergy and antibiotic prescribing. JAMA Network Open, 2021; 4(4): e218367-e218367.
- Organization WH. The selection and use of essential medicines: report of the WHO Expert Committee on Selection and Use of Essential Medicines, 2023 (including the 23rd WHO Model List of Essential Medicines and the 9th WHO Model List of Essential Medicines for Children). World Health Organization, 2024.
- Aricò MO, Valletta E, Caselli D. Appropriate use of antibiotic and principles of antimicrobial stewardship in children. Children, 2023; 10(4): 740.
- Garnier A-S, Drablier G, Briet M, Augusto J-F. Nephrotoxicity of amoxicillin and third-generation cephalosporins: an updated review. Drug Safety, 2023; 46(8): 715-724.
- Ong JJ, Aguirre I, Unemo M, Kong FY, Fairley CK, Hocking JS, et al. Comparison of gastrointestinal side effects from different doses of azithromycin for the treatment of gonorrhoea. Journal of Antimicrobial Chemotherapy, 77(7), 2011-2016.
- Thi TVL, Pham EC, Dang-Nguyen D-T. Evaluation of children's antibiotics use for outpatient pneumonia treatment in Vietnam. Brazilian Journal of Infectious Diseases, 2024; 28(4): 103839.
- Torres A, Kuraieva A, Stone GG, Cilloniz C. Systematic review of ceftaroline fosamil in the management of patients with methicillin-resistant Staphylococcus aureus pneumonia. European Respiratory Review, 2023; 32(170).
- Yehualaw A, Taferre C, Bantie AT, Demsie DG. Appropriateness and pattern of antibiotic prescription in pediatric patients at Adigart General Hospital, Tigray, Ethiopia. BioMed research international, 2021; 2021(1): 6640892.
- Zulfkar Q, Humaira A, Mohd AD, Afshana Q. THE GROWING THREAT OF ANTIBIOTIC RESISTANCE: MECHANISMS, CAUSES, CONSEQUENCES, AND SOLUTIONS. International Journal of Cognitive Neuroscience and Psychology, 2025; 3(3): 28-36.

