The Role of Platelet-Rich Fibrin (PRF) in Periodontal Plastic Surgery: A Contemporary Review of Evidence-Based Applications
Chariklia Neophytou1,*, Maria Dorda2, Aikaterini Balouri2, Panagiota Dimitriadou2, Avra-Maria Neofytou2 and Leonidas Batas1
1Department of Preventive Dentistry, Periodontology and Implant Biology, Dental School, Aristotle University of Thessaloniki, Greece
2Dentist, Dental School, Aristotle University of Thessaloniki, Greece
Received Date: 06/08/2025; Published Date: 10/09/2025
*Corresponding author: Chariklia Neophytou, Department of Preventive Dentistry, Periodontology and Implant Biology, Dental School, Aristotle University of Thessaloniki, Greece
Abstract
Objective: To evaluate current evidence on the clinical and biological applications of platelet-rich fibrin (PRF), including injectable PRF (i-PRF), in periodontal plastic surgery procedures such as gingival phenotype modulation, root coverage, and wound healing optimization.
Materials and Methods: A structured literature review was conducted using data from randomized controlled trials, systematic reviews, and in vitro studies published up to May 2025. Studies were included if they evaluated PRF's role in soft tissue augmentation, particularly in comparison with Connective Tissue Grafts (CTG), and if they reported outcomes such as Gingival Thickness (GT), root coverage, or postoperative healing.
Results: PRF demonstrated consistent benefits in enhancing gingival thickness, supporting root coverage procedures, and improving wound healing. i-PRF combined with microneedling resulted in a 65.0% increase in gingival thickness over six months. When used with Coronally Advanced Flap (CAF) procedures, PRF improved relative root coverage and clinical attachment levels, though CTG remained superior for keratinized tissue gain. Notably, application of PRF to palatal donor sites during CTG harvesting was associated with significantly reduced postoperative pain and accelerated healing, enhancing patient comfort and recovery. The regenerative performance of PRF is also influenced by blood handling protocols, emphasizing the need for standardized centrifugation parameters.
Conclusion: PRF is a versatile and minimally invasive biomaterial that enhances soft tissue regeneration in periodontal plastic surgery. It may serve as an effective adjunct or alternative to CTG in selected clinical scenarios and offers clear advantages in donor site healing. Standardization of preparation protocols and further long-term research are essential to fully define its role in mucogingival therapy.
Keywords: Platelet-rich fibrin; i-PRF; Gingival recession; Periodontal plastic surgery; Connective tissue graft; Microneedling; Growth factors; Wound healing
List of Abbreviations: ACD – Acid Citrate Dextrose; A-PRF – Advanced Platelet-Rich Fibrin; CAF – Coronally Advanced Flap; CAL – Clinical Attachment Level; CHA – Compound Hyaluronic Acid; CM – Collagen Matrix; CRC – Complete Root Coverage; CTG – Connective Tissue Graft; ECM – Extracellular Matrix; EGF – Epidermal Growth Factor; FGG – Free Gingival Graft; GT – Gingival Thickness; H-PRF – Horizontal Platelet-Rich Fibrin; HGFs – Human Gingival Fibroblasts; i-PRF – Injectable Platelet-Rich Fibrin; IGF-1 – Insulin-like Growth Factor-1; KMW – Keratinized Mucosa Width; KTW – Keratinized Tissue Width; L-PRF – Leukocyte- and Platelet-Rich Fibrin; LSCC – Low-Speed Centrifugation Concept; MN – Microneedling; mRC – Mean Root Coverage; PD – Probing Depth; PDGF – Platelet-Derived Growth Factor; PREMs – Patient-Reported Experience Measures; PRF – Platelet-Rich Fibrin; PRP – Platelet-Rich Plasma; PROMs – Patient-Reported Outcome Measures; RCT – Randomized Controlled Trial; RCF – Relative Centrifugal Force; rRC – Relative Root Coverage; SEM – Scanning Electron Microscopy; TCP – Tissue Culture Plastic; TGF-β – Transforming Growth Factor-Beta; VAS – Visual Analog Scale; VEGF – Vascular Endothelial Growth Factor; VISTA – Vestibular Incision Subperiosteal Tunnel Access
Introduction
Periodontal plastic surgery encompasses a range of procedures aimed at correcting or improving the morphology, position, and amount of gingival and alveolar mucosa. A major clinical focus within this field lies in the management of soft tissue defects such as gingival recession and thin periodontal phenotypes, which are not only esthetically concerning but can compromise oral health and long-term treatment outcomes. In recent years, regenerative medicine has introduced innovative approaches to soft tissue augmentation, among which Platelet-Rich Fibrin (PRF) has emerged as a biologically active and autologous biomaterial with considerable clinical promise.
Originally introduced by [1], PRF is a second-generation platelet concentrate derived from centrifuged whole blood, without the use of anticoagulants or exogenous agents. It creates a fibrin matrix rich in platelets, leukocytes, and cytokines, capable of stimulating neovascularization, cellular migration, and extracellular matrix remodeling [2,3]. Compared to platelet-rich plasma (PRP), PRF offers a more sustained release of key growth factors, such as PDGF, TGF-β, VEGF, and IGF-1, thereby better mimicking the natural wound healing environment [4].
Of particular interest is the development of injectable PRF (i-PRF), which enables its use in minimally invasive clinical settings. i-PRF demonstrates enhanced biocompatibility and angiogenic potential, supporting fibroblast proliferation, collagen synthesis, and tissue regeneration [4,5]. Recent in vitro studies have shown that i-PRF significantly increases the expression of PDGF, TGF-β, and collagen type I mRNA levels in gingival fibroblasts, outperforming traditional PRP in several key metrics such as cell migration and gene expression [4].
Parallel to advancements in biologics, procedural innovations such as Microneedling (MN), a technique originally rooted in dermatology, have been repurposed for oral soft tissue therapy. MN creates controlled micro-injuries that activate the body’s repair mechanisms, promoting collagen production and enhancing the efficacy of topically or locally applied regenerative agents. In a pioneering clinical study [6], demonstrated that combining MN with i-PRF injections significantly improved gingival thickness in patients with thin periodontal phenotypes, outperforming i-PRF alone.
Furthermore, a growing body of clinical evidence supports the integration of PRF into Coronally Advanced Flap (CAF) procedures for root coverage in gingival recession cases. Systematic reviews and meta-analyses have revealed that CAF combined with PRF results in improved relative Root Coverage (rRC) and Clinical Attachment Levels (CAL) compared to CAF alone [5]. Although Connective Tissue Grafts (CTGs) remain the gold standard for maximum root coverage and keratinized tissue gain, PRF is favored for its lower patient morbidity, reduced postoperative pain, and autologous nature [7].
Given the evolution in both material science and surgical techniques, this review aims to synthesize current evidence on the use of PRF, particularly i-PRF, in periodontal plastic surgery. Emphasis will be placed on its biological properties, clinical applications in phenotype modulation and gingival recession coverage, and potential advantages over traditional grafting materials. Through a comprehensive analysis of the literature, the clinical efficacy, limitations, and future prospects of PRF-based therapies will be critically examined.
Methodology of the Review
This narrative literature review was conducted to synthesize current clinical and preclinical evidence regarding the use of PRF and i-PRF in periodontal plastic surgery. The focus was on their applications in gingival recession coverage, periodontal phenotype modulation, and soft tissue augmentation. The review followed a structured approach to identify, screen, and analyze relevant high-quality studies, including Randomized Controlled Trials (RCTs), systematic reviews, and in vitro and in vivo investigations.
Search Strategy and Inclusion Criteria
A comprehensive electronic search was performed across PubMed, Cochrane Library, Scopus, and Google Scholar databases for studies published up to May 2025. The search terms included the Boolean string: "PRF" AND ("plastic surgery" OR "root coverage" OR "keratinized tissue" OR "soft tissue"). Only articles published in English in peer-reviewed journals were considered. Additional sources were identified by manual cross-referencing of citations within selected articles.
Studies were included if they met the following criteria:
- Investigated the use of PRF or i-PRF in periodontal plastic or mucogingival surgery.
- Were designed as RCTs, systematic reviews, meta-analyses, or controlled in vitro or in vivo studies.
- Reported quantitative outcomes related to Gingival Thickness (GT), Keratinized Tissue Width (KTW), CAL, relative or Complete Root Coverage (CRC), or Patient-Reported Outcome Measures (PROMs) (e.g., pain, esthetic satisfaction).
- Employed standardized and validated measurement techniques.
Studies were excluded if they:
- Lacked specific clinical endpoints related to soft tissue regeneration.
- Did not use PRF or any of its derivatives as a primary or adjunctive treatment.
- Were case reports, narrative reviews, letters to the editor, or expert opinion pieces without empirical data.
Data Extraction and Analysis
From each selected study, key data were extracted, including study design, sample size, population characteristics, PRF type (e.g., leukocyte- and platelet-rich fibrin [L-PRF], i-PRF), surgical technique (e.g., CAF, Vestibular Incision Subperiosteal Tunnel Access [VISTA], MN), outcome variables (GT, KTW, rRC, CAL), and statistical significance of results.
Where available, additional information was collected on:
- Histological assessments.
- Growth factor release profiles.
- Cellular responses such as fibroblast proliferation, migration, and gene expression.
In vitro studies were reviewed to provide mechanistic insight into PRF’s biological activity, particularly its influence on wound healing dynamics, tissue remodeling, and soft tissue regeneration at the molecular level.
All included studies were appraised critically for methodological rigor, relevance to clinical practice, and reproducibility of findings. Evidence strength was assessed based on study design hierarchy, sample size, follow-up duration, and statistical robustness. Studies with limited sample sizes, high heterogeneity, or absence of a control group were interpreted with appropriate caution.
Results
Biological Mechanisms of PRF
PRF is a second-generation autologous platelet concentrate developed as a biocompatible scaffold for tissue regeneration. Unlike its predecessor, PRP, PRF does not require anticoagulants or bovine thrombin, thereby preserving the physiological coagulation cascade and enhancing biological compatibility [1,2]. The resulting three-dimensional fibrin matrix entraps platelets and leukocytes, enabling a sustained release of key growth factors that modulate various stages of wound healing.
Fibrin Architecture and Growth Factor Release
The fibrin matrix in PRF acts as a reservoir for a variety of cytokines and growth factors, including platelet-derived growth factor (PDGF), transforming growth factor-beta (TGF-β), vascular endothelial growth factor (VEGF), insulin-like growth factor (IGF-1), and epidermal growth factor (EGF). These molecules play crucial roles in chemotaxis, angiogenesis, fibroblast proliferation, and extracellular matrix deposition [3,4].
Notably, the kinetic release profile of growth factors in PRF contrasts with that of PRP. While PRP exhibits a rapid burst release within hours, PRF demonstrates a prolonged release extending over 7 to 10 days [4]. This temporal extension aligns more closely with the natural phases of wound healing, potentially enhancing tissue maturation and vascular integration.
In a comparative in vitro analysis [4], found that i-PRF exhibited significantly greater long-term release of PDGF-AA, PDGF-AB, EGF, and IGF-1 than PRP, despite lower initial concentrations. Interestingly, PRP demonstrated higher total levels of VEGF and TGF-β1 after 10 days. These findings suggest that variations in centrifugation force and duration selectively affect the cellular composition and bioactivity of the resulting concentrates.
Figure 1 illustrates the relative growth factor release profiles of PRP and i-PRF, highlighting differences in their regenerative potential.
Cellular Responses and Molecular Activation
PRF's biological potential is also reflected in its ability to stimulate cell behaviors critical to periodontal regeneration. In vitro studies demonstrate that both PRP and i-PRF exhibit excellent biocompatibility with human gingival fibroblasts (HGFs), but i-PRF induces significantly greater cell migration and upregulation of regenerative gene expression [4]. Specifically, i-PRF enhanced the mRNA levels of TGF-β and PDGF at 3- and 7-days post-seeding, and significantly increased collagen type I (COL1a2) expression compared to both PRP and control tissue culture plastic. This profile implies a stronger pro-regenerative and matrix-remodeling stimulus by i-PRF.
The superior cellular activity of i-PRF may be attributable to the low-speed centrifugation concept (LSCC), which retains higher numbers of leukocytes and platelets in the upper plasma layer [8]. Leukocytes contribute to regenerative processes by secreting additional cytokines and orchestrating cellular recruitment during healing [2].
Fibrin as a Scaffold and Barrier
Beyond its role as a delivery vehicle for growth factors, the PRF matrix itself provides mechanical and spatial support for cellular migration and attachment. Its high-density fibrin network supports capillary ingrowth and may serve as a temporary extracellular matrix, facilitating neotissue organization. Moreover, PRF membranes have demonstrated the capacity to act as biologically active barriers in guided tissue and bone regeneration (GBR/GTR) procedures, mimicking both physical and biological functions of traditional barrier membranes [4].
The addition of hyaluronic acid to PRF, as investigated by [9], further enhanced fibroblast proliferation and migration in vitro, pointing to a synergistic effect between bioactive scaffolds and exogenous biostimulators. These data support the potential for future formulation improvements of PRF-based materials in clinical periodontics.
Blood Collection and Centrifugation Protocols
The biological efficacy of PRF is highly dependent on the specific parameters used during blood collection and centrifugation. PRF is produced by collecting venous blood in tubes without anticoagulants and subjecting it to centrifugation shortly after collection, typically within one minute, to initiate the natural coagulation cascade. The choice of centrifuge, rotor type (fixed-angle vs. horizontal), Relative Centrifugal Force (RCF), spin time, and tube material (e.g., glass or silica-coated) all critically influence the cellular composition and fibrin architecture of the final PRF product [5,10]. For example, i-PRF is commonly obtained by centrifuging whole blood at 700 rpm (approximately 60×g) for 3 minutes, producing a liquid formulation with high concentrations of platelets and leukocytes in the upper plasma layer [4]. LSCC have been shown to preserve more regenerative cells and result in greater long-term growth factor release, while horizontal centrifugation further improves fibrin network uniformity [8,9]. Therefore, strict adherence to validated protocols and consistent centrifuge calibration are essential to ensure the reproducibility and clinical effectiveness of PRF-based therapies.
Clinical Applications of PRF in Periodontal Plastic Surgery
The clinical utility of PRF in periodontal plastic surgery has expanded substantially in the past decade. As a minimally invasive, autologous regenerative agent, PRF has demonstrated benefits in soft tissue augmentation, root coverage procedures, and enhancement of periodontal phenotype. Its application has been particularly promising in cases where conventional grafting materials such as CTGs may be contraindicated due to patient-related or procedural limitations.
1. Phenotype Modulation in Thin Gingiva
GT and KTW are essential components of the periodontal phenotype, which influences the outcomes of surgical interventions and the long-term stability of periodontal health. Individuals with thin periodontal phenotypes are more susceptible to gingival recession and experience less favorable healing following surgical manipulation (Jepsen et al., 2018; [11].
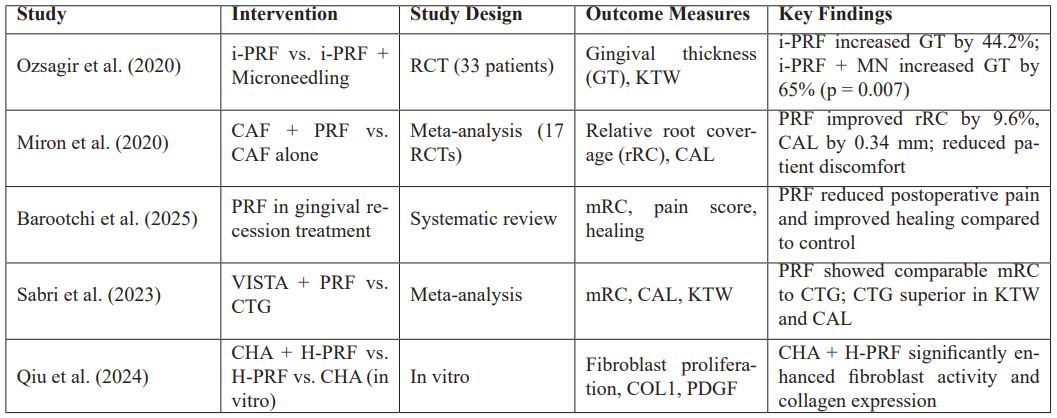
A landmark RCT by Ozsagir et al. (2020) evaluated the efficacy of i-PRF with or without MN in increasing GT among individuals with a thin phenotype. Over a 6-month follow-up, i-PRF alone increased GT by 44.2%, while MN combined with i-PRF resulted in a 65.0% increase. The difference between the two interventions was statistically significant (p = 0.007), favoring the combined approach. Although a minor increase in KTW was observed only in the MN + i-PRF group (~0.17 mm), it was not deemed clinically significant. No adverse events were reported, and recession depth remained stable, indicating both safety and efficacy of the protocol.
These findings suggest that i-PRF, especially when combined with MN, can effectively enhance GT without the need for invasive grafting procedures. Such minimally invasive phenotype modulation is particularly valuable as a preconditioning strategy prior to orthodontic or restorative treatments in patients with fragile soft tissues [12,13].
2. Root Coverage and Gingival Recession Therapy
CAF techniques remain the standard of care for the treatment of Miller Class I and II gingival recession defects. However, the search for less invasive alternatives to CTG has led to the growing use of PRF as a bioactive adjunct. A meta-analysis by [5] encompassing 17 RCTs concluded that CAF combined with PRF significantly improved rRC by 9.6% (p = 0.05) and CAL by +0.34 mm (p < 0.0001) compared to CAF alone. Importantly, the studies reported reduced postoperative discomfort and improved patient satisfaction when PRF was used, especially in lieu of donor grafts.
When PRF was compared directly to CTG, the latter remained superior in achieving maximum root coverage and KTW gain. For instance, CTG was associated with a 3.97% greater rRC and 0.50 mm greater KTW gain (Miron et al., 2020). Nevertheless, the benefits of PRF in reducing donor site morbidity and enhancing soft tissue healing make it an attractive alternative in selected clinical scenarios, particularly when sufficient keratinized tissue is already present [7,14].
Further support for PRF's utility comes from the VISTA technique. In multiple RCTs, VISTA combined with PRF showed root coverage outcomes comparable to those achieved with CTG. Although CTG was favored for increasing KTW and CAL, PRF showed better PROMs and less surgical morbidity [15]. Thus, PRF can be considered a viable alternative to CTG in patients where comfort and minimal invasiveness are prioritized.
3. PRF in Donor Site Healing and Morbidity Reduction
Beyond its use as a primary grafting material, PRF has demonstrated significant benefits when applied to donor sites, particularly the palate, during CTG harvesting. Several studies have reported that PRF application to palatal wounds leads to accelerated healing, reduced postoperative pain, and enhanced epithelialization. These effects are attributed to the fibrin matrix’s ability to act as a biologically active wound dressing, promoting angiogenesis and delivering sustained release of growth factors such as PDGF, TGF-β, and VEGF [4,14]. In RCTs, covering palatal donor sites with PRF membranes significantly reduced Visual Analog Scale (VAS) pain scores and shortened healing times compared to conventional healing by secondary intention. These findings underscore PRF’s value not only as a regenerative agent in recipient sites but also as a minimally invasive tool to mitigate donor site morbidity, thereby improving overall patient satisfaction in periodontal plastic surgery.
4. Wound Healing and Patient-Centered Outcomes
One of the most significant advantages of PRF over traditional grafting methods lies in its ability to enhance soft tissue healing and reduce postoperative morbidity. Several RCTs and systematic reviews have documented significantly lower pain scores, faster healing times, and reduced swelling in PRF-treated groups compared to CTG (Moraschini & Barboza, 2016; [4]. This is attributed to the anti-inflammatory properties and angiogenic effects of growth factors released from the PRF matrix [3].
A systematic review by [14] confirmed these findings, showing that patients treated with PRF experienced statistically lower postoperative discomfort and quicker functional recovery. These outcomes have crucial implications for patient compliance and acceptance of surgical therapy, especially in esthetic zones where repeat procedures may otherwise be necessary.
A comparative overview of clinical and in vitro outcomes from studies using PRF is presented in Table 1.
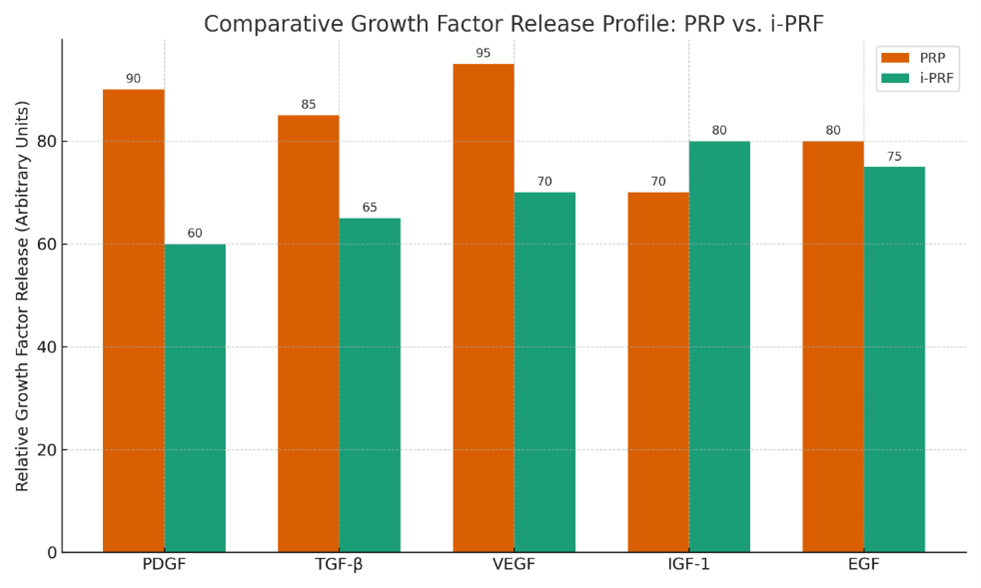
Figure 1: Comparative Growth Factor Release Profile (PRP vs. i-PRF).
A bar chart showing early vs. sustained release of PDGF, TGF-β, VEGF over time.
Table 1: Summary of Clinical and Preclinical Outcomes from Studies Investigating the Use of PRF in Periodontal Plastic Surgery.
Discussion
The use of PRF in periodontal plastic surgery has introduced a biologically driven approach to soft tissue management that complements or, in selected cases, substitutes traditional autologous grafts. Unlike CTGs, which involve additional surgical morbidity and longer postoperative recovery, PRF offers the advantage of being minimally invasive, autologous, and rich in growth factors conducive to tissue regeneration.
One of the central strengths of PRF lies in its multifunctional biological profile. Beyond acting as a scaffold, PRF serves as a delivery system for a broad spectrum of cytokines and growth factors, including PDGF, TGF-β, and VEGF, which orchestrate key events in wound healing, such as fibroblast migration, angiogenesis, and extracellular matrix remodeling. These biologic effects are most pronounced in i-PRF, which has demonstrated enhanced ability to stimulate gene expression related to collagen synthesis and cellular proliferation in vitro.
In addition to its biological activity, PRF's ease of application and autologous origin render it particularly appealing in patients for whom donor site surgery is contraindicated or undesirable. The absence of donor site morbidity, combined with patient-reported reductions in pain and surgical trauma, supports its growing popularity in esthetically driven mucogingival therapy. However, PRF is not a universal replacement for CTG. In clinical scenarios requiring maximal KTW or CRC, CTG remains the superior approach due to its long-term stability and volume retention.
An important and increasingly recognized use of PRF is as a healing adjunct at the palatal donor site during CTG procedures. When a CTG is required, applying PRF to the exposed donor area can significantly accelerate soft tissue healing, reduce inflammation, and improve patient comfort. This approach is especially valuable in cases involving thin palatal mucosa, multiple graft harvests, or patients with low pain tolerance. PRF can serve as a protective biologic dressing, enhancing epithelialization while minimizing postoperative bleeding and discomfort. Its hemostatic and anti-inflammatory properties, coupled with its capacity to promote angiogenesis, make it a reliable aid in reducing donor site morbidity and facilitating faster recovery.
A critical consideration emerging from the literature is the variability in PRF preparation protocols. The final biological composition of PRF is highly dependent on centrifugation speed, time, rotor type, and tube material. These factors directly influence the number of platelets and leukocytes retained, the density of the fibrin matrix, and the release kinetics of bioactive molecules. For example, the LSCC and horizontal centrifugation have been associated with improved cellular yield and growth factor concentration compared to traditional protocols. Inconsistencies in preparation may account for the heterogeneity of clinical outcomes observed across studies, emphasizing the need for standardized, reproducible techniques.
Furthermore, while several in vitro studies have demonstrated the biological plausibility of PRF in enhancing soft tissue regeneration, the translation of these findings to predictable clinical outcomes remains an area requiring further investigation. Many studies are limited by small sample sizes, short follow-up periods, or lack of histologic verification. The heterogeneity in study designs, outcome measures, and PRF formulations complicates the ability to conduct high-level meta-analyses and restricts the generalizability of current evidence.
Emerging formulations such as advanced PRF (A-PRF), horizontal PRF (H-PRF), and PRF combined with bioactive agents like hyaluronic acid (CHA-PRF) show promise in enhancing regenerative outcomes. These innovations highlight a trend toward optimizing both the biological and mechanical properties of PRF, potentially expanding its indications in periodontal and peri-implant soft tissue applications. However, robust clinical trials comparing these novel formulations against established therapies are still lacking.
Overall, while PRF cannot fully replace autogenous grafts in all periodontal plastic procedures, its versatility, biocompatibility, and patient-centered benefits warrant serious consideration. As the field advances, greater emphasis on methodological consistency, long-term outcome data, and patient-reported experiences will be essential to fully define PRF's role in contemporary periodontal therapy.
Limitations
While the clinical and biological data supporting the use of PRF in periodontal plastic surgery are promising, several limitations must be acknowledged. First, variability in PRF preparation protocols, including differences in centrifugation speed, rotor type, temperature control, and tube material, introduces significant heterogeneity across studies (Dohan Ehrenfest et al., 2018). This lack of standardization makes it difficult to draw definitive conclusions about the efficacy of one PRF protocol over another or to compare outcomes directly between studies.
Second, many of the available RCTs suffer from small sample sizes and short follow-up periods, often limited to 6 or 12 months. Long-term stability of soft tissue gains achieved with PRF, especially in the context of gingival recession coverage, remains insufficiently documented (Miron et al., 2020; Barootchi et al., 2025). Similarly, most clinical trials have not included histological analyses to confirm the quality of regenerated tissue, leaving uncertainty regarding the true regenerative capacity of PRF-based therapies.
Another limitation is the absence of consistent patient-reported outcome measures (PROMs) and patient-reported experience measures (PREMs) in many studies. Although PRF is frequently associated with lower morbidity and improved comfort, quantifiable evidence of these benefits is still limited in scope and often reliant on visual analog scales without further qualitative analysis (Moraschini & Barboza, 2016).
Finally, while in vitro studies such as those by [4,9] offer mechanistic insights into PRF’s cellular and molecular effects, translating these findings into predictable clinical outcomes requires further validation through well-controlled in vivo studies and multicenter RCTs.
Conclusion
Platelet-rich fibrin (PRF) represents a biologically active, cost-effective, and minimally invasive approach to soft tissue regeneration in periodontal plastic surgery. Its ability to stimulate angiogenesis, promote fibroblast activity, and enhance extracellular matrix remodeling positions it as a valuable adjunct in the management of gingival recession, thin periodontal phenotype, and postoperative wound healing.
Injectable formulations such as i-PRF further expand the clinical utility of PRF by offering greater ease of application and the potential for synergy with adjunctive procedures like MN. While PRF does not yet replace connective tissue grafts in terms of absolute tissue gains, it offers superior patient comfort, reduced morbidity, and adequate esthetic outcomes, particularly when baseline keratinized tissue is sufficient.
To fully integrate PRF into routine clinical protocols, future research must address existing limitations through standardized preparation protocols, long-term clinical trials, and robust evaluation of patient-centered outcomes. As the evidence base continues to expand, PRF is poised to play an increasingly prominent role in personalized, biologically driven periodontal therapy.
References
- Choukroun, J., Adda, F., Schoeffler, C., & Vervelle, A. (2001). An opportunity in perio-implantology: PRF. Implantodontie, 42, 55–62.
- Miron, R. J., Choukroun, J. (2017). Platelet rich fibrin in regenerative dentistry: Biological background and clinical indications. Wiley-Blackwell.
- Choukroun, J., & Ghanaati, S. (2018). Reduction of relative centrifugation force within injectable PRF (i-PRF) concentrates the cells and growth factors. Platelets, 29(6), 585–590.
- Miron, R. J., Fujioka-Kobayashi, M., Bishara, M., Zhang, Y., Hernandez, M., & Choukroun, J. (2017). Platelet-rich fibrin and soft tissue wound healing: A systematic review. Tissue Engineering Part B: Reviews, 23(1), 83–99.
- Miron, R. J., et al. (2020). Use of platelet-rich fibrin for the treatment of gingival recessions: A systematic review and meta-analysis. Clinical Oral Investigations, 24, 2543–2557.
- Ozsagir, Z. B., Saglam, E., Sen Yilmaz, B., Choukroun, J., & Tunali, M. (2020). Injectable platelet-rich fibrin and microneedling for gingival augmentation in thin periodontal phenotype: A randomized controlled clinical trial. Journal of Clinical Periodontology, 47(4), 489–499.
- Novaes, A. B., & Palioto, D. B. (2019). Platelet-rich fibrin and other autologous preparations in regenerative periodontal and implant therapy. Periodontology 2000, 79(1), 76–89.
- Dohan Ehrenfest, D. M., Pinto, N. R., Pereda, A., Jimbo, R., Corso, M. D., Kang, B. S., & Ghanaati, S. (2018). The impact of the centrifuge characteristics and centrifugation protocols on the cells, growth factors, and fibrin architecture of a leukocyte- and platelet-rich fibrin (L-PRF) clot and membrane. Platelets, 29(2), 171–184.
- Qiu, Y., et al. (2024). Novel approach to soft tissue regeneration: In vitro study of compound hyaluronic acid and horizontal platelet-rich fibrin combination. Journal of Applied Oral Science, 32, e20230294.
- Dohan Ehrenfest, D. M., Rasmusson, L., & Albrektsson, T. (2014). Classification of platelet concentrates: From pure platelet-rich plasma (P-PRP) to leucocyte- and platelet-rich fibrin (L-PRF). Trends in Biotechnology, 27(3), 158–167.
- Müller, H.-P., & Eger, T. (2002). Masticatory mucosa in subjects with different periodontal phenotypes. Journal of Clinical Periodontology, 29(10), 837–842.
- Holmes, J. D., Sulik, W. D., & Osborn, J. (2005). Gingival augmentation techniques to increase keratinized tissue. Pract Proced Aesthet Dent, 17(8), 577–582.
- Morris, M. L., Klein, P. A., & Gapski, R. (2017). Gingival biotype modification using platelet-rich fibrin: A pilot study. International Journal of Periodontics & Restorative Dentistry, 37(3), 441–447.
- Barootchi, S., Tavelli, L., & Wang, H.-L. (2025). Platelet-rich fibrin in the treatment of gingival recessions: A systematic review and meta-analysis of randomized clinical trials. Periodontology 2000, 97(1), 101–121.
- Sabri, H., Samavati-Jame, F., Sarkarat, F., Wang, H.-L., & Zadeh, H. H. (2023). Clinical efficacy of Vestibular Incision Subperiosteal Tunnel Access (VISTA) for treatment of multiple gingival recession defects: A systematic review, meta-analysis and meta-regression. Clinical Oral Investigations, 27(1), 1–15.

