Study Protocol for a Randomized Controlled Trial Assessing Prevention of Metabolic Acidosis in Preterm Neonates by Replacing Sodium Chloride with Sodium Acetate in Parenteral Nutrition (PROTECT Trial)
Adnan Mirza1, Farjam Ahmed Zakai1, Gul Ambreen2, Shabina Ariff1, Kashif Hussain2, Imran Nisar1, Sohail Salat1 and Omar Irfan1,3*
1Department of Pediatrics and Child Health, The Aga Khan University Hospital, Stadium Road, Karachi, Pakistan
2Department of Pharmacy, Aga Khan Hospital, Karachi, Pakistan
3Independent Consultant, Milton, Ontario, Canada
Received Date: 08/04/2025; Published Date: 05/08/2025
*Corresponding author: Dr. Omar Irfan, Department of Pediatrics and Child Health, The Aga Khan University hospital, Stadium Road, Karachi; Independent Consultant, Milton, Ontario, Canada
Abstract
Introduction: Providing balanced dietary support for neonates who are extremely preterm (<28 weeks) and of very low birth weight (VLBW) (<1.5kg) remains a challenge for pediatricians. Parenteral nutrition (PN) can serve as early nutritional support and hence is critical in the overall management. Sodium supplementation through the PN route has mostly been Sodium Chloride (So-C). However, due to the limited excretion capacity of preterm babies’ kidneys, sodium replacement by So-C puts infants at risk of hyperchloremia, linked to the development of metabolic acidosis. Metabolic acidosis is associated with pulmonary hypertension, bronchopulmonary dysplasia (BPD), and intraventricular hemorrhage (IVH). Furthermore, severe renal manifestations, such as renal tubular acidosis and then renal failure, can develop after hyperchloremic acidosis. In patients with metabolic acidosis due to acute chloremia, Sodium Acetate (So-A) can be used to replenish plasma bicarbonate levels and blood pH. This study aims to assess the effectiveness of So-A for preventing/treating metabolic acidosis and associated neonatal comorbidities compared to the So-C group.
Methods and analysis: This is a two-arm, open-label, phase III randomized controlled trial at the Neonatal Intensive Care Unit (NICU) of Aga Khan University Hospital (AKUH). Neonates <33 weeks gestational age who need PN in the first 72 hours of life will be randomized to receive either So-A (intervention) or So-C (control) as part of their PN regimen for a maximum of 28 days. Primary outcomes include the prevalence of metabolic acidosis, which will be measured by blood pH, base excess, and bicarbonate. Secondary outcomes will be the development of neonatal comorbidities, length of ventilation, sodium bicarbonate therapy usage, and weight gain and survival. The trial will recruit 200 neonates (100 per arm), according to power calculations to identify meaningful differences in metabolic acidosis and comorbidities. Data analysis will utilize t-tests, Chi-square tests, logistic regression, and receiver operating characteristics (ROC) curve analysis to identify the best dosing of So-A.
Ethics and dissemination: The Ethical Research Committee of AKUH (ID#2024-6523-29448) has approved the study. The findings will be disseminated in national and international forums and peer-reviewed journals.
Trial registration number: NCT06545565
Background
Despite the recent developments in neonatal care, providing balanced dietary support for neonates who are extremely preterm (<28 weeks) and of very low birth weight (VLBW) (<1.5kg) remains a challenge for pediatricians. During week one of neonatal life, parenteral nutrition (PN) can serve as early nutritional support and hence is critical in the overall management. Due to the prematurity of the neonatal gastrointestinal tract, and having poor sucking and swallowing reflexes, any unrestricted nutrition plan may potentially lead to complications, which can include necrotizing enterocolitis (NEC) [1,2]. Furthermore, in addition to neonatal kidneys restricting the ability to conserve electrolytes, the preterms have a limited renal tubular ability to reabsorb sodium, leading to further sodium loss in the urine [3,4].
Sodium supplementation through the PN route has mostly been Sodium Chloride (So-C) [2]. However, due to the limited excretion capacity of preterm babies’ (28 to less than 32 weeks) kidneys, sodium replacement by So-C puts infants at risk of hyperchloremia [3-10], which is further linked to the development of metabolic acidosis [7,8]. Metabolic acidosis is associated with pulmonary hypertension, bronchopulmonary dysplasia (BPD), and intraventricular hemorrhage (IVH)[11-14], leading to increased requirement of ventilation support and bicarbonate supplementation [7]. Furthermore, severe renal manifestations, such as renal tubular acidosis leading to renal failure, can develop after hyperchloremic acidosis [15-19].
To reduce the risk of hyperchloremic metabolic acidosis in preterms, the 2018 European Society for Paediatric Gastroenterology Hepatology and Nutrition (ESPGHAN) published a guideline that recommends using alternate electrolyte solutions in PN that are chloride-free, such as Sodium Acetate (So-A) or sodium lactate (SL) [20]. In patients with metabolic acidosis due to acute chloremia, So-A can be used to replenish plasma bicarbonate levels and blood pH [21,22]. There is a paucity of trials evaluating the role of So-A as PN, especially in VLBW infants [23,7]. A 2020 randomized controlled trial (RCT) [23] of 52 infants born before 33 weeks gestation with birth weights under 1,301 grams revealed that the So-A group had reduced metabolic acidosis, required bicarbonate supplementation, and significant reduction in the incidence of BPD, compared to So-C. Another RCT [7] from 1997 found that acetate reduced the incidence of hyperchloremia from 77% to 25% and improved base excess levels from day 5 onwards. Given the paucity of evidence for So-A in PN, So-C has been the most suitable choice for mineral support in PN for many Neonatal Intensive Care Units (NICUs) worldwide.
Objectives
- To investigate the clinical efficacy of So-A in preventing and treating metabolic acidosis during the first four weeks of life in early preterm neonates (< 33 weeks of gestational age).
- To investigate the efficacy of So-A for lowering the rate of comorbidities in early preterm neonates (< 33 weeks of gestational age).
- To determine the optimal dosing of So-A for decreasing metabolic acidosis in preterm neonates of < 33 weeks of gestational age who received So-A in PN (Only interventional group).
Methods
Design: This will be an open-label phase III RCT to compare So-A and So-C, given daily as the component of PN over the first 28 days of neonatal life. We will administer the treatments within the first 72 hours of birth.
Setting: The trial will be conducted in the NICU of the Aga Khan University Hospital (AKUH), which is a 700-bed, not-for-profit tertiary care hospital receiving country-wide referrals.
Randomization: Infants will be randomized to one of the two interventions, So-A or the standard So-C therapy, before the commencement of PN. An independent pharmacist at the PN Preparation Unit of AKUH will perform randomization using a computer-generated list upon participant enrolment. Randomization will be performed according to block randomization of four, using sequentially numbered sealed and opaque envelopes.
- The interventional group: This group will receive So-A as neonatal PN.
- Control Group: The control group will receive So-C as neonatal PN.
Selection and Withdrawal of Participants
Study Population and Eligibility Criteria
Inclusion criteria:
- Written informed consent obtained by parents/legal representative before the initiation of PN.
- All the neonates up to 72 hours since birth, who were admitted to the NICU of AKUH and received PN during 28 days of their life.
- Gestational age < 33 weeks
Exclusion criteria:
Infants with the below conditions will be excluded:
- Inborn errors of metabolism.
- Genetic/congenital conditions that affect neurodevelopment or require multiple surgeries.
- Severe metabolic alkalosis in critically ill neonates is defined as a persistent elevation of the serum pH above 7.45, and it also involves a primary increase in serum bicarbonate (HCO3-) concentration > 25mEq/L.
- Severe Hypernatremia in critically ill neonates is defined as persistently high serum sodium levels > 150 mmol/L.
- Severe liver failure.
- Infants with multiple congenital abnormalities.
- Severe perinatal asphyxia.
Subject withdrawal criteria:
Any individual has the right to withdraw from the study at any time (mentioned in informed consent). In case of withdrawal, the data of that individual will only be included for analysis if the stay of the neonate is ≥ 7 days and received PN therapy for ≥ 3 consecutive days during 28 days of neonatal life.
A description of the “stopping rules” or “discontinuation criteria” :
For individual subjects, the criteria for discontinuation of intervention include, when an individual.
1) will not need PN therapy,
2) will have Severe hypernatremia [Severe Hypernatremia in critically ill neonates is defined as persistently high serum sodium levels > 150 mmol/L]
3) metabolic alkalosis [Severe metabolic alkalosis, in critically ill neonates, is defined as a persistent elevation of the serum pH above 7.45, and it also involves a primary increase in serum bicarbonate (HCO3-) concentration > 25mEq/L]
Intervention- Trial Medications:
So-A is not available in a commercially prepared form for injectable use (to add in PN), therefore, it will be prepared by the AKUH pharmacy for this trial.
PN will be formulated in accordance with the ESPEN/ESPGHAN Guidelines on Pediatric Parenteral Nutrition [20], as well as the Clinical Nutrition Review from 2018 [1]. At the time of each infant's enrollment in the study, PN will be tailored to include either the specified intervention of standard amino acids (SA) or specialized amino acids (SC). Additional details regarding the PN formulation will adhere to the unit’s established protocol. The pharmacist responsible for preparing the PN will be informed about the assigned SA or SC and will also be aware of the clinical condition of the infant. PN for both intervention groups will be dispensed in indistinguishable compounded bags. The sodium type used will not be concealed among the list of ingredients. If necessary, the attending medical team will have the authority to modify the composition, such as in instances of severe alkalosis.
Retention and Follow-up
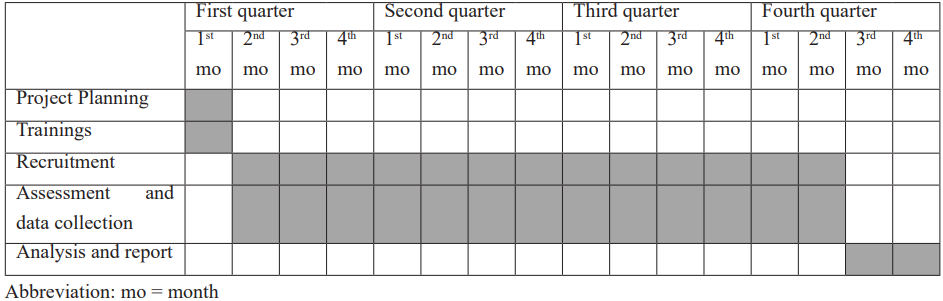
All the subjects will receive the trial medication added to the PN bag till the PN therapy continues during NICU stay up to a maximum of 28 days of life. The participants will undergo daily follow-ups during their hospital stay until discharge (for 28 days) or until death. The study procedure chart and data to be collected at different time points are shown in Table 1 and Table 2, respectively. The Gantt chart of the study plan is reported in Table 3.
Assessment of Efficacy of Intervention
Primary efficacy parameters include metabolic acidosis, i.e., Blood pH, base excess (BE), and bicarb levels. Secondary efficacy parameters include prevention of comorbid conditions (BPD, IVH, PDA, ROP, Sepsis, and jaundice), duration of ventilation, use of plasma volume expanders, inotropes, and sodium bicarbonate therapy, and changes in weight and survival.
Methods and timing for assessing, recording, and analyzing efficacy parameters: The data collected will include maternal and infant demographics, blood investigation reports, and clinical outcome results. The primary outcomes will be assessed from pre-PN on the first day of life (DOL), then 2– 7 DOL, and days 10, 13, 16, 20, 25, and 28 of life irrespective of the duration of PN therapy. Blood sampling techniques will include using either an in-dwelling arterial catheter or capillary sampling. For day 1 of PN therapy samples, the latest blood gas within 6-12 hours of PN initiation will be used. In the case of several samples being taken on the following days, the first recorded value will be taken before the nursing staff's morning shift transfer to standardize the sampling times. The daily doses of So-A in the PN will be adjusted by the PN processing pharmacist according to the patient’s daily serum levels, and the same pharmacist will maintain the data for it.
Routine blood investigation will be performed on 1, 3, 5,7, 10, 13, 16,20,25, and 28 DOL, as per the unit protocol. Patient case notes will be reviewed for outcome data. PN, total fluid, and enteral feed intakes will be charted and recorded daily until 28 days of neonatal life. As PN is not the only source of electrolytes, daily electrolytes (especially sodium and chloride) provided through all the sources (PN, feed, and fluids) will be recorded from the daily case notes.
Assessment of Safety of Intervention:
- Adverse event: Although the patient will be monitored and doses will be tapered according to the serum pH values and serum sodium levels, but patient may have a minimal risk of experiencing a few adverse events due to existing disease conditions and other treatment approaches in NICU.
- Severe Metabolic alkalosis in critically ill neonates is defined as a persistent elevation of the serum pH above 7.45, and it involves a primary increase in serum bicarbonate (HCO3-) concentration > 25mEq/L (at least 3 consecutive readings in 24 hours).
- Severe Hypernatremia is defined as serum sodium levels > 150 mmol/L (at least 3 consecutive readings in 24 hours).
- Serious Adverse Event: There are no serious adverse events associated with the use of So-A, but prolongation of hospital stays and patient death resulting from to adverse effects of this medication will be considered serious adverse effects.
Risk mitigation plan:
Patients hospitalized in the NICU of AKUH will be under the special care and close monitoring of trained nurses. A qualified and trained team of physicians will always be available to address the urgent and critical needs of neonates.
Methods and timing for assessing, recording, and analyzing safety parameters: Both the parameters, i.e., serum PH and electrolytes, will be monitored as part of this protocol as
- The serum pH and base excess (BE) will be recorded from pre-PN on the first day of life (DOL), then 2– 7 DOL, and days 10, 13, 16, 20, 25, and 28 of life, and more frequently on patients with pH>7.45.
- Serum sodium levels: will be assessed on 1, 3, 5,7, 10, 13, 16, 20, 25, and 28 DOL and may be more frequent in patients with > 150 mmol/L.
Follow-up of subjects after adverse events: Within 6-8 hours of having higher serum pH and sodium levels, levels will be reassessed. In case of the above-mentioned effects, doses of So-A will be tapered/adjusted accordingly by the primary physician in direct coordination with the clinical pharmacist.
Reporting and Recording Procedures of Adverse Events: As the mentioned possible adverse effects are not solely related to the use of So-A and are routine intensive care management, they will be reported to the primary care team only and recorded by the investigator.
Reporting and Recording Procedures of Serious Adverse Events: The mentioned possible serious adverse events are not solely related to the use of So-A and are routinely experienced in critically ill and preterm neonates in intensive care units. However, after the confirmation by the primary care team and the principal investigator, these events will be recorded and reported on the board of ERC.
Table 1: Study Procedure Chart.
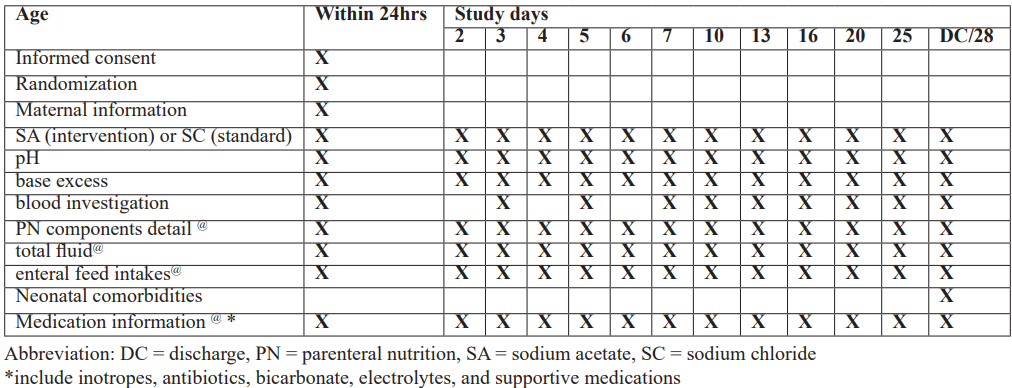
Table 2: Data to be collected at different time points.
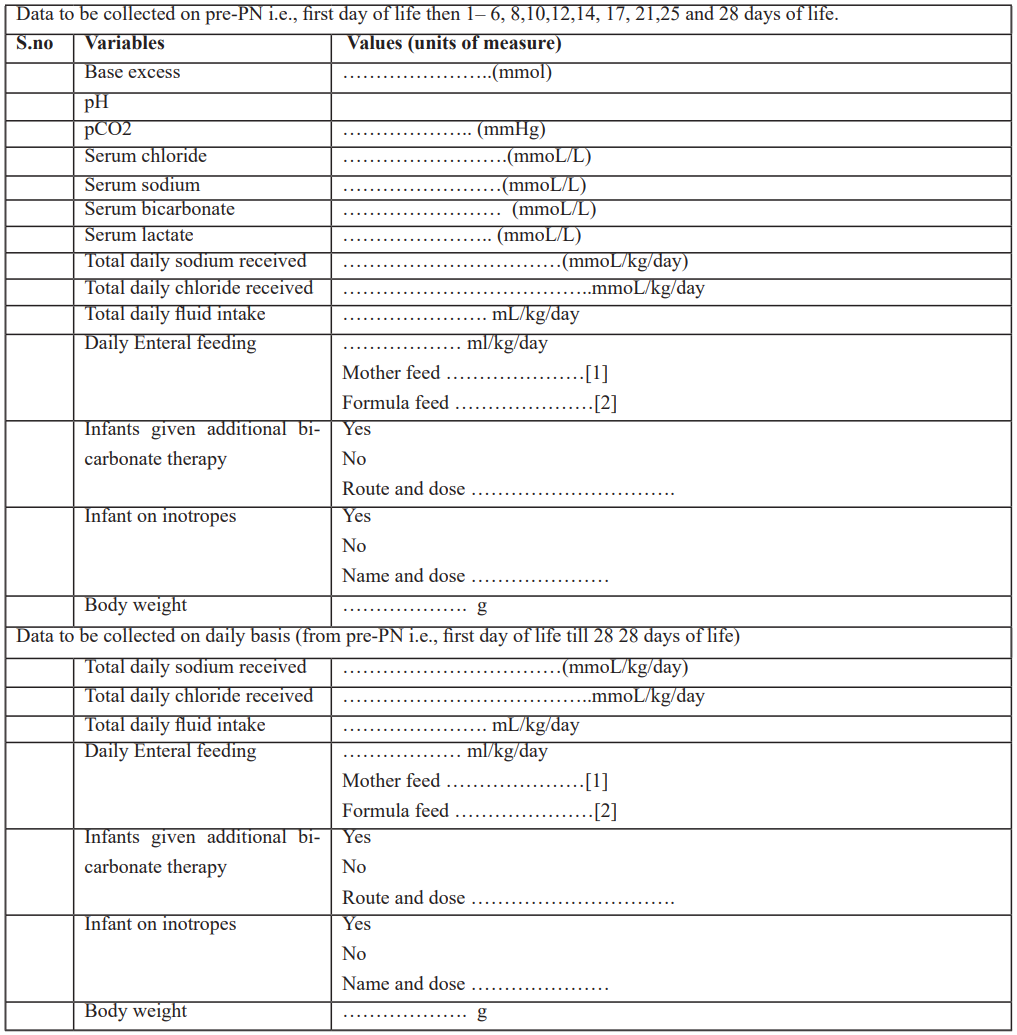
Table 3: Gantt chart presenting the study plan.
Data Analysis
Sample Size Calculations
A sample of 92 neonates per group (rounded to 100 neonates) will be required to achieve 90% power to detect a difference of 1.3 in mean pCO2 (kPa) level with an SD of 0.10 at a 5% level of significance using a two-sided t-test. The baseline pCO2 (kPa) value was found as 7.3 [23]. Using the difference of mean pCO2 (kPa), a sample size of 200 (100 neonates in each arm) is taken which is adequate for the primary outcome and secondary outcomes of interest. This will allow adjustment for all the other causes of metabolic acidosis in preterm neonates and other neonatal co-morbidities in the regression analysis.
Plan of analysis
All the statistical analysis will be performed on the statistical software STATA version 15.0. Mean ± SD will be reported for continuous variables, while n (%) will be reported for categorical variables to describe the baseline and clinical characteristics of the population. The student’s t-test or Mann-Whitney U test will be used for the comparison between both groups in all aspects. The incidence of metabolic acidosis will be compared between both groups using the Chi-square test. A p-value <0.05 will be considered significant. To compare the rate of comorbidities between the two groups Chi-square test will be performed. Logistic regression will be used to predict the risk of metabolic acidosis by using So-A doses. The Youden index will be used to obtain cut-off values for So-A doses.
Ethics:
Ethical Approval and Consent to Participate
Ethical approval from the ethical Research committee of AKU (ID#2024-6523-29448) has been granted. Filled data collection forms are the property of AKUH. In the event of any publication regarding this study, patient identity will remain confidential.
Timeline and monitoring and evaluation plan
Timeline:
12-13 months for recruitment and data collection and at least 2 months for data analysis of the study for the intervention group (So-A) and control group (So-C)
Monitoring and evaluation
Data will be gathered electronically and will be automatically uploaded to the server of AKU through the Internet. Data will be stored for an extended period and will reside in a data repository. Access to this repository will be restricted to only data management staff working on the project using AKU-LAN ID login.
Sustainability:
Once we have ascertained the effectiveness of So-A in the neonatal population and optimal dosing range to prevent/treat metabolic acidosis and associated morbidities, we will then develop the So-A guideline for neonates and would then apply for extramural grants for doing trials for the effectiveness of So-A in different age groups and do various interventions in our context.
Declarations:
Funding: Aga Khan University.
Conflicts of interest/Competing interests: The authors have no competing interests to declare that are relevant to the content of this article.
Ethics approval: The Ethical Research Committee of AKUH (ID#2024-6523-29448) has approved the study.
Consent to participate: Written informed consent obtained from parents/legal representatives before the initiation of PN.
Consent for publication: Written informed consent was obtained from parents/legal representatives for the publication of results.
Availability of data: Not applicable
Code availability: Not applicable
Acknowledgements: None
Authorship contribution statement: Adnan Mirza, Shabina Ariff, Kashif Hussain, Imran Nisar, and Sohail Salat came forth with the concept and designed the study. The literature review was carried out by Gul Ambreen, Shabina Ariff, Kashif Hussain, Imran Nisar, Sohail Salat, and Omar Irfan. Adnan Mirza, Gul Ambreen, Shabina Ariff, and Sohail Salat provided the clinical viewpoint for the article. Gul Ambreen, Kashif Hussain, Imran Nisar, and Sohail Salat, moderated and revised the manuscript. All authors approved the final version of the manuscript.
References
- Mihatsch WA, Braegger C, Bronsky J, Cai W, Campoy C, Carnielli V, et al. ESPGHAN/ESPEN/ESPR/CSPEN guidelines on pediatric parenteral nutrition. Clinical Nutrition, 2018; 37(6): 2303-2305.
- Valentine CJ, Puthoff TD. Enhancing parenteral nutrition therapy for the neonate. Nutr Clin Pract, 2007; 22: 183-193.
- Barnett HL, Vesterdal J. The physiologic and clinical significance of immaturity of kidney function in young infants. J Pediatr, 1953; 42: 98-119. |
- Heller H. The renal function of newborn infants. J Physiol, 1944; 102: 429-440.
- Ekblad H, Kero P, Takala J. Slow Sodium Acetate infusion in the correction of metabolic acidosis in premature infants. Am J Dis Child, 1985; 139: 708-710.
- Groh-Wargo S, Ciaccia A, Moore J. Neonatal metabolic acidosis: effect of chloride from normal saline flushes. JPEN J Parenter Enteral Nutr, 1988; 12: 159-161.
- Peters O, Ryan S, Matthew L, Cheng K, Lunn J. Randomised controlled trial of acetate in preterm neonates receiving parenteral nutrition. Arch Dis Child Fetal Neonatal Ed, 1997; 77: F12-15.
- Richards CE, Drayton M, Jenkins H, Peters TJ. Effect of different chloride infusion rates on plasma base excess during neonatal parenteral nutrition. Acta Paediatr, 1993; 82: 678-682.
- Kermorvant-Duchemin E, Iacobelli S, Eleni-Dit-Trolli S, Bonsante F, Kermorvant C, Sarfati G, et al. Early chloride intake does not parallel that of sodium in extremely low-birth-weight infants and may impair neonatal outcomes. J Pediatr Gastroenterol Nutr, 2012; 54: 613-619.
- Iacobelli S, Kermorvant-Duchemin E, Bonsante F, Lapillonne A, Gouyon JB. Chloride balance in preterm infants during the first week of life. Int J Pediatr, 2012; 2012: 931597.
- Jobe AH, Bancalari E. Bronchopulmonary dysplasia. Am J Respir Crit Care Med, 2001; 163: 1723-1729.
- Kenet G, Kuperman AA, Strauss T, Brenner B. Neonatal IVH--mechanisms and management. Thromb Res, 2011; 127 Suppl 3: S120-122.
- Low JA, Lindsay BG, Derrick EJ. Threshold of metabolic acidosis associated with newborn complications. Am J Obstet Gynecol, 1997; 177: 1391-1394.
- Cooke RW. Factors associated with periventricular hemorrhage in very low birth weight infants. Arch Dis Child, 1981; 56: 425-431.
- Handy JM, Soni N. Physiological effects of hyperchloraemia and acidosis. Br J Anaesth, 2008; 101: 141-150.
- McCague A, Dermendjieva M, Hutchinson R, Wong DT, Dao N. Sodium Acetate infusion in critically ill trauma patients for hyperchloremic acidosis. Scand J Trauma Resusc Emerg Med, 2011; 19: 24.
- Skutches CL, Sigler MH, Teehan BP, Cooper JH, Reichard GA. Contribution of dialysate acetate to energy metabolism: metabolic implications. Kidney Int, 1983; 23: 57-63.
- Tsai IC, Huang JW, Chu TS, Wu KD, Tsai TJ. Factors associated with metabolic acidosis in patients receiving parenteral nutrition. Nephrology (Carlton), 2007; 12: 3-7.
- Vinay P, Prud'Homme M, Vinet B, Cournoyer G, Degoulet P, Leville M, et al. Acetate metabolism and bicarbonate generation during hemodialysis: 10 years of observation. Kidney Int, 1987; 31: 1194-1204.
- Jochum F, Moltu SJ, Senterre T, Nomayo A, Goulet O, Iacobelli S, et al. ESPGHAN/ESPEN/ESPR/CSPEN guidelines on pediatric parenteral nutrition: Fluid and electrolytes. Clin Nutr, 2018; 37(6 Pt B): 2344-2353.
- Cash RA, Toha KM, Nalin DR, Huq Z, Phillips RA. Acetate in the correction of acidosis secondary to diarrhea. Lancet, 1969; 2: 302-303.
- Watten RH, Gutman RA, Fresh JW. Comparison of acetate, lactate, and bicarbonate in treating the acidosis of cholera. Lancet, 1969; 2: 512-514.
- Ali A, Ong EY, Singh BK, Cheah FC. Comparison Between Sodium Acetate and Sodium Chloride in Parenteral Nutrition for Very Preterm Infants on the Acid-Base Status and Neonatal Outcomes. Pediatric Gastroenterology, Hepatology & Nutrition, 2020; 23(4): 377.

