Antibiotic Prescription in Septic Shock
Nabih I*, Erragh A, Chaabi S, Yaseen F, Nsiri A and Alharrar R
Surgical Emergency Intensive Care unit, Ibn Rochd University Hospital Center, Faculty of Medicine and Pharmacy of Casablanca, Hassan II University, Morocco
Received Date: 22/06/2025; Published Date: 22/07/2025
*Corresponding author: Nabih Ibtissam, Surgical Emergency Intensive Care unit, Ibn Rochd University Hospital Center, Faculty of Medicine and Pharmacy of Casablanca, Hassan II University, Morocco
Abstract
Introduction: Septic shock remains a critical emergency in intensive care with a high mortality rate, particularly exacerbated in low-income countries due to logistical constraints limiting access to essential antibiotics. This study aims to evaluate the impact of initial antibiotic therapy on the mortality of patients in septic shock by analyzing the timing, duration, and appropriateness of the treatments administered.
Patients and Methods: This is a retrospective descriptive and analytical study conducted at the Ibn Rochd University Hospital in Casablanca between January 2019 and December 2023. A total of 117 patients diagnosed with septic shock according to Sepsis-3 criteria were included. Demographic, clinical, biological, and therapeutic data were collected and analyzed, particularly focusing on the relationship between the characteristics of antibiotic therapy (timing, duration, and appropriateness) and mortality.
Results: Among 1,349 admissions to the intensive care unit, 117 cases of septic shock were recorded (incidence: 8.6%). The majority of patients were male (sex ratio: 1.54), with a mean age of 58.5 years and frequent comorbidities, notably diabetes (47%) and hypertension (22%). Clinically, fever (64.1%), tachypnea (23.9%), and hemodynamic instability (28.2%) were the most common signs. Biologically, abnormalities such as anemia (84.6%), thrombocytopenia (47%), and elevated CRP (68.5%) were predominant.
Early administration of antibiotics before admission, performed in 48.7% of patients, significantly reduced mortality. However, inadequate treatment increased the risk of death. The overall mortality rate was 67.2% and was associated with delays in management. While the SOFA score strongly predicted mortality, prior antibiotic therapy did not influence its progression.
Conclusion: The study emphasizes the importance of rapid, targeted antibiotic therapy in accordance with international guidelines, especially in low-income countries where logistical barriers worsen clinical disparities.
Introduction
Septic shock remains a critical emergency in intensive care, with a mortality rate still high despite advancements in the optimization of its management [1]. This syndrome, resulting from a severe systemic infection, is often exacerbated by the occurrence of multivisceral organ failures, further compromising the prognosis of patients. Among the key components of management, rapid and effective control of the infectious source plays a determining role, in which antibiotic therapy occupies a central position.
The literature consistently emphasizes the importance of early and appropriate antibiotic administration in septic shock patients to significantly reduce mortality [2]. The timeliness and quality of this initial antibiotic therapy are crucial prognostic factors, rivaling in importance the hemodynamic and circulatory interventions [2]. However, despite these recommendations, significant disparities remain in their implementation, particularly in low- income countries, where logistical and structural constraints, such as the limited availability of certain essential antibiotics, can complicate the rapid and adequate initiation of treatment.
These logistical barriers, coupled with limited resources, exacerbate the challenges of identifying and treating infections in intensive care settings. They also contribute to the variability of clinical practices and a higher mortality rate, despite efforts to adhere to international guidelines.
In this context, the primary objective of our study was to assess antibiotic practices in terms of combinations and adherence to recommendations, taking into account the timing of initiation, duration, and therapeutic appropriateness. Secondary objectives included the analysis of predictive factors for mortality, such as the SOFA score and patient characteristics.
Patients and Methods
We conducted a retrospective descriptive and analytical study involving all patients who presented with septic shock and were admitted to the surgical emergency intensive care unit at Ibn Rochd University Hospital in Casablanca, over a four-year period from January 2019 to December 2023.
Study Population:
Inclusion Criteria: We included all patients over the age of 18 who were hospitalized for sepsis or septic shock and required antibiotic therapy. The definitions used for sepsis and septic shock were based on the third international consensus conference, "Sepsis-3."
- Sepsis was defined as a SOFA score ≥ 2 in patients with a suspected
- Septic shock was defined, in the absence of lactate measurement, as sepsis associated with the need for vasopressor drugs to maintain a mean arterial pressure (MAP) ≥ 65 mmHg.
Exclusion Criteria: Patients admitted with a SOFA score < 2 or those with organ failures unrelated to an infectious cause were excluded from the study.
Data Collection
Demographic, biological, microbiological, and therapeutic data were collected using a structured form on Microsoft Excel. Statistical analysis was performed using SPSS-22 and Microsoft Excel 2016.
Objectives and Analyses
The main objective of this study was to evaluate whether the antibiotic therapy administered followed current recommendations and to determine the impact of the quality and duration of antibiotic therapy on patient mortality. To achieve this, we conducted several analyses:
1. Timing of Antibiotic Administration and Mortality:
- The relationship between the timing of antibiotic administration and mortality was assessed using the correlation coefficient (r), which measures the strength and direction of the linear relationship between these variables.
- The closer the coefficient is to 1, the stronger the positive correlation; conversely, a coefficient close to -1 indicates a negative correlation, and a coefficient close to 0 reflects the absence of a linear relationship.
2. Inadequate Antibiotic Therapy and Mortality:
- The relevance of the antibiotic therapy was evaluated using Gyssens’ classification (Figure 1), which analyzes the quality of drug choice, dosage, and treatment duration.
- Patients were divided into two groups: survivors and deceased, with a comparison of data between the two groups using the chi-square A difference was considered significant for p ≤ 0.05.
3. SOFA Score and Mortality:
- The relationship between the SOFA score and mortality was studied using
Somers’ D test, a multivariate analysis method adapted for ordinal data.
Ethical Considerations
The collection of patient records was carried out in accordance with the principles of anonymity and confidentiality, following the applicable ethical guidelines.
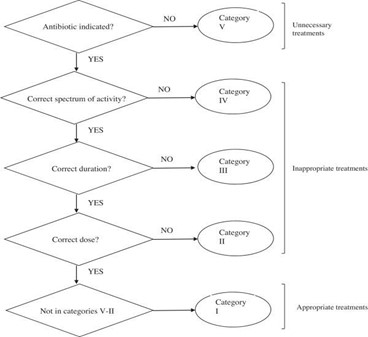
Figure 1: Antibiotic prescription classification based on Gyssens’ algorithm [3].
Results
1. General Characteristics
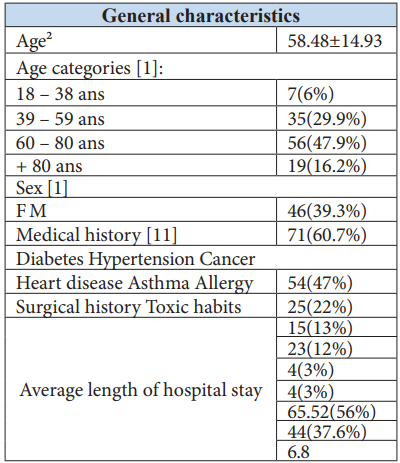
During the study period, 117 cases of septic shock were recorded out of a total of 1,349 hospitalizations, representing an incidence of 8.6%. The mean age was 58.48 years, with a standard deviation of 14.93 and a range between 16 and 90 years. The majority of patients (47.9%) were aged between 60 and 80 years. Men were more affected than women, with a sex ratio of 1.54. One or more underlying chronic diseases were identified in 79.5% of the patients, with diabetes being the most prevalent (47%), followed by hypertension (22%).
Among the patients, 56% had undergone prior surgery, and 37.6% had a history of substance use. The average length of hospitalization was 6.8 days. These data are detailed in Table 1.
Table 1: General characteristics.
2. Clinico-Biological Parameters
Clinically, the signs observed in our patients included fever in 64.1% of cases, confusion in 13.7%, tachypnea in 23.9%, and hemodynamic instability in 28.2% of cases.
Biologically, anemia was present in 84.6% of cases, and leukocytosis was observed in 59.8% of patients. Regarding platelet levels, thrombocytopenia was identified in 47% of cases, while thrombocytosis was noted in 16.2%. A low prothrombin time (PT) was found in 76.9% of cases, and an extended activated partial thromboplastin time (aPTT) was observed in 48.7%.
Electrolyte imbalances were detected in 41.9% of cases, increased creatinine levels in 58.1%, hepatic cytolysis in 33%, elevated CRP in 68.5%, and hypoalbuminemia in 58%.
The median SOFA score was 6, with an interquartile range (IQR) of 5–7.
3. Antibiotic Therapy
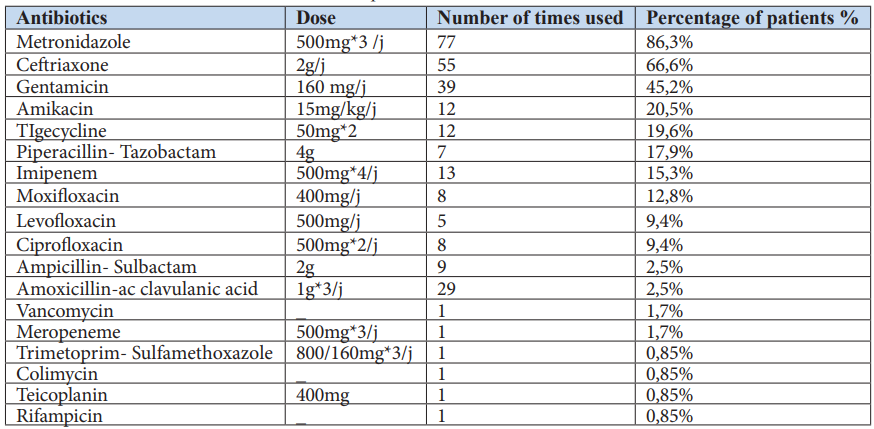
Antibiotic therapy was initiated in 48.7% of patients prior to their admission to the intensive care unit. Residual antibiotic levels were not measured in our patients.
The table below summarizes all the antibiotics prescribed to the patients in our study.
Table 2: Antibiotics prescribed in the intensive care unit.
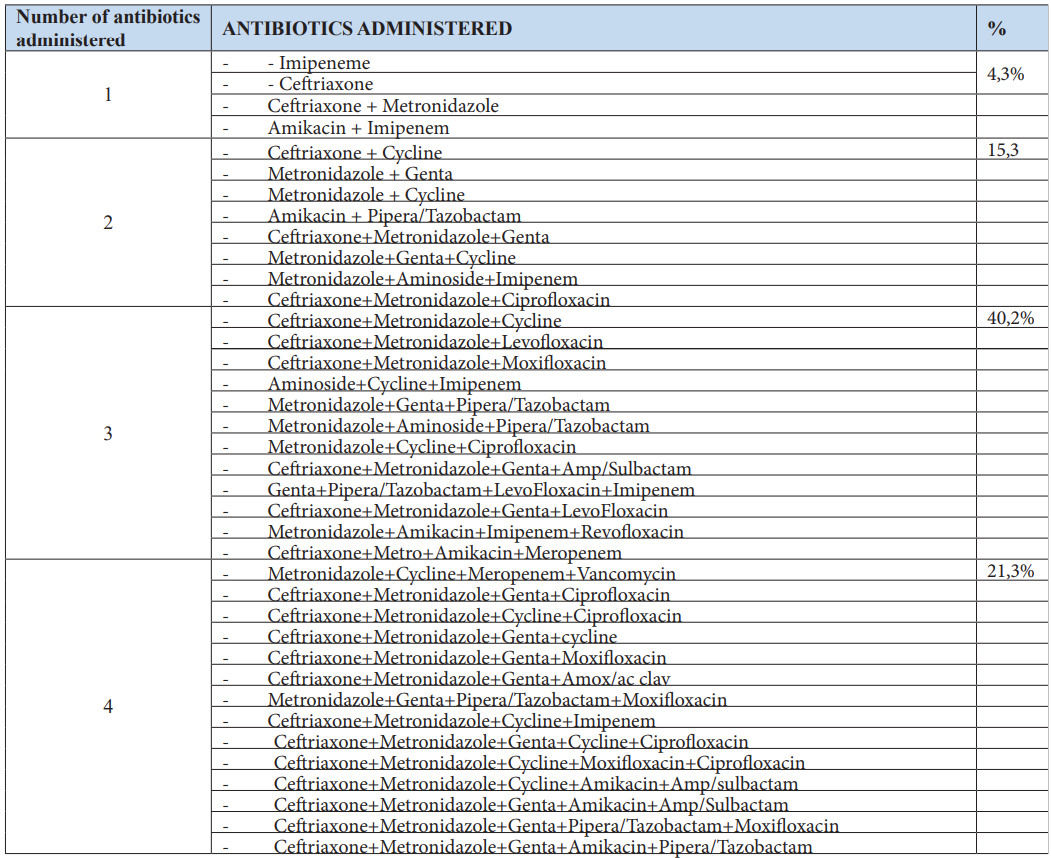
Several combinations of antibiotics were prescribed to patients throughout their stay in the intensive care unit. The table below summarizes the different combinations used.
Table 3: Most commonly prescribed antibiotic combinations for ICU patients.

The average duration of antibiotic treatments was 6.61 days, ranging from 1 to 21 days.
Mortality Analysis
The overall mortality rate was 67.2%.
1. Mortality and Timing of Antibiotic Therapy:
Table 4: Analysis of mortality in relation to time to administration.
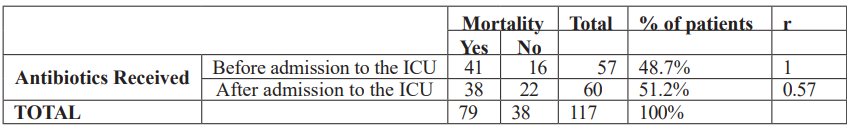
There is a strong linear correlation between mortality and the initiation of antibiotic therapy before admission to the intensive care unit in bivariate analysis. This indicates that patients who did not receive antibiotic therapy prior to ICU admission had a higher mortality rate compared to those who received it beforehand.
2. Mortality and duration of antibiotherapy :
Table 5: Analysis of mortality in relation to duration of antibiotherapy.

To determine whether there is a link between mortality and insufficient duration of antibiotic therapy, we conducted a series of statistical tests. All tests demonstrated a close relationship between the duration of antibiotic therapy and mortality.
Table 6: Correlation Tests (Duration of Antibiotic Therapy and Mortality).
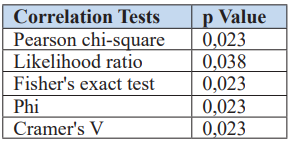
Mortality in the group with a duration of antibiotic therapy of less than 4 days was significantly higher than in the group with a duration of more than 4 days.
3. Mortality and the Number of Antibiotics Administered:
Table 7: Analysis of mortality in relation to the number of antibiotics received.
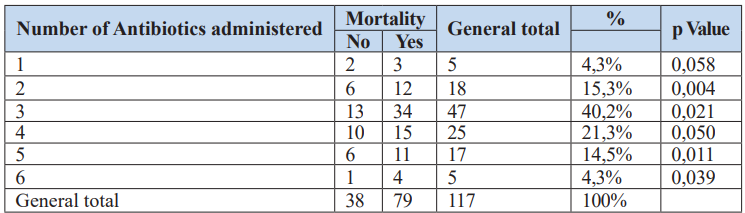
The analysis of the association between the number of antibiotics received and mortality concluded that patients who received 2, 3, 4, 5, or 6 antibiotics had a higher mortality rate compared to those who received only one antibiotic.
4. Mortality and Gyssens Classification:
Ten records, representing 8.5% of the patients, could not be assessed using the Gyssens classification due to insufficient data. Antibiotic therapy was deemed appropriate in 57.2% of cases. Errors in administration were identified in 42.6% of cases, primarily involving a non-compliant spectrum in 29.8% and non-compliant duration in 12.8% of cases. The mortality rate of patients according to the Gyssens classification is summarized in Figure 2 below.
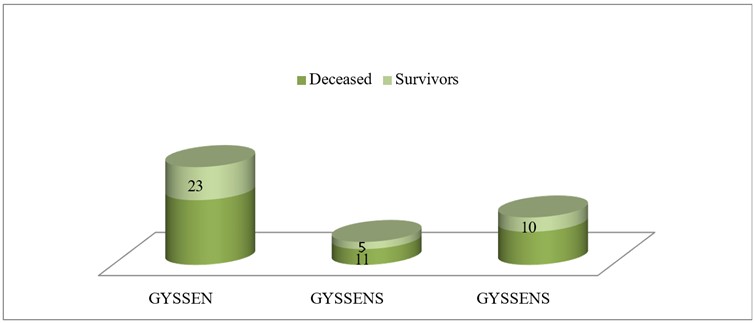
Figure 2 : Mortality according to the GYSSENS classification.
Table 8: Analysis of mortality in relation to the GYSSENS classification.
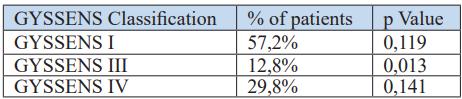
GYSSENS III (a: excessively long duration: 31.26%; p = 0.8 and b: excessively short duration: 68.75%; p= 0.023)
It was concluded that patients who received antibiotic therapy of inadequate duration had a higher mortality rate (p = 0.013) compared to those who received antibiotic therapy with an inappropriate spectrum (p = 0.14).
5. Mortality and SOFA score:
The SOFA score was calculated for 92.4% of the patients, while 7.6% of the patients could not be assessed due to insufficient data.
Table 9: Analysis of mortality in relation to the SOFA score.
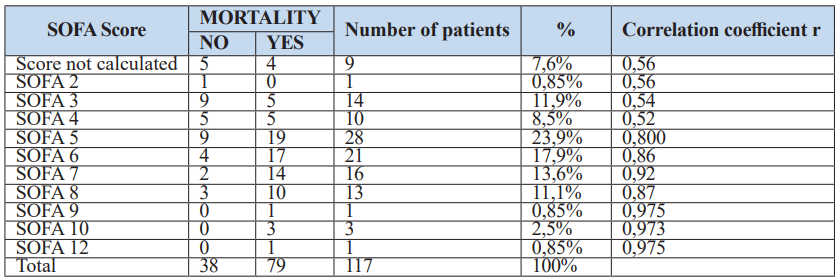
The higher the SOFA score, the greater the correlation with mortality. The value of the summing d = 0.022
6. SOFA score and prior antibiotherapy:
We compared patients who received antibiotherapy before resuscitation and those who received it only after admission to the intensive care unit on the basis of the rise in the SOFA score.
Table 10: Analysis of the relationship between changes in SOFA score and whether or not patients had received prior antibiotherapy.
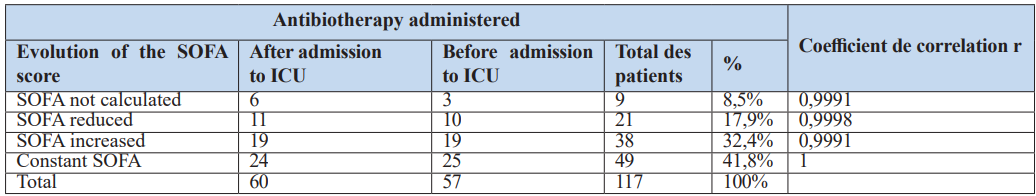
These results indicate that despite the early administration of antibiotics before ICU admission, the SOFA score does not change. This suggests that prior antibiotic therapy does not impact the evolution of the SOFA score.
6. Multivariate Analysis:
Predictive factors for mortality identified using logistic regression and confirmed by the Cox & Snell R-squared test, with a predictive accuracy of 90%, were:
- Inadequate antibiotic duration (GYSSENS III).
- SOFA score >
- Administration of 3 or fewer antibiotics, or more than 5 antibiotics.
Discussion
Our study highlighted several key aspects of the management of septic shock patients in our intensive care unit. The analysis revealed an incidence of 8.6% for septic shock, primarily affecting elderly patients with significant comorbidities, such as diabetes (47%) and hypertension (22%). These findings align with the literature, which indicates that patients with chronic comorbidities are more likely to develop septic shock due to their immunological and metabolic vulnerability [4-6].
1. Incidence of Septic Shock
The prevalence of septic shock observed in our study was 8.6%, a result consistent with international literature, where the incidence of septic shock typically ranges between 6% and 12% of ICU admissions [7]. A recent meta-analysis by Jean-Louis Vincent et al. reported incidence rates from 7% to 23% across various hospital settings [8]. Variations in reported rates may be attributed to methodological differences between studies, particularly regarding diagnostic criteria. Since the introduction of Sepsis-3 criteria, which are based on the SOFA score, estimates of sepsis and septic shock incidence have evolved, enabling more precise identification of severe cases requiring intensive care [8].
2. Demographic Characteristics and Comorbidities
The mean age of patients was 58.5 years, with a male predominance (sex ratio: 1.54). These findings align with recent studies indicating that septic shock primarily affects older individuals, with higher incidence rates among men [9,10]. This gender disparity may be related to hormonal and behavioral factors that influence immune response and exposure to risk factors [9].
The role of comorbidities, particularly diabetes (47%) and hypertension (22%), is well-documented in the literature. These conditions increase susceptibility to infections and exaggerated inflammatory responses [4,5].
3. Clinical and Biological Manifestations
The clinical data, including fever, tachypnea, and hemodynamic instability, along with biological abnormalities such as anemia, thrombocytopenia, and hypoalbuminemia, observed in our study are well-established markers of septic shock [7]. Fever reflects a systemic inflammatory response to an underlying infection, while tachypnea is often associated with metabolic acidosis or pulmonary dysfunction, indicating the severity of respiratory failure [11]). Hemodynamic instability, a key criterion for septic shock according to Sepsis-3 definitions, manifests as hypotension requiring vasopressors to maintain adequate perfusion [7].
Biologically, anemia may result from blood loss or marrow suppression due to inflammation, while thrombocytopenia is often linked to excessive platelet consumption, particularly in disseminated intravascular coagulation (DIC) [11]. Hypoalbuminemia reflects capillary leakage caused by endothelial dysfunction and is an indicator of severity [12,13]. Collectively, these parameters underscore the complex pathophysiology of septic shock, involving excessive inflammation, vascular dysregulation, and multiorgan failure, which are essential for diagnosis and management.
4. Timing of Antibiotic Administration
Only 48.7% of patients in our study received antibiotics before ICU admission. This delay can be attributed to several context-specific factors, including limited immediate availability of certain antibiotics, prolonged diagnostic delays due to restricted access to microbiological tools, and late recognition of clinical symptoms in patients often admitted in critical condition.
Current guidelines, such as the 2021 Surviving Sepsis Campaign, emphasize the importance of administering broad-spectrum antibiotics within one hour of diagnosing septic shock to limit infection progression and reduce the risk of organ failure [14]. Studies have shown that delayed initiation of antibiotic therapy is closely associated with a significant increase in mortality. For instance, each hour of delay correlates with a 7.6% increase in mortality, highlighting the critical importance of prompt management [15,16]. These results underscore the need to strengthen diagnostic and therapeutic protocols to ensure timely initiation of antibiotic treatment in line with best clinical practices.
5. Choice of Antibiotics and Combinations
Metronidazole (86.3%) and third-generation cephalosporins (66.6%) were the most frequently used antibiotics, reflecting a strategy aimed at covering a wide range of pathogens, including anaerobes and Gram-negative bacilli, commonly involved in abdominal and polymicrobial infections associated with septic shock [17]).
The administration of multiple antibiotics simultaneously—sometimes up to six agents—while potentially justified for complex or resistant infections, raises significant concerns. These combinations increase the risks of toxicity, opportunistic infections like Clostridioides difficile, and bacterial resistance, a particularly critical issue in intensive care [18]. International guidelines, such as the 2016 Surviving Sepsis Campaign, recommend systematic reevaluation of antibiotic therapy after 48–72 hours based on microbiological results to reduce overuse and optimize targeted treatment
(16). These findings highlight the importance of implementing antimicrobial stewardship programs in ICUs to streamline antibiotic use, minimize bacterial resistance, reduce adverse effects, and optimize associated costs [19].
6. Duration of Antibiotic Therapy
The average duration of antibiotic treatment in our study was 6.61 days, slightly below current recommendations, which suggest a duration of 7 to 10 days for most severe infections, depending on clinical response and microbiological data [16].
An inappropriate treatment duration, particularly if too short, was clearly associated with increased mortality in our study, a finding corroborated by the literature. Hellyer et al. demonstrated that short durations increase the risk of relapse, persistent infection, or resistance emergence, while excessive prolongation may lead to side effects and microbiome imbalances [20]. The use of biomarkers such as procalcitonin can help individualize and optimize treatment duration, reducing unnecessary exposure while maintaining equivalent clinical efficacy [21]. Rigorous clinical assessment is essential to adjust antibiotic durations and improve outcomes in sepsis or septic shock patients.
7. Impact of Antibiotic Therapy on Mortality
Our study revealed an overall mortality rate of 67.2%, with higher mortality among patients who did not receive antibiotics before ICU admission. Recent guidelines, including the 2021 Surviving Sepsis Campaign, emphasize the administration of broad-spectrum antibiotics within one hour of diagnosing septic shock, significantly reducing mortality [14]. Inappropriate antibiotic therapy, as identified through the Gyssens classification, was associated with increased mortality, corroborating findings by Weinberger et al., who demonstrated that inadequate treatment doubles the risk of death [22]. Combining prompt initiation with adjustments based on microbiological results is crucial to improving outcomes.
8. Impact of SOFA Score on Mortality and the Role of Prior Antibiotic Therapy
The SOFA score is a key tool for assessing organ failure and predicting mortality in sepsis patients. Our study shows a direct correlation between higher SOFA scores and mortality, with scores above 5 significantly associated with increased risk. These findings align with recent literature showing that each one-point increase in the SOFA score is linked to a significant rise in mortality.
Huang et al. developed a model combining the SOFA score with comorbidity indices, demonstrating superior performance in predicting hospital mortality in sepsis [23]. Furthermore, dynamic trajectories of the SOFA score within the first 72 hours, as analyzed by Palmowski et al., were robust predictors of 30-day mortality [24].
Regarding antibiotic therapy, our study reveals that its administration before ICU admission does not directly impact SOFA score trajectories, although rapid initiation is associated with an overall reduction in mortality. Karakike et al. showed that early SOFA score changes, particularly within the first seven days, are key indicators of treatment response and survival in septic shock [25]. Li et al. highlighted the importance of integrating the SOFA score with other parameters, such as procalcitonin, to optimize survival predictions and guide therapeutic decisions [26]. These results confirm that while the SOFA score is a reliable predictor, its utility is enhanced when integrated into a multimodal approach, including the timeliness and optimization of antibiotic therapy.
9. Study Limitations
Our study has certain limitations inherent to its retrospective nature, such as reliance on available medical records and the absence of specific microbiological data. However, as a single-center study, it provides a detailed and contextualized analysis of local practices, although validation in larger populations may be needed to confirm the generalizability of the results.
Conclusion
Our study highlights the crucial importance of early and appropriate antibiotic administration in patients with septic shock. Delays or inadequacies in initial treatment are closely associated with increased mortality. However, in the context of low-income countries, these challenges are exacerbated by significant logistical barriers, including limited availability of essential antibiotics, budgetary constraints, and insufficient infrastructure for optimal care. These realities complicate the implementation of international recommendations and amplify disparities in clinical outcomes.
Timely intervention, combined with the careful selection of antimicrobial agents, remains a cornerstone of management. Our findings underscore the necessity of adapting clinical protocols to local constraints and implementing antimicrobial stewardship programs tailored to these settings. These programs should include better resource allocation, continuous medical training, and improvements in supply chains to ensure adequate availability of treatments.
By strengthening these strategies, it would be possible to significantly improve patient outcomes and reduce inequalities in access to quality care. These observations also call for future research to evaluate the impact of these interventions in resource-limited environments, taking into account local socio-economic and epidemiological factors. Such efforts will not only align clinical practices with the latest standards but also better address the specific needs of patients in resource- constrained settings.
References
- Mahapatra S, Heffner Septic Shock. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2024.
- Shrestha GS, Kwizera A, Lundeg G, Baelani JI, Azevedo LCP, Pattnaik R, et International Surviving Sepsis Campaign guidelines 2016: the perspective from low-income and middle-income countries. Lancet Infect Dis, 2017; 17(9): 893‑895. doi: 10.1016/S1473-3099(17)30436-3.
- Gyssens IC, van den Broek PJ, Kullberg BJ, Hekster YA, van der Meer JW. Optimizing antimicrobial A method for antimicrobial drug use evaluation. J Antimicrob Chemother, 1992; 30(5): 724‑747. doi: 10.1093/jac/30.5.724.
- Fleischmann C, Scherag A, Adhikari NKJ, Hartog CS, Tsaganos T, Schlattmann P, et al. Assessment of Global Incidence and Mortality of Hospital-treated Current Estimates and Limitations. Am J Respir Crit Care Med, 2016; 193(3): 259‑272. doi: 10.1164/rccm.201504- 0781OC.
- Costantini E, Carlin M, Porta M, Brizzi MF. Type 2 diabetes mellitus and sepsis: state of the art, certainties and missing Acta Diabetol, 2021; 58(9): 1139‑1151. doi: 10.1007/s00592- 021-01728-4.
- Rudd KE, Johnson SC, Agesa KM, Shackelford KA, Tsoi D, Kievlan DR, et Global, regional, and national sepsis incidence and mortality, 1990-2017: analysis for the Global Burden of Disease Study. Lancet, 2020; 395(10219): 200‑211. doi: 10.1016/S0140-6736(19)32989-7.
- Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, et The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA, 2016; 315(8): 801‑810. doi: 10.1001/jama.2016.0287.
- Vincent JL, Jones G, David S, Olariu E, Cadwell KK. Frequency and mortality of septic shock in Europe and North America: a systematic review and meta-analysis. Crit Care, 2019; 23(1): 196. doi: 10.1186/s13054-019-2478-6.
- Li Y, Li H, Zhang D, Zhang Y, Li J, Li Y, et al. Sex differences in the SOFA score of ICU patients with sepsis or septic shock: a nationwide Crit Care, 2024; 28(1): 49. doi: 10.1186/s13054-024- 04996-y.
- Li Q, Chai W, Wang X, Cheng L, Cai X, Fu J, et Epidemiological analysis of septic shock in the plateau region of China. Front Med, 2022; 9: 968133. doi: 10.3389/fmed.2022.968133.
- Kumar Sepsis roadmap: What we know, what we learned, and where we are going. Clin Immunol, 2020; 210: 108264. doi: 10.1016/j.clim.2019.108264.
- Raia L, Zafrani Endothelial Activation and Microcirculatory Disorders in Sepsis. Front Med, 2022; 9: 907992. doi: 10.3389/fmed.2022.907992.
- Fernández-Sarmiento J, Schlapbach LJ, Acevedo L, Santana CR, Acosta Y, Diana A, et Endothelial Damage in Sepsis: The Importance of Systems Biology. Front Pediatr, 2022; 10: 828968. doi: 10.3389/fped.2022.828968.
- Evans L, Rhodes A, Alhazzani W, Antonelli M, Coopersmith CM, French C, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock Intensive Care Med, 2021; 47(11): 1181‑247. doi: 10.1007/s00134-021-06506-y.
- Kumar A, Safdar N, Kethireddy S, Chateau D. A survival benefit of combination antibiotic therapy for serious infections associated with sepsis and septic shock is contingent only on the risk of death: a meta-analytic/meta-regression study. Crit Care Med, 2010; 38(8): 1651‑1664. doi: 1097/CCM.0b013e3181e96b91.
- Rhodes A, Evans LE, Alhazzani W, Levy MM, Antonelli M, Ferrer R, et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: Intensive Care Med, 2017; 43(3): 304‑377. doi: 10.1007/s00134-017-4683-6.
- Kalil AC, Metersky ML, Klompas M, Muscedere J, Sweeney DA, Palmer LB, et Management of Adults with Hospital-acquired and Ventilator-associated Pneumonia: 2016 Clinical Practice Guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis, 2016.
- Tamma PD, Aitken SL, Bonomo RA, Mathers AJ, van Duin D, Clancy Infectious Diseases Society of America Guidance on the Treatment of Extended-Spectrum β-lactamase Producing Enterobacterales (ESBL-E), Carbapenem-Resistant Enterobacterales (CRE), and Pseudomonas aeruginosa with Difficult-to-Treat Resistance (DTR-P. aeruginosa). Clin Infect Dis, 2021; 72(7): 1109‑1116. doi: 10.1093/cid/ciaa1478.
- Bassetti M, Rello J, Blasi F, Goossens H, Sotgiu G, Tavoschi L, et Systematic review of the impact of appropriate versus inappropriate initial antibiotic therapy on outcomes of patients with severe bacterial infections. Int J Antimicrob Agents, 2020; 56(6): 106184. doi: 10.1016/j.ijantimicag.2020.106184.
- Hellyer T, Mantle T, McMullan R, Dark How to optimise duration of antibiotic treatment in patients with sepsis? BMJ, 2020; 371: m4357. doi: 10.1136/bmj.m4357.
- Gutiérrez-Pizarraya A, León-García MDC, De Juan-Idígoras R, Garnacho-Montero J. Clinical impact of procalcitonin-based algorithms for duration of antibiotic treatment in critically ill adult patients with sepsis: a meta-analysis of randomized clinical Expert Rev Anti Infect Ther, 2022; 20(1): 103‑112. doi: 10.1080/14787210.2021.2013252.
- Weinberger J, Rhee C, Klompas A Critical Analysis of the Literature on Time-to-Antibiotics in Suspected Sepsis. J Infect Dis, 2020; 222(Suppl 2): S110‑118. doi: 10.1093/infdis/jiaa246.
- Huang Y, Gao Y, Quan S, Pan H, Wang Y, Dong Y, et al. Development and internal-external validation of the ACCI-SOFA model for predicting in-hospital mortality of patients with Sepsis-3 in the ICU: a multicenter retrospective cohort study. Shock, 2024; 61(3): 367‑374. doi: 1097/SHK.0000000000002043.
- Palmowski L, Nowak H, Witowski A, Koos B, Wolf A, Weber M, et Assessing SOFA score trajectories in sepsis using machine learning: A pragmatic approach to improve the accuracy of mortality prediction. Lazzeri C, éditeur. PLOS ONE, 2024; 19(3): e0300739. doi: 10.1371/journal.pone.0300739.
- Karakike E, Kyriazopoulou E, Tsangaris I, Routsi C, Vincent JL, Giamarellos-Bourboulis The early change of SOFA score as a prognostic marker of 28-day sepsis mortality: analysis through a derivation and a validation cohort. Crit Care, 2019; 23(1): 387. doi: 10.1186/s13054-019-2652-x.
- Li W, Wang M, Zhu B, Zhu Y, Xi X. Prediction of median survival time in sepsis patients by the SOFA score combined with different predictors. Burns Trauma, 2020; 8: tkz006. doi: 10.1093/burnst/tkz006.

