Calcitriol Revises Aromatase Gene Expression in Ehrlich Solid Tumor Bearing Mice Exposed to Low Dose Gamma Radiation
ZNermeen M El Bakary*, Khaled Sh Azab, Shaden M Hanafy, Ghada M Abdel Aziz and Aysam M Fayed
Radiation Biology Department, Radiation Research Division, National centre for Radiation Research and Technology, Atomic Energy Authority, Egypt
Molecular biology Department, Institute of Genetic Engineering and Biotechnology, University of Sadat City, Egypt
Received Date: 02/02/2021; Published Date: 15/03/2021
*Corresponding author: Nermeen M El Bakary, Radiation Biology Department, Radiation Research Division, National center for Radiation Research and Technology, Atomic Energy Authority, Egypt. Email: nermeen_ahmed11@yahoo.com
Abstract
Background: Dysregulation of aromatase expression had been monitored in many types of cancer. Our study aimed to evaluate the possible role of calcitriol (Cal; Vit D3-OH) or/and low dose of gamma radiation in regulation of aromatase gene expression and the regression of tumor proliferation in murine model (EST; Ehrlich solid tumor bearing mice).
Methods: Mice with ≈1 cm3 EST were received (i.p. injection) day after day repeated doses of Calcitriol (Cal) (0.05µg/mouse) for 14day or/and exposed to 0.5 Gy gamma radiation (low dose) delivered as one shot at dose rate 0.48 Gy/min.
Results: Our results demonstrated that, mRNA expression of aromatase, levels of cyclooxygenase (COX2) and prostaglandin (PGE2) in addition to volume of the tumor are significantly decreased while caspase 3 level is significantly increased in EST mice treated with Cal or/and exposed to 0.5 Gy gamma ray compared to untreated EST bearing mice. However, the most pronounced improvements in all of the measured parameters were obviously indicated in EST mice group treated with Cal and exposed to gamma radiation. This was accomplished by suppression of inflammatory markers which cause down regulation in aromatase mRNA expression as well as augmenting apoptosis by inducing Caspase3 concentration.
Conclusion: It could be concluded that the exposure to low dose gamma radiation potentiate the action of Calcitriol against tumor growth in the subjected murine model which represent a prospective policy for the management of solid tumor and decreasing the possibilities of tumor drug resistance.
Keywords: Calcitriol; Gamma radiation; Aromatase; COX2; PGE2; Caspase3; Tumor Volume
Introduction
The medical history of cancer began millennia ago. Historical findings of patients with cancer date back to ancient Egyptian and Greek civilizations, where this disease was predominantly treated with radical surgery and cautery that were often ineffective, leading to the death of patients [1]. Searching for novel approaches to control malignancies is the noble target of scientists working in the field of cancer research and many other related fields. Currently, several researchers are focused on the development of cell therapies, anti-tumor vaccines, and new biotechnological drugs that have already shown promising results in preclinical studies, therefore, in the near future, we will certainly assist to a new revolution in the field of medical oncology [2,3,4].
For many years, breast cancer has had the highest incidence of all cancers in women worldwide. There are significant data implicating estrogen and its metabolites in the development of breast cancer. Estrogen binding to its receptor results in proliferation of estrogen-sensitive epithelial tissues. With rapid proliferation, there is the potential for genetic errors and consequent predisposition to malignant cellular transformation [5]. Alternatively, estrogen metabolites may have intrinsic genotoxic effects and cause DNA damage and cellular transformation [6]. Postmenopausal combined hormone therapy increases the risk of breast cancer, the chances of dying from breast cancer, and the likelihood that the cancer may be found only at a more advanced stage. There are many evidences linking between estrogens, largely contributing to breast cancer development and other types of cancer [7,8,9]. Estrogen activates tumor growth and inhibits apoptosis through mechanisms mediated by its interaction with estrogen receptors [10].
Aromatase (CYP19), a cytochrome P450 enzyme, responsible for estrogens biosynthesis is important in the growth of breast cancers in both pre- and postmenopausal women. It is highly expressed in human breast cancer tissue compared to normal breast tissue. Further, in situ estrogen production is more effective than circulating estrogen in the promotion of breast cancer. The tumor aromatase has been shown to stimulate breast cancer growth via an autocrine and a paracrine manner [11]. Estrogen synthesized locally becomes the major source of the hormone in the breast after menopause when circulating estrogen levels from the ovaries dramatically decline [12].
As aromatase catalyzes the final and rate-limiting step in the biosynthesis of estrogen, inhibitors of this enzyme are effective targeted therapy. The Aromatase Inhibitors (AIs), a standard treatment for postmenopausal patients is effective in adjuvant and first-line metastatic setting. Aromatase inhibitors lower estrogen levels by stopping aromatase from changing other hormones into estrogen (Estrogen can fuel the growth of breast cancer cells). Preclinical data in resistance models demonstrated that the crosstalk between estrogen and other signaling pathways is an important resistant mechanism. Blockade of these signaling pathways is an attractive strategy to overcome the resistance to AI. There are several clinical trials to evaluate novel targeted therapies to reverse resistance to Ais [13,14,15].
We are interested in Vit D3 (Cholecalciferol) which is derived from food and is essential precursor to the potent steroid hormone, 25- dihydroxy vitamin D3, 1α,25(OH)2D3, or calcitriol. Hormonally active Vit D (calcitriol) has been claimed for its critical importance in the case of health or disease. The decrease in vitamin D content leads to group of abnormalities advanced to serious diseases whereas, the over consumption of Vit D leads to several undesired symptoms [16]. The inhibitory action of calcitriol on aromatase expression has been subjected to investigation in our present study.
Evidences have been accumulated to indicate that calcitriol modulates calcium and bone metabolism. These include anti-proliferation, anti-angiogenesis, pro-apoptosis, pro differentiation, and immune regulation in a cell and tissue [17]. Also, the calcitriol signaling plays an important role in the development of normal mammary gland [18].
Studies on vitamin D receptor (VDR) knockout mice provide evidence that calcitriol signaling through the VDR opposes estrogen driven proliferation of mammary epithelial cells and maintains normal differentiation [19].
Low dose gamma irradiation has been authenticated as a promising auxiliary strategy to improve the immune adaptive response and its implication during the strategy of cancer treatment indicates many intriguing results [3,4,20].
Feinendegen reported that radiation hormesis is the hypothesis that low doses of ionizing radiation (A low dose of IR is a dose that creates a burst of hits and ROS that is adequate to stimulate the protective systems and produce observable health benefits, within the region of and just above natural background levels in the range 0.01-0.50 Gy ) [21] are beneficial, stimulating the activation of repair mechanisms that protect against disease, that are not activated in absence of ionizing radiation [22]. Hence, the radiation adaptive response direct DNA repair and gives the impression to be a central origin of the prospective hormetic effect [23].
The pro inflammatory molecules; prostaglandins (PGs) and its synthesis rate limiting enzyme, cyclooxygenase-2 (COX2) play an important role in the development and progression of cancer [24]. Many acknowledged evidences addressed the correlation between over expression of Aromatase and expression of inflammatory molecules (PGS and COX2) [25,26]
The irradiation of EST bearing mice at low dose was taken as argue of immune system [27,28]. this work explored the role of vitamin D3 analogue; Calcitriol as an Aromatase inhibitor in regulating the cancer proliferation in mice treated with low dose and to what extent it can reduce the possibility of cancer treatment failure in Ehrlich Solid Tumor (EST) bearing mice exposed to low radiation dose.
Material and Methods
Animals
Female swiss albino mice (1 years old) weighing (30-35g) were obtained from the National Cancer Institute (Cairo University, Giza, Egypt). Animals were housed in plastic cages and maintained under standard conditions of temperature, humidity and 12 h light/dark cycles along the experimental period. They were provided with a pellet diet containing all the necessary nutritive elements. Food and water were available throughout the experimental periods ad libitum. Mice were left for acclimatization period of 1 week before the start of the experiment.
Animal experimentation were consistent with the guidelines of Ethics by the Guide for the Care and Use of Laboratory Animals (National Research Council, 1996) in accordance with the recommendations for the proper care and use of laboratory animals approved by Animal care committee of the National Center for Radiation Research and Technology (NCRRT), Cairo, Egypt.
Calcitriol (Cal)
Calcitriol (Vitamin D3 capsule; 1000 IU) was obtained from Puritan's Pride Company (Cairo, Egypt). It was dissolved and diluted (1:10 v/v) in saline. Each mouse received intraperitoneal (i.p) injection of 0.05µg/mouse once every 48 h for 14 days [29].
Tumor Inoculation:
The initial inoculums of Ehrlich Ascites Carcinoma (EAC) cells, kindly provided by the National Cancer Institute, Cairo University, and Giza, Egypt) were propagated in our laboratory. This was performed by weekly i. p. injection of freshly ascites fluid withdrawn from a donor mouse (bearing 6-8 days old ascites tumor) into 4 female mice each weighing 35g approximately using sterile disposable syringes under aseptic conditions. Whereas, the cell line of EAC was preserved by ip injection of 2.5 million cells per animal. To count the EAC before ip, the bright line hemocytometer was used. Physiological sterile saline solution was used for the injection and dilution. Solid tumor of Ehrlich carcinoma was obtained by the intramuscular transplantation of 2.5 x106 viable Ehrlich cells into the right thigh muscle of the lower limb of healthy mice, and obtained after two weeks of inoculation (zero time of the experiment) [3].
Tumor Volume Monitoring
Tumor volume was measured at different time intervals during the experimental period using a Vernier caliper on the 15th, 21th, and 28st days from the time the tumor reached 1 cm3 during the experimental period. The volume of solid tumor was calculated using the following formula according to Papadopoulos et al [30]. [A × B2× 0:52], where A and B are the longest and the shortest diameter of tumor, respectively
Low Dose Gamma Irradiation:
Whole body gamma irradiation to mice was performed using a Gamma cell 40 Cesium Irradiator (Atomic Energy of Canada Ltd., Ontario, Canada) located in the National Center for Radiation Research and Technology (NCRRT) of the Atomic Energy Authority (EAEA, Cairo, Egypt) which ensured a homogenous dose distribution all over the irradiation tray. Mice were placed in a specially designed well-ventilated acrylic container and each mouse was whole body irradiated at 0.5Gy [31], at a dose rate of 0.48Gy/min calculated by the authorization of the Dosimeter Department in the NCRRT.
Animal grouping
A total number of 90 female Swiss albino mice were randomly allocated into 6 equally- sized groups as follows:
Group 1 (Positive control, E): Mice bearing solid Ehrlich tumor
Group 2 (Irradiated, R): Mice of this group were whole body exposed to 0.5 Gy of γ-radiation at the day 15 of the experiment.
Group3 (Calcitriol, Cal): Mice of this group were intraperitoneally injected with Cal (0.05 µg Cal /mouse) every other day for 14 day starting at 14 days from initiation of the experiment (from zero time).
Group 4 (E+ R): Mice of this group are bearing solid tumor and then exposed to 0.5 Gy of whole body γ-radiation.
Group 5 (E+ Cal): Mice of this group are intraperitoneally injected with 0.05 µg Cal/mouse once every other day for 14 days starting on the 15th day from experiment.
Group 6 (E+ R+ Cal): Mice of this group are exposed to 0.5 Gy whole body γ-irradiation and intraperitoneally injected with 0.05 µg Cal/mouse once every other day for 14 days starting on the 15thday from the day of Ehrlich cells inoculation.
Samples preparation
At the day after complete all treatments according to the experiment suggested regimen (15th days from the experiment zero time); mice were anesthetized using diethyl ether. The blood was collected on heparinized tubes by using syringe for heart puncture and the collected blood were preserved at -80 ºC for the molecular analysis (RT-PCR). Animals were sacrificed and undergo necropsy. Portions of solid tumor were dissected, rinsed in ice cold isotonic saline, blotted dry with a filter paper and preserved at -20 ºC for ongoing biochemical analysis.
RT-qPCR assessment of Aromatase gene expression
Entire cellular RNA was extracted from the thigh muscles by use of RNeasy® Mini Kit (Qiagen, Hilden, Germany). The procedures were done according to the manufacturer’s instructions. The total RNA concentration and also the purity of RNA samples were determined by using the Nano Drop spectrophotometer (Thermo Fisher Scientific, USA). Purity of samples was determined by measuring absorbance at 260 and 280 nm, respectively.
First-strand complementary DNA (cDNA) was synthesized using Thermo Scientific™ Revert Aid™ First-Strand cDNA Synthesis Kit (Fermantas, Thermo Fisher Scientific Inc, Runcorn, UK).
Real-time polymerase chain reaction (PCR) amplification and analysis were performed in an optical 96-well plate in ABI PRISM 7500 Fast Sequence Detection System Thermal Cycler (Applied Biosystems, Foster City, CA, USA) using Power SYBRR Green PCR Master Mix (Applied Biosystems).
The relative expression of the real-time reverse transcriptase PCR products was determined by the ΔΔCt method. This method calculates a relative expression to housekeeping gene using the equation: fold induction = 2^ - (ΔΔCt). Where ΔΔ Ct=Ct [gene of interest (unknown sample)-Ct housekeeping gene (unknown sample)] - [Ct gene of interest (calibrator sample) - Ct housekeeping gene (calibrator sample)] [32].
Primers used for aromatase RNA gene expression and glyceraldehyde -3- phosphate dehydrogenase (GAPDH) as a housekeeping gene are as follow
Table 1: Sequences of primers used to accomplish RT-PCR amplification.

Biochemical Analysis
Caspase 3 was assayed in solid EC tumor by a sandwich enzyme-linked immunosorbent assay (ELISA) mouse kit provided by R&D Systems, Inc. (Minneapolis, USA according to the kits manufacture procedures. Whereas, Prostaglandin level in solid EC tumor was determined by a sandwich enzyme-linked immunosorbent assay (ELISA) mouse kit provided by Enzo life science (Postfach, Switzerland). The COX-2 enzyme activities were detected spectrophotometrically in tissue homogenate. Cyclooxygenases catalyze the synthesis of prostaglandins from arachidonic acid. The COX component converts arachidonic acid to Prostaglandin G2 (PGG2) whereas the peroxidase component reduces the PGG2 to the corresponding alcohol, Prostaglandin H2 (PGH2). In this kit the TMPD serves as a reducing agent, TMPD gets oxidized, so electrons flow from the TMPD to PGG2 and the appearance of the oxidized TMPD is monitored at 590 nm. The rate of increase in absorbance was taken for calculating the activity of the enzyme [33].
Statistical analysis
All data were subjected to the Shapiro–Wilk test for normality (p >0.05) to ensure the normal distribution. After confirmation of data normal distribution statistical analysis of the difference between means was performed using one-way analysis of variance (ANOVA). When significant F-ratio was present, the post hoc Duncan’s test for multiple comparisons was used to evaluate the statistical significance difference between various groups (p < 0.05). Statistical Package for Social Science (SPSS) version 20.0 (SPSS Inc., Chicago, IL, USA) were used to run all of the statistical analyses included in the present study.
Results
mRNA expression of Aromatase
The present study comprises the assessment of Aromatase gene expression using RT-PCR technique. Table (2) Shows that the highest mRNA expression of Aromatase was found in the Ehrlich solid tumor of E mice group which considered as +ve control group whereas, the lowest level was recorded in group 3 (Cal) mice. The results demonstrated the most expression of Aromatase mRNA in untreated solid tumor considered as 100% expression and the administration of several equal doses of Cal reduced the expression to 78.3%. Whereas, the exposure of Ehrlich bearing mice to single dose of 0.5Gy reduced to 76% compared to expression of that in the untreated Ehrlich bearing mice. The most pronounced reduction amongst groups was observed in E+ R+ Cal.
Table 2: Comparative mRNA expression (arbitrary units) of Aromatase in the right thigh muscle or solid tumor of different animal groups.
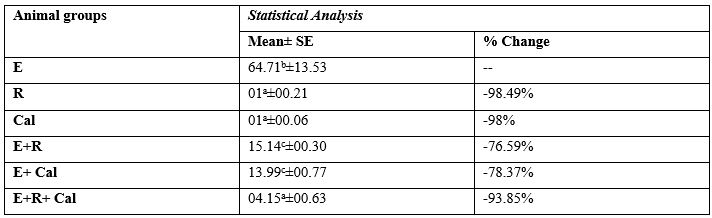
R: Radiation E: Ehrlich Cal: calcitriol
Each value represents Mean± SE (n=15)
Values with dissimilar superscript letters are considered significantly different (p <0.05).
%: Percentage of change from the values of mRNA in mice bearing Ehrlich solid tumor (E).
Percent change = T-C/C* 100
Inflammatory markers
COX2
The protein concentration of COX2 in different animal groups showed a great variation as shown in Table (3).
The result illustrated a marked down regulation of COX- 2 protein in (E+R+Cal) gp of mice in comparison to (E+R) gp or (E+Cal) gp.
Table (3): Cyclooxygenase-2 (COX-2) concentration in different animal groups
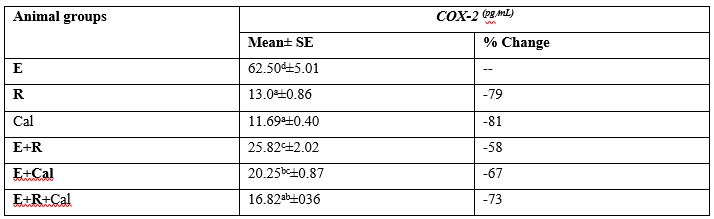
R: Radiation E: Ehrlich Cal: calcitriol
Each value represents Mean± SE (n=15)
Values with dissimilar superscript letters are considered significantly different (p <0.05).
%: Percentage of change from the values in mice bearing Ehrlich solid tumor (E).
Percent change = T-C/C* 100
PGE2
A significant increase of tumor PGE2 concentration in (E) gp of mice and (E+R) gp of mice compared to the other gps as shown in (Table 4)
In mice bearing solid tumor and pretreated with Cal a remarkable (p<0.05) drop in tumor PGE2 level compared to its equivalent value in E mice was recorded. Whole body exposure of E mice to 0.5 Gy of gamma radiation either alone or along with Cal injection produced a pronounced decline in tumor PGE2 levels when matched up to E mice. The result obtained revealed that tumor of E+ R+ Cal mice group demonstrated the sharpest decline in tumor PGE2 level compared with its equivalent value in the tumor of E+ Cal and E+ R mice groups.
Table 4: Prostaglandin E2 (PGE2) level in different animal groups.
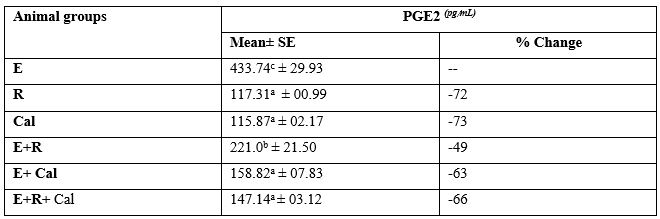
R: Radiation E: Ehrlich Cal: calcitriol
Each value represents Mean± SE (n=15)
Values with dissimilar superscript letters are considered significantly different (p <0.05).
%: Percentage of change from the values in mice bearing Ehrlich solid tumor (E).
Percent change = T-C/C* 100
Apoptosis:
The experimental data relating to the activities of caspase3 in different animals’ groups is presented in Table (5).
Table shows that E mice manifested a significant (p<0.05) decline in cleaved caspase-3 protein compared to its equivalent value in other animal groups.
Treatment of E mice with Cal resulted in a significant elevation in caspase-3 concentration, compared to E mice. Further, exposure of E mice to γ-irradiation either alone or in combination with Cal treatment induces significant (p<0.05) increase in cleaved caspase-3 protein compared to its equivalent value in E mice. However, the combination of the two treatment modalities (E+ R+ Cal) shows the most pronounced increases of caspase-3 on account of E+ R or E+ Cal mice.
Table 5: Caspase-3 protein expression in different animal groups.
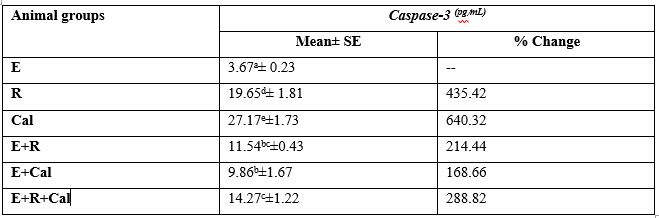
R: Radiation E: Ehrlich Cal: calcitriol
Each value represents Mean± SE (n=15)
Values with dissimilar superscript letters are considered significantly different (p <0.05).
%: Percentage of change from the values in mice bearing Ehrlich solid tumor (E),
Percent change = T-C/C* 100
Tumor proliferation
The volume of solid tumor in the right thigh of mice was measured three times starting after pulp appearance in E mice. The data obtained has been represented in Table (6) and figure (1). Table displays the effect of Calcitriol (Cal), Gamma irradiation (R) and their combination on the growth of solid tumor throughout 28 days’ post EAC inoculation. Treatment of animals with Cal starting after the tumor volume reached 1 cm3 (14 days after tumor transplantation) resulted in marked inhibition of tumor growth as observed at 15, 21 and 28 days from the day of EAC inoculation. The exposure of EAC-bearing mice (E) to γ-irradiation resulted in a considerable reduction in the tumor size. Also, significant reduction of tumor volume was identified in irradiated mice bearing tumor and treated with Cal (E+ R+ Cal mice group) at 15, 21 and 28 days from the day of EAC inoculation compared to that E mice. It worth mention that, the (E +R+ Cal) mice signify the most reduced tumor volume as compared with other groups.
Table 6: Influence of Cal administration or/and exposure to 0.5 Gy gamma radiation on Tumor growth (mm3) in different animal.
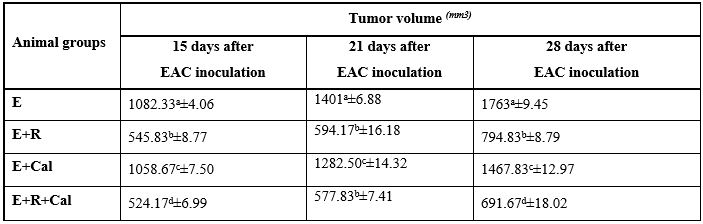
R: Radiation E: Ehrlich Cal: calcitriol
Each value represents Mean± SE (n=15)
Values with dissimilar superscript letters are considered significantly different (p <0.05).
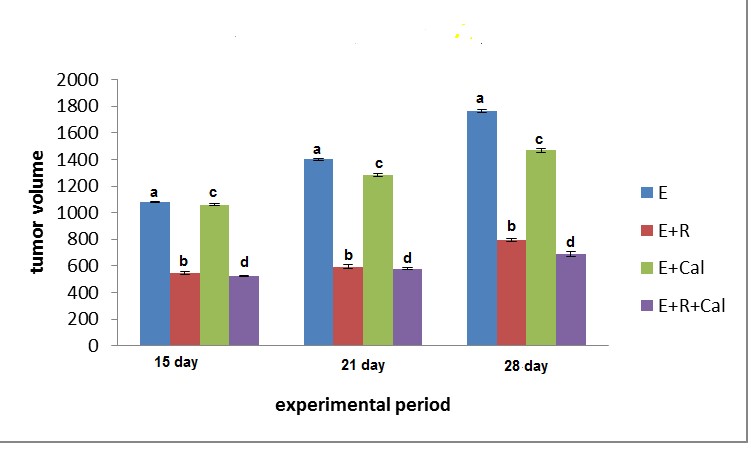
Figure 1: Influence of Cal administration or/and exposure to 0.5 Gy gamma radiation on Tumor growth (mm3) in different animal.
R: Radiation E: Ehrlich Cal: calcitriol
Each value represents Mean± SE (n=15)
Values with dissimilar superscript letters are considered significantly different (p <0.05).
Discussion
The development of original approaches in cancer treatment to arrive new level of success in the continuous battle against cancer required searching deeply in the relative tiny details. One of the interesting tricky points is the relation between estrogens and breast cancer. Our study focused on the aromatase enzyme a rate limiting enzyme in the pathway of estrogen synthesis. Likewise, the present work comprises the examination of calcitriol (vit D3-OH) or/and low dose gamma irradiation as novel approaches targeting the adjustment of aromatase expression, reducing the tumor proliferation and overcoming the disadvantage of using ordinary aromatase inhibitors.
While, aromatase (CYP19) catalyzes the synthesis of estrogens from androgenic precursors and ovaries are the principal source of circulating estrogens in premenopausal women, many other tissues including the breast express aromatase and hence have the capacity to locally synthesize estrogens [34]. Tissue specific promoters differentially regulated by various transcription factors are controlled the aromatase expression in these different situates [35]. Lønning et al. stated that circulating pool of estrogens may be more important for breast carcinogenesis than local estrogen synthesis upregulated in postmenopausal benign breast tissue or malignant breast tumors [36].
In the present study, the data obtained pointed out to significantly higher (p<0.05) mRNA expression of aromatase in solid tumor of murine model (EST bearing mice) compared with its corresponding values in the other mice groups (Table 2). These results could highlight the relation between locally synthesized estrogen and the development of cancer. Zhao et al. disclosed the hyper proliferation and neoplastic damage caused by estrogen and its metabolites [37].
Worth mention, mouse model is not being an exact mirror of estrogen production in humans since mouse aromatase is expressed in fewer tissues than human aromatase [38]. However; this was going over in our work as a highly expressed aromatase gene was observed in solid tumor of mice inoculated with EAC cells (Table 2). The upregulation of aromatase gene expression could be attributed to the presence of multiple exons available for use in aromatase gene. The regulation of tissue specific aromatase gene by alternative uses of these exons has been identified [39]. During cancer development exon 1 termed exon I.4 is often toggled from exon I.4 (1b) to exon I.3 (1c) or exon PII (1d), (1b) which causes enhancement of aromatase expression in cancer-associated adipocytes and fibroblasts. Moreover, the aromatase gene is further regulated at both the transcriptional and post-transcriptional levels through protein kinase A (PKA)-, protein kinase C (PKC)-, and tyrosine kinase receptor-mediated signaling pathways that employ prostaglandin E2 and class 1 cytokines. Epigenetic modifications of the aromatase gene and microRNA mediated aromatase regulation may play a critical role in cancer progression. Several genetic polymorphisms in the aromatase gene may be prognostic factors of disease and may influence response to aromatase inhibitors.
Lately, inhibition of aromatase has become one of the key strategies in breast cancer treatment. In the present work, we recorded a significant decrease in the expression of aromatase in EST mice treated with calcitriol or/and exposed to low dose gamma irradiation (LDR) with remarkable advantage of mice received Cal and exposed to LDR (Table 2). The results obtained signalize obviously the capability of calcitriol to reduce aromatase gene expression even applied lonely or with LDR. As an inception, calcitriol could exert anti proliferative, pro-apoptotic and pro-differentiation actions in several malignant cells including breast cancer cells. Thoroughly, it can inhibit the expression of CYP19A1 gene encoding aromatase enzyme that catalyzes estrogen synthesis from androgenic precursors. In addition, calcitriol repress nuclear receptor responsible for estrogen actions; Era [40]. These mutual actions (opposing the estrogen effect and inhibition of aromatase mRNA expression) trim down the levels of the estrogenic hormones as well as their biological activities within the breast [41]. It was previously mentioned the presence of different promoters distributed over a regulatory region of the CYP19A1 gene permit regulation of the aromatase gene in a tissue selective manner [42]. The main beneficial action of calcitriol is its ability to selectively diminished aromatase transcription. Promoters responsible for aromatase gene expression not only differ between normal tissues but also between normal and cancer tissue. So, tissue-selective CYP19A1 promoters have been proposed as a possible strategy for development of selective aromatase modulators (SAM) [43]. Wheler et al. mentioned that AIs, a main therapeutic approach to treat breast cancer, prevent progression or recurrence of cancer breast in postmenopausal women after primary therapy have a serious disadvantageous of inhibiting estrogen synthesis at any sites as bone [44,45].
Whereas, calcitriol could inhibit the production of estrogen receptors as well as suppress other growth signaling pathway selectively in tumor cells [46]. Furthermore, the improvement recorded in aromatase expression in mice exposed to low dose gamma radiation (Table 2) could be due to enhancement of the immune responses because there are no consequential evidences of direct effect of low level of gamma radiation on the machinery of aromatase gene expression. However, LDR may potentiate the action of Cal on the unique exon’s arrangement in the structure of aromatase gene.
Just about the same issue, our experimental results identify significant increases in the protein expression of COX2 and PGE2 synthesis synchronous with highly expressed aromatase mRNA (Tables 2-4). We hypothesized the correlation between inflammatory response and aromatase gene expression in the development of cancer. We expected the contribution of Inflammation/ Aromatase axis to the increased risk of hormonal developing cancer. One could attribute the changes induced in COX 2 and PGE2 protein to the inflammatory burden implicated after inoculation of EAC cells in the right thigh of normal mice which in turn work to enhance aromatase expression. In agreement with this view Grivennikov et al. reported that the inflammatory responses play influential roles at different stages of tumor development, including initiation, promotion, malignant conversion, invasion, and metastasis [47]. The most, if not all, solid malignancies trigger an intrinsic inflammatory response that builds up a pro tumorigenic micro environment [35]. Tak and Firestein, nominate nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) as a link between inflammation, immunity to cancer development and progression [48]. NF-κB, highly activated at sites of inflammation in diverse diseases, can induce transcription of pro inflammatory cytokines, chemokines, adhesion molecules, matrix metalloproteinases (MMPs), COX2 [49,50].
Cyclooxygenases COX1 and COX2 are rate limiting enzymes in the production of PGE2 from arachidonic acid. The major pathophysiological functions of PGE2 are bringing out pyrexia, pain sensation, and inflammation. So, the analgesic and anesthetic effects of the most widely used non-steroidal anti-inflammatory drugs (NSAIDs) are thought to be driven by inhibition of the production of PGE2. Going to the point, COX-2-derived PGE2 working to stimulates the cyclic adenosine monophosphate (cAMP) to PKA signal transduction pathway that activate CYP19 transcription resulting in increased aromatase expression [51]. Looking upon EST mice received doses of Cal or/and exposed to LDR, significant decreases in the levels of COX2 and the concentration of PGE2 were observed (Table 3,4). Calcitriol might play a noticeable role in preventing the exaggeration of inflammation in the tumor tissue of murine mice. Han et al. stated that pro inflammatory cytokines and COX 2 proteins are significant remarks of in situ active inflammatory responses [52]. Meanwhile the decreases happened in the COX2 protein expression and PGE2 concentration in EST mice received calcitriol treatment denotes its anti-inflammatory capacity. It could be suggested that calcitriol barricade the COX2 inducible cytokines in tumor tissue and consequently checked the production of COX2 in tumor tissue. The COX2 enzymatic activity upregulation could be attributed to the activation of several tumor necrosis factor alfa (TNF-α) dependent intracellular signaling pathway which plays a key role in the control of COX-2 induction. Different protein kinases and transcriptional factors that are greatly regulated by TNF-α are contributed to COX2 upregulation. The expression of COX2 mRNA and protein is often enhanced in various human cell types by inflammatory cytokines such as TNF-α [28]. As COX2 is the key enzyme in pathway of production of pro inflammatory PGs, synthesis and biological actions of PGs significantly withdrawn [41]. Furthermore, calcitriol could decrease PGE2 via enhancing the expression of 15-Hydroxyprostaglandin dehydrogenase (15-PGDH), the enzyme initiating PG catabolism [53].
Besides, low-dose radiation can be beneficial to living organisms, since the mild oxidative stress induced by low-dose radiation induces adaptive responses, including enhanced cell damage repair/protection [3,54]. Thus, the decreases in COX2 expression and PGE2 concentration in EST mice exposed to low dose gamma radiation could be interpreted in the view of the hormetic effect of LDR. Further, in EST mice exposed to LDR and received calcitriol demonstrate prevalent reduction of COX2 and PGE2 expression holding up the view of LDR advancing action of calcitriol.
Our data shows tumor micro environments rich with COX-2 and PGE2 (Table 3,4), elevated tumor apoptosis resistance manifested by decrease in cleaved caspase3 levels (Table 5) as well as tumor enlargement (Table 6). The low concentration of Caspase3 observed in tumor tissue (addressing inhibition of apoptosis) might be considered a consequence event follow COX2 and PGE2 over expression. Favaloro et al. revealed that regulation of apoptosis is linked to several pathophysiological disorders, including autoimmune disorders, Alzheimer’s disease, and cancer [55]. During tumor transformation COX-2 over expression and activation caused increased production of PGE2, which can stimulate tumor growth by enhancing angiogenesis and invasiveness as well as inhibiting immune surveillance and apoptosis [56]. Further the over expressed COX2 up regulate Bcl2 localized to outer membrane of mitochondria leading to inhibition of pro apoptotic proteins Bax and Bak. These proapoptotic factors, work on mitochondrial membrane to promote permobilization and release of cytochrome c which is an important signal in apoptosis cascade.The mitochondrial release of cytochrome c in cytosol has been shown to be rate limiting for the activation of caspases and endonucleases [57]. Li et al. reported that cytosolic cytochrome c activates procaspase-9 by binding to Apaf1 in the presence of dATP, leading to caspase-9 activation and subsequent activation of downstream effector caspases, including caspase-3, with triggering of apoptosis [58].
He et al. stated that Caspases, a family of cysteine proteases that cleave their substrates on the carboxy-terminal side of specific aspartic acid residues [59]. These proteases are generally present in healthy cells as inactive zymogens, but when stimulated they undergo autolytic cleavage to become fully active. Cleaved caspase3 (an executioner caspase) is one of proteases or enzymes can kill the cell by degrading proteins indiscriminately. Thus, it could be mention that COX2 and PGE2 rich microenvironment hinders discharge of cytochrome c to cytosol and afterward suppress apoptotic cascade.
Standing on the previous we have two cohesive points to be followed in concern of calcitriol or and low dose gamma radiation impact against cancer proliferation in EST bearing mice. First which we already discuss is the Over expression of aromatase gene and second is the inhibition of apoptosis in response to highly expressed cycloxygenase2 and PGE2 which we are going to interpret.
The experimental data indicate that calcitriol administration or/and LDR exposure to EST mice induced significant increases in Caspase3 level compared with E mice (Table 5). The reported increases in caspase 3 tailored the promotion of apoptotic process in tumor cells after calcitriol administration. Krishnan et al. affirmed the anticancer effects of Calcitriol due to its ability to suppress proliferation and promotes apoptosis and differentiation of malignant cells and inhibits tumor angiogenesis and invasion [60].
Essentially most pathways which are leading to apoptosis are ultimately regulated by Bcl-2 family of proteins. The Bcl-2 family of proteins is divided into two subclasses that either promote (BclXS, Bax) or suppress (Bcl-2, Bcl-XL, Mcl-1) apoptosis. These proteins can form hetero- and homodimers and the ratio of apoptosis promoters to apoptosis suppressors is one determinant of cellular response. The increases in Bcl-2 expression were observed in a number of cancers including prostate, lung, and breast and is often associated with advanced stages of the disease [61]. Expected upregulation of Bcl2 could be confirmed by the over expression of COX2 and PGE2 (greatly linked toBcL2 prompting) recorded in the present study. On the other hand, the inhibition of this inflammatory signs due to Cal (Table 2,3) could prevent the up regulation of this pre survival proteins. As Bcl2 protein is critical in preventing the cell from initiating apoptosis, calcitriol have been sought to provide potential anti-cancer therapies by making cancer cells more susceptible to apoptosis-inducing agents.
In addition, the improvement in Caspase 3 expression in EST mice exposed to LDR could interpreted in the view of LDR hormetic effect. The LDR activation of apoptosis could be executed in cancer cells by different mechanisms like inhibition of inflammatory signs, inhibition of aromatase as well as promotion of proapoptotic proteins3,4. Xin et al. reported that LDR induces apoptosis of tumor cells and has numerous beneficial effects on normal tissues, including radiation homeostasis and adaptive response [62].
In conclusion Calcitriol and low dose gamma irradiation could introduce transferable potentiating effect towards the cancer proliferation via selective suppression of tumor tissue m-RNA aromatase and amendment of inflammatory response which trigger apoptosis in tumor cells in murine model of EST. This may represent a prospective therapeutic policy for the management of solid tumor and decreasing the possibilities of tumor therapeutic resistance.
Conflicting Interests
The Authors declare that there is no conflict of interest.
References
- Sudhakar A. History of cancer, ancient and modern treatment methods. J. Cancer Sci. Ther. 2009; 1: 1–4.
- Falzone L, Salomone S, Libra M. evolution of Cancer Pharmacological Treatments at the Turn of the Third Millennium. Front Pharmacol. 2018; 9: 1300.
- El Bakary NM, Alsharkawy AZ, et al. Role of Bee Venom and Melittin on Restraining Angiogenesis and Metastasis in γ-Irradiated Solid Ehrlich Carcinoma-Bearing Mice. Integr Cancer Ther. 2020; 19: 1534735420944476.
- Hafez EN, Moawed FS, et al. Gamma Radiation-Attenuated Toxoplasma gondii Provokes Apoptosis in Ehrlich Ascites Carcinoma-Bearing Mice generating long-Lasting Immunity. Tech Cancer Res&Treat. 2020; 19: 1-11.
- Henderson BE, Feigelson HS. Hormonal carcinogenesis. Carcinogenesis. 2000; 21(3): 427–433.
- Yager JD. Endogenous estrogens as carcinogens through metabolic activation. J Natl Cancer Inst Monogr. 2000; 27: 67–73.
- Kaminska M, Ciszewski T, et al. Breast cancer risk factors. Prz Menopauzalny. 2015; 14(3): 196–202.
- Sun YS, Zhao Z, Yang ZN. Risk factors and preventions of breast cancer. Int J Biol Sci. 2017; 13(11): 1387–1397.
- Ozsoy A, Barca N, Dolek BA. The relationship between breast cancer and risk factors: a single-center study. Eur J Breast Health. 2017; 13(3): 145–149.
- Lewis-Wambi JS and Jordan VC. Estrogen regulation of apoptosis: how can one hormone stimulate and inhibit? Breast Cancer Res 2009; 206: 1-12.
- Chen S. Aromatase and breast cancer. Front Biosci 1998; 6: d922-923.
- Brodie A, Long Band Lu Q. Aromatase expression in normal breast. Breast cancer Res Treat1998; 49: S85-91.
- Iwase H. Current topics and perspectives on the use of aromatase inhibitors in the treatment of breast cancer. BreastCancer 2008; 15: 278-290.
- Lønning PE, Geisler J, Bhatnager A. Development of aromatase inhibitors and their pharmacologic profile. Am J Clin Oncol. 2003; 26(4 Suppl 1): S3–S8.
- Dombernowsky P, Smith I, Falkson G, et al. Letrozole, a new oral aromatase inhibitor for advanced breast cancer: double-blind randomized trial showing a dose effect and improved efficacy and tolerability compared with megestrol acetate. J Clin Oncol. 1998; 16(2): 453–461.
- Chumsri S, Howes T, Bao T, et al. Aromatase, Aromatase Inhibitors, and Breast Cancer. J Steroid Biochem Mol Biol 2011; 125: 13-22.
- Jeon S, Shin E. Exploring vitamin D metabolism and function in cancer. Exp Mol Med. 2018; 50(4): 20.
- Saracligil B, Ozturk B, Unlu A, et al. The effect of vitamin D on MCF-7 breast cancer cell metabolism. BratislLekListy 2017; 118: 101-106.
- Welsh J, Wietzke JA, Zinser GM, et al. Vitamin D-3 receptor as a target for breast cancer prevention. J Nutr, 2003; 133: 2425S–2433S.
- Feinendegen LE. Evidence for beneficial low-level radiation effects and radiation hormesis. Br J Radiol 2005; 78: 3–7.
- Dar TB, Henson RM, Shiao SL. Targeting Innate Immunity to Enhance the Efficacy of Radiation Therapy. Front Immunol. 2018; 9: 3077.
- Calabrese EJ, Baldwin LA. Scientific foundations of hormesis. Crit Rev Toxicol.2001;31(4 & 5): 351-624.
- Dobrzyński L, Fornalski KW, Socol Y, et al. Modeling of Irradiated Cell Transformation: Dose- and Time-DependentEffects. Radiat Res 2016; 186: 396–406.
- Brueggemeier RW, Su B, Sugimoto Y, et al. Aromatase and COX in breast cancer enzyme inhibitor and beyond. J Steroid Biochem Mol Biol 2007; 106: 16-23.
- Dannenberg AJ, Altorki NK, et al. Cyclo-oxygenase 2: a pharmacological target for the prevention of cancer. Lancet Oncol 2001, 2:544-551.
- Harris RE, Chlebowski RT, Jackson RD, et al. Breast cancer and non-steroidal anti-inflammatory drugs: prospective results from the Women’s Health Initiative. Cancer Res 2003; 63: 6096-6101.
- Medhat AM, Azab KS, Said MM, et al. Antitumor and radiosensitizing synergistic effects of apigenin and cryptotanshinone against solid Ehrlich carcinoma in female mice. TumourBiol 2017; 39: 1–13.
- Azab KS, Meky NH, El- Deghidy EAM, et al. Response of COX2/PGE2 Inflammatory pathway to brown Seaweed Extract in Rats Exposed to Gamma Radiation. World J Nucl SciTechnol2017; 7: 189-205.
- Milliken EL, Zhang X, Flask C, et al. EB1089, a vitamin D receptor agonist, reduces proliferation and decreases tumor growth rate in a mouse model of hormone-induced mammary cancer. Cancer Lett 2005; 229: 205–215.
- Papadopoulos D, Kimler BF, Estes NC, et al. Growth delay effect of combined interstitial hyperthermia and branchytherapy in a rat solid tumor model. Anticancer Res 1989; 9: 45-47.
- Rödel F, Frey B, Gaipl U, et al. Modulation of inflammatory immune reactions by low-dose ionizing radiation: molecular mechanisms and clinical application. Curr Med Chem 2012; 19:1741-1750.
- Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method.%J. methods,%25, 2001;402-408.
- Kulmacz RJ and Lands WE. Requirements for Hydroperoxide by the Cyclooxygenase and Peroxidase Activities of Prostaglandin H Synthase. Prostaglandins 1983; 25: 531-540.
- Simpson E, Santen RJ. Celebrating 75 years of oestradiol. J Mol Endocrinol 2015; 55: T1–T20.
- Simpson ER, Clyne C, Rubin G, et al. Aromatase-a brief overview. Annu Rev Physiol 2002; 64: 93–127.
- Lønning PF, Haynes BP, Straume AH, et al. Exploring breast cancer estrogen disposition: the basis for endocrine manipulation. Clin Cancer Res 2011; 17: 4948–4958.
- Zhao H, Zhou L, Shangguan AJ, et al. Aromatase expression and regulation in breast and endometrial cancer. J Mol Endocrinol 2016; 57(1): R19–R33.
- Zhao H, Pearson EK, Brooks DC, et al. A humanized pattern of aromatase expression is associated with mammary hyperplasia in mice. Endocrinology 2012; 153: 2701–2713.
- Bulun SE, Chen D, Moy I, et al. Aromatase, breast cancer and obesity: a complex interaction. Trends Endocrinol Metab 2012; 23: 83-89.
- Swami S, Krishnan AV, Feldman D. 1alpha, 25-Dihydroxy vitamin D3 down-regulates estrogen receptor abundance and suppresses estrogen actions in MCF-7 human breast cancer cells. Clin Cancer Res 2000; 6: 3371–3379.
- Krishnan AV, Swami S, Peng L, et al. Tissue selective regulation of Aromatase expression by calcitriol: implications for breast cancer therapy. Endocrinology 2010; 151: 32-42.
- Bulun SE, Simpson ER. Aromatase expression in women's cancers. Adv Exp Med Biol 2008; 630: 112–132.
- Lundqvist J, Norlin M, Wikvall K. 1α, 25-Dihydroxyvitamin D3 exerts tissue-specific effects on estrogen and androgen metabolism. Biochim Biophys Acta 2011; 1811: 263–270.
- Wheler J, Johnson M, Seidman A. Adjuvant therapy with aromatase inhibitors for postmenopausal women with early breast cancer: evidence and ongoing controversy. Semin Oncol 2006; 33: 672–680.
- Confavreux CB, Fontana A, Guastalla JP, et al. Estrogen-dependent increase in bone turnover and bone loss in postmenopausal women with breast cancer treated with anastrozole. Prevention with bisphosphonates. Bone 2007; 41: 346–352.
- Welsh J. Targets of vitamin D receptor signaling in the mammary gland. J Bone Miner Res 2007; 22: V86–V90.
- Grivennikov SI, Greten FR, Karin M. Immunity, Inflammation, and Cancer. Cell 2010; 140: 883–899.
- Mantovani A, Allavena P, Sica A, et al. Cancer-related inflammation. Nature 2008; 454: 436–444.
- Tak PP, Firestein GS. NF-kB: a key role in inflammatory diseases. J ClinInvest 2001; 107: 7-11.
- Han ZN, Boyle DL, Manning AM, et al. AP-1 and NF-kappa B regulation in rheumatoid arthritis and murine collagen-induced arthritis. Autoimmunity 1998; 28: 197–208.
- Afify SM, Seno M. Conversion of Stem Cells to Cancer Stem Cells: Undercurrent of Cancer Initiation. Cancers (Basel). Epub ahead of print 11 march 2019.
- Subbaramaiah K, Morris PG, Zhou XK, et al. Increased levels of COX-2 and prostaglandin E2 contribute to elevated aromatase expression in inflamed breast tissue of obese women. Cancer Discov 2012; 2: 356–365.
- Swami S, Krishnan AV, Moreno J, et al. Calcitriol and Genistein Actions to Inhibit the Prostaglandin Pathway: Potential Combination Therapy to Treat Prostate Cancer. J Nutr 2007; 137: 205S–210S.
- Kojima S, Tsukimoto M, Shimura N, et al. Treatment of Cancer and Inflammation with Low-Dose Ionizing Radiation: Three Case Reports. Dose Response 2017; 15: 1559325817697531.
- Favaloro B, Allocati N, Graziano V, et al. Role of Apoptosis in disease. Aging (Albany NY) 2012; 4: 330–349.
- Kuo CL, Chi CW , Liu TY. Modulation of Apoptosis by Berberine through Inhibition of Cyclooxygenase-2 and Mcl-1 Expression in Oral Cancer Cells. In Vivo 2005; 19: 247-252.
- Martinou JC, Desagher S, Antonsson B. Cytochrome c release from mitochondria: all or nothing. Nat Cell Biol 2000; 2: E41–E43.
- Li P, Nijhawan D, Budihardjo I, et al. Cytochrome c and dATP-dependent formation of Apaf-2/Caspase-9 complex initiates an apoptotic protease cascade. Cell 1997; 91: 479–489.
- He B, Lu N, Zhou Z. Cellular and Nuclear Degradation during Apoptosis. Curr Opin Cell Biol 2009; 21: 900–912.
- Krishnan AV, Swami S, Feldman D. The potential therapeutic benefits of vitamin D in the treatment of estrogen receptor positive breast cancer. Steroids 2012; 27: 1107-1112.
- Blutt SE, McDonnell TJ, Polek TC, et al. Calcitriol-induced apoptosis in LNCaP cells is blocked by overexpression of Bcl-2. Endocrinology 2000; 141: 10–17.
- Xin Y, Zhang HB, Tang TY, et al. Low‑dose radiation‑induced apoptosis in human leukemia K562 cells through mitochondrial pathways. Mol Med Rep 2014; 10: 1569-1575.

