Two Make Company – When Less Is More and Double Is Better – Evidence to Practice of Antiplatelet Therapy in Stroke Care
Wagner M1, Borutta MC2 and Koehn J1,*
¹Department of Neurology, ANregiomed, Ansbach
²Department of Neurology, University Hospital Erlangen-Nuremberg, Erlangen
Received Date: 07/11/2025; Published Date: 10/12/2025
*Corresponding author: Dr. med. Julia Koehn, PD, Department of Neurology, ANregiomed, Escherichstr. 1, 91522 Ansbach, Germany
Abstract
According to the World Stroke Organisation (WSO), globally more than 77 million patients suffer from stroke [1], with nearly one quarter being recurrent events. Of these, 87% are etiologically linked to modifiable risk factors [2]. Current guidelines recommend to treat affected patients on certified Stroke Units (STUs); while STU coverage varies widely by regions, of now, there are thousands of STUs worldwide. A key component of STU care is the management of cerebro- and cardiovascular risk factors as part of secondary prevention. Initiating antiplatelet therapy (APT), either as monotherapy or dual therapy, represents one of the cornerstones of treatment for non-cardioembolic stroke.
This review chapter summarizes essential aspects of Dual Antiplatelet Therapy (DAPT):
(i) pivotal studies on common indications for DAPT (non-cardioembolic, minor strokes and transient ischemic attacks [TIA], evidence comparing DAPT with thrombolytic therapy, and DAPT in specific scenarios — such as symptomatic intracranial stenosis and extracranial carotid artery stenosis), (ii) recent evidence from 2025 DAPT studies, and (iii) a practical summary addressing key questions: who benefits from DAPT, when should therapy be initiated, how long should it be continued — and where are its limitations.
Keywords: Iischemic stroke; Transient ischemic attack; Antiplatelet therapy; Secondary prevention
List of Abbreviations: STU: Stroke Unit; DAPT: Dual Antiplatelet Therapy; HR: Hazard Ratio; ARR: Absolute Risk Reduction; NNT: Number Needed to Treat; NIHSS: National Institute of Health Stroke Scale; TIA: Transient Iachemic Attack; ABCD²: Clinical prediction score to determine the risk for stroke in the days following a transient ischemic attack; NNH: Number Needed to Harm; mRS: modified Ranking Scale; INR: International Normalized Ratio; AMM: aggressive medical management; BP: Blood Pressure; LDL: Low-Density Lipoprotein; CEA: Carotid Endarterectomy; CAS: carotid artery stenting; OMT: Optimized Medical Therapy; ESRS: Essen Stroke Risk Score; ICASB: Intracranial Atherosclerosis Burden; ICA Anterior Circulation; PCA: Posterior Circulation; CAR: Carotid Artery Risk
Common Indications for DAPT
DAPT in Non-Cardioembolic Minor Stroke and TIA - A Decade of Data
Evidence supporting the use of Dual Antiplatelet Therapy (DAPT) in ischemic stroke dates back more than 10 years. In 2013, the CHANCE trial provided the first indications from Chinese study data that dual therapy might offer benefits [3]. Five years later, the POINT trial confirmed these findings using international data, albeit with a slightly longer treatment duration [4]. In 2019, pooled data from both studies were published, concluding that the main benefit of DAPT occurs early—within the first few days after the event [5]. Subsequently, evidence accumulated rapidly: in 2020, the THALES trial with ticagrelor [6]; in 2021, CHANCE-2 investigating the impact of a genetic background — specifically CYP2C19 — on treatment efficacy [7]; in 2023, INSPIRES extended the initiation window for DAPT to 72 hours [8]; and most recently, in 2024, ATAMIS examined patients with more severe strokes [9]. Collectively, these studies have generated highly relevant data over a decade, directly influencing daily clinical practice.
In 2013, the multicenter, randomized, double-blind CHANCE trial enrolled over 5,000 patients to assess the benefit of early (within 24 hours of symptom onset) and short-term DAPT (aspirin + clopidogrel for 21 days), followed by clopidogrel monotherapy until day 90, compared with aspirin plus placebo. Results showed a clear reduction in recurrent strokes, with the greatest benefit within the first few days and no relevant increase of major bleeding complications; NNT≈29. Notably, patients were enrolled very early after the event, aspirin dosing was 75mg, and generalizability was limited due to the exclusively Asian cohort [3].
POINT also demonstrated short-term benefits of DAPT (primarily within the first few days), followed by an increased bleeding risk. The trial included 4,881 patients randomized within 12 hours after symptom onset. The DAPT group received clopidogrel 600mg on day 1, then 75mg/day until day 90, plus aspirin 50–325 mg/day (dose determined by center). The control group received placebo plus aspirin at the same dose range. The composite vascular endpoint (stroke, myocardial infarction, vascular death) occurred in 5.0% of the DAPT group versus 6.5% in the aspirin group (HR 0.75, p=0.02). And again, the main benefit occurred within the first week after randomization (ARR 1.5 percentage points; NNT≈67). Major bleeding complications occurred in 0.9% vs 0.4% respectively (HR 2.32, p=0.02) [4].
Pooled analyses from CHANCE and POINT confirmed earlier findings: DAPT is effective and safe within the first 21 days after ischemic stroke, with the greatest benefit during the early phase after the event (event rate reduction DAPT vs aspirin monotherapy: day 0–21: 5.2% vs 7.8%, HR 0.66, p < 0.001; day 0–10: 4.7% vs 7.1%, HR 0.65, p < 0.001) [5].
THALES was the first trial to evaluate ticagrelor in combination with aspirin for DAPT. With a NNT of 91, again a benefit was found in patients with non-cardioembolic minor stroke (NIHSS ≤5) or high-risk TIA (ABCD² ≥6) regarding the primary endpoint (stroke — ischemic or hemorrhagic — or death) at 30 days, thus offering an alternative to clopidogrel, particularly in cases of clopidogrel nonresponse [6].
With this background, CHANCE-2 (multicenter, randomized, double-blind, placebo-controlled, 202 centers in China) further analyzed the use of ticagrelor + aspirin compared to clopidogrel + aspirin in carriers of CYP2C19 loss-of-function alleles (*2/*3). Inclusion criteria: minor stroke or high-risk TIA, therapy initiation ≤24 hours. Regarding recurrent strokes, the authors suggested a ticagrelor-based DAPT benefit (ARR of 1.6%), however, it should be noted that this purely Asian study cohort had an above-average LOF allele frequency compared to other ethnicities, thus limiting generalizability of the results [7].
Even when therapy was initiated later — within 72 hours after stroke onset — the INSPIRES data demonstrated a benefit of DAPT over aspirin monotherapy (HR 0.79, NNT≈53) in patients with minor stroke (NIHSS ≤3) or high-risk TIA (ABCD² ≥4). Within this study, most participants (87.2%) were enrolled between 24–72 hours post-stroke (12.8% ≤24 hours). Consistent with prior data, benefit was greatest within the first 3 weeks (recurrent stroke until 90 days: 7.3% [DAPT] vs 9.2% [aspirin], p=0.008). Moderate to severe bleeding occurred in 0.9% (DAPT) vs 0.4% (aspirin), yielding a NNH of 200 [8].
Most recently, the potential target population was expanded as part of the ATAMIS study which enrolled patients with moderately severe stroke (NIHSS 4–10). The primary efficacy endpoint at day 7 (early neurological deterioration, NIHSS increase ≥2 points not due to bleeding) occurred in 4.8% of the DAPT group versus 6.7% of the aspirin group. However, this early moderate benefit did not translate into improved functional outcome at 90 days, where no differences were observed between groups [9].
Use of DAPT Compared to Thrombolytic Therapy
The rationale for research comparing DAPT with intravenous thrombolysis lies in the lack of convincing evidence for the efficacy of i.v. thrombolysis in patients with minor stroke (NIHSS ≤3), whereas the use of DAPT has been extensively studied (see studies above).
The multicenter, randomized ARAMIS trial was the first to directly compare DAPT (clopidogrel + aspirin) with i.v. alteplase in a non-inferiority design regarding the primary endpoint of a favorable functional outcome (mRS 0–1) at 90 days. Study design: Inclusion of patients with acute ischemic stroke, NIHSS ≤5, within 4.5 hours of symptom onset; Group 1: i.v. alteplase (0.9mg/kg), followed by guideline-based antithrombotic therapy after 24 hours; Group 2: clopidogrel + aspirin for 12 days, then monotherapy. And the findings: DAPT appeared non-inferior to i.v. thrombolysis in minor stroke (mRS 0–1 at 90 days: DAPT 93.8%, alteplase 91.4%), with a more favorable safety profile (symptomatic intracerebral hemorrhage: DAPT 0.3%, alteplase 0.9%; any bleeding complication: DAPT 6/371, alteplase 19/352). Regarding limitations, again the restricted generalizability (participants from 38 centers in China) should be mentioned. Furthermore, the study compared DAPT against the active standard (alteplase), therefore conclusions regarding possible efficacy in comparison with placebo are precluded. Finally, the crossover rate was remarkably high (20%), and the study lacks subgroup analyses, e.g., patients with vessel occlusions. Nevertheless, after ARAMIS DAPT appears to be a safe and effective alternative to i.v. thrombolysis in this selected patient population [10].
DAPT in Intracranial Symptomatic Stenosis
Early evidence has set the tone regarding therapeutic approaches for stroke patients with underlying symptomatic intracranial stenosis. Following the WASID trial (2005), warfarin was abandoned as an option for these cases, and antiplatelet therapy was confirmed as treatment standard [11]. In 2011, SAMMPRIS demonstrated that aggressive medical management (cholesterol, blood pressure, lifestyle) combined with short-term DAPT for 3 months seems generally superior to early stenting [12]. These findings clearly led to guideline recommendations against routine stenting in symptomatic intracranial stenosis ≥70%. However, subgroup analyses of the studies mentioned above did not provide consistent evidence for a specific pharmacological regimen in symptomatic intracranial stenosis.
WASID compared warfarin (target INR 2.0–3.0) with aspirin (2 × 650mg/day) in 569 patients within 30 days after an ischemic event and 50–99% symptomatic intracranial stenosis, randomized across 59 U.S. centers. Regarding the primary endpoints (ischemic stroke, intracranial hemorrhage, or vascular death), anticoagulation offered no benefit; in fact, the trial was terminated early due to safety concerns [11].
Data from SAMMPRIS have also been standard practice for years. In 2011, 451 patients with symptomatic 70–99% intracranial stenosis (TIA/stroke ≤30 days) were randomized 1:1 into the two study arms in this multicenter trial: to either PTAS (Wingspan stent) + aggressive medical management (AMM) or AMM alone. AMM included a long-term therapy with aspirin 325mg/day, clopidogrel 75mg/day for 90 days, intensive risk factor control (SBP <140 mmHg or <130 mmHg in diabetes; LDL <70 mg/dL with statines), and a structured lifestyle program. Primary endpoints included stroke or death within 30 days after randomization (or post-procedure), and subsequent territorial ischemic stroke until study end. Data revealed that PTAS had a 30-day primary endpoint risk >14%, compared to <6% within the AMM alone group. Long-term outcomes also favored medical management, and bleeding complications were significantly lower with AMM (5/227 [2.2%] vs. 22/224 [9.8%]; p<0.001). It should be mentioned that these results were largely driven by high periprocedural complication rates in PTAS. Nevertheless, the results led to guideline recommendations that are still valid today i) against routine stenting of symptomatic intracranial stenosis ≥70%, ii) stenting should be reserved for selected cases of AMM failure, iii) in addition to 90 days of DAPT (aspirin + clopidogrel), successful AMM includes intensive LDL and BP control [12].
With regard to the management of patients with intracranial stenosis, subgroup analyses of further studies did not provide a consistent picture. The corresponding data from CHANCE did not prove a clear additional benefit of DAPT over aspirin monotherapy [3]. In THALES, the combination therapy appeared superior in atherosclerotic stenosis, but the study also included extracranial plaques [6]. Finally, CHANCE-2 did not demonstrate superiority of ticagrelor over clopidogrel in this subgroup [7]. Although these data did not confirm superiority of any specific pharmacological regimen, current recommendations support early-phase DAPT in patients with intracranial stenosis, alongside aggressive risk factor management as the cornerstone of therapy.
DAPT in Extracranial Carotid Stenosis
For antithrombotic therapy in patients with symptomatic internal carotid artery (ICA) stenosis, the primary reference is the 2023 European Society for Vascular Surgery – ESVS – guideline [13]. Here, in patients with Carotid Endarterectomy (CEA), aspirin therapy is routinely recommended perioperatively, while DAPT may be considered in cases of high embolic risk. In contrast, it is well known that DAPT (aspirin + clopidogrel) for ≥4 weeks is recommended after Carotid Artery Stenting (CAS), followed by lifelong single antiplatelet therapy, preferably clopidogrel 75mg daily. In case no immediate revascularization is planned, short-term DAPT for 21 days followed by clopidogrel monotherapy is recommended after TIA or minor stroke. This reflects the expedited European Stroke Organization recommendation and is referenced in the ESVS guideline.
The German S3 guideline on extracranial carotid stenosis (2020, currently under revision) also implemented DAPT [14], recommending that patients presenting very early (within 12 hours of symptom onset) with mild neurological deficits (high-risk TIA or stroke with NIHSS ≤4) may be considered for DAPT (aspirin + clopidogrel) for 10–21 days.
Most recent data on this topic comes from ECST-2, which compares optimal medical therapy versus optimal medical therapy plus revascularization in (also) symptomatic stenoses; these results are discussed in section 3.
Current DAPT Data – Update from 2025
The following section summarizes recent studies and sub-analyses on DAPT and its implications for daily practice, particularly regarding risk stratification and additional diagnostic criteria published in 2025. From the INSPIRES trial [15, 16, 17, 18], several post-hoc analyses were published focusing on risk stratification to enable more targeted patient selection. Another study examined the benefit of DAPT in relation to intracranial atherosclerosis burden, promoting a step towards imaging-guided patient selection [19]. Real-world data explored topography-driven DAPT benefit (20), and finally ECST-2 reaffirmed the urgency of optimized medical therapy (OMT) in patients with low/intermediate recurrence risk [21].
In a post-hoc analysis, Zhang et al. analyzed DAPT effectiveness using the Essen Stroke Risk Score (ESRS). Patients with ESRS <3 showed clear superiority of DAPT during the recommended treatment period (HR 0.67), whereas those with ESRS ≥3 had no demonstrable advantage over aspirin monotherapy (HR 0.92). These seemingly paradoxical results — one might expect high-risk patients to benefit most from intensified therapy — were attributed to two factors: i) higher incidence of non-platelet-mediated events (e.g., cardiac embolism, progression of other vascular disease) in high ESRS patients, reducing DAPT responsiveness; ii) increased bleeding complications in this group, diminishing overall benefit. As a practical message from the analyses, it is suggested that the ESRS may be used as a simple decision-making aid: ≤72h DAPT particularly for ESRS <3 [15].
A further sub-analysis of the INSPIRES data showed that DAPT significantly reduced recurrent stroke risk in patients with newly diagnosed diabetes, whereas those with long-standing diabetes did not benefit. One possible explanatory model focused on early pathophysiological mechanisms (hyperglycemia-triggered platelet activation, inflammation, endothelial stress) that remain modifiable shortly after diagnosis. In contrast, chronic diabetes involves non-platelet-mediated or structural etiologies (diffuse atherosclerosis, microangiopathy) and comorbidities. Although hypothesis-driven, these findings support a certain degree of phenotype-guided patient selection for DAPT [16].
In a further post-hoc analysis with pathophysiological considerations, Wang et al. investigated the impact of baseline neutrophil count on DAPT benefit within the early post-stroke phase. Within the group with a low neutrophil count, no additional benefit of DAPT over aspirin monotherapy was observed, whereas the authors demonstrated a significant benefit in the middle tertile group (HR~0.55). Finally, in the group with a high neutrophil count, DAPT effects remained significant without increased bleeding risk. This analysis also discussed possible immunological aspects in stroke etiology: neutrophils represent a simple inflammation marker — greater inflammatory-thrombotic activation may predict stronger DAPT benefit [17].
Although the data from an INSPIRES post-hoc analysis on metabolic syndrome did not yield any further “user-relevant” results, the statement by Zhang Xia et al. is worth mentioning in brief, in that the presence of metabolic syndrome does not appear to have a relevant effect on the relative benefit or moderate-to-severe bleeding risk associated with initiating DAPT in the early phase after stroke [18].
In a multicenter prospective study, Liu et al. examined the impact of intracranial atherosclerosis burden (ICASB) on the potential benefit of DAPT in minor stroke. ICASB was calculated based on i) the number of affected vessels, ii) the degree of stenosis, and iii) the sum across all vessels (ICASB ≥4 = high burden). As primary outcome, the authors defined recurrent infarction (ischemic or hemorrhagic) at 90 days and 1 year post-index event. Compared to antiplatelet mono-therapy, DAPT showed no advantage in terms of the primary endpoints in the overall cohort. However, patients with a high ICAS score showed a clear advantage from initiating DAPT, with an absolute risk reduction of ~7 percentage points (NNT≈14). There was no DAPT advantage in patients with low ICAS burden (HR~1.1), and with regard to safety aspects, there was no significant interaction between ICAS burden and the occurrence of intracranial hemorrhage. These findings reinforce evidence supporting DAPT in minor stroke patients with underlying intracranial stenosis/atherosclerosis [19].
A propensity-matched analysis of REDAPT data compared early DAPT initiation (≤ 48 h) between patients with anterior (ICA) versus posterior circulation (PCA) ischemia. Although not RCT data, findings suggest similar 90-day recurrent infarction rates under early DAPT after minor stroke, regardless of the vascular territory (≈3.1% ICA vs 2.9% PCA), providing no basis for topography-driven restrictions on DAPT use [20].
The 2-year interim results of ECST-2 were published to address outdated evidence on revascularization and evolving medical management. This prospective, randomized trial compared optimized medical therapy (OMT) versus OMT plus revascularization (CEA or stenting) in patients with ≥50% ICA stenosis (asymptomatic or symptomatic with low/intermediate recurrence risk). At 30 centers in Canada and Europe, 429 patients were randomized. The authors stratified risk according to the CAR score (Carotid Artery Risk), that estimates individual 5-year ipsilateral stroke risk under OMT. Regarding OMT, strict LDL and BP targets were defined, and patients received short-term DAPT (3 months after TIA/minor stroke, or up to 6 weeks post-CAS). Using a Win-Ratio-Model, the primary endpoint “death or fatal stroke” was weighted and defined. 2 years after randomization, results did not deliver an additional benefit of revascularization over OMT alone (Win-Ratio 1.01; 95% CI 0.60–1.70; p=0.97). Furthermore, a subgroup analysis – asymptomatic vs symptomatic ICA stenosis – did not show significant differences between therapeutic strategies, so that for CAR score <20% (low/intermediate risk), optimal risk factor control appears essential and not inferior to revascularization at 2 years. Strategies for higher-risk patients require individual assessment, and 5-year data are pending. Finally, it should be noted that within ECST-2, DAPT was standardized only as part of OMT, and alternative treatment durations were not studied. Given evidence for net benefit primarily within the first 21–30 days post-event and increasing bleeding risk with prolonged therapy, the optimal DAPT duration in carotid stenosis under modern OMT remains an open question for future research [21].
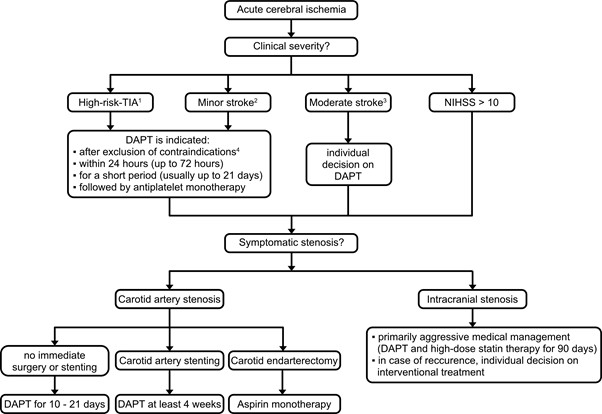
Figure: Algorithm for decision-making on DAPT in acute ischemic stroke. (DAPT, dual antiplatelet therapy [1]; ACBD2-Score ≥ 6 according to THALES trial or ≥ 4 according to POINT trial [2]; NIHSS ≤ 5 according to THALES trial or ≤ 3 according to POINT trial [2]; NIHSS 4 – 10 according to ATAMIS trial [4]; for example, non-cardioembolic etiology).
Conclusion, Practical Application, and Outlook
Based on the presented data, the following key points can be summarized:
- In the absence of contraindications, antiplatelet therapy should be initiated early in patients with minor stroke and high-risk TIA, starting with short-term dual therapy —typically aspirin and clopidogrel for 21 days — followed by single antiplatelet therapy.
- Moderate Stroke (NIHSS 4–10): DAPT shows an early stabilizing effect; however, a clear 90-day functional benefit is not proven, requiring individualized decision-making.
- Even with a delayed DAPT initiation (24–72 hours) a persistent benefit is observed, though safety considerations become increasingly important.
- In intracranial stenosis, an aggressive medical management remains the standard, including high-dose statin therapy and DAPT for 90 days.
- In symptomatic ICA stenosis without immediate CEA or CAS, DAPT for 10–21 days is recommended; after CAS, at least 4 weeks of DAPT; after CEA, usually aspirin monotherapy.
- In the setting of “minor stroke, non-disabling deficits within 4.5 hours”, DAPT appears non-inferior to thrombolysis, likely due to stabilization and secondary prevention. Careful patient selection remains essential.
- Choice of P2Y12 inhibitor: clopidogrel remains standard; ticagrelor is an option in CYP2C19 loss-of-function or extensive atherosclerosis.
Looking Ahead:
Future strategies will likely involve more targeted DAPT use, with imaging playing a central role — as higher ICAS burden may correlate with greater benefit.
Therapy duration will require further refinement: current evidence supports maximum efficacy in the early secondary prevention window (10–21 days), though effects beyond 30 days remain plausible. In the future, balancing benefit against bleeding risk even more precisely will be critical.
ECST-2 data suggest that in low/intermediate-risk carotid stenosis, optimized medical therapy (including DAPT and statin) is a reasonable alternative to early revascularization. Whether prolonged DAPT is beneficial in selected extracranial ICA stenosis cases remains to be clarified in future trials.
Real-world data from 2025 provide insights into subgroup-specific considerations (anterior vs posterior circulation, metabolic syndrome, inflammatory markers), potentially refining indications even further.
In short: “Double yes — but targeted, short, and smart.”
Author Contributions: M.W. and J.K.: concept and design, literature acquisition and analysis, drafting the article, final approval of the submitted version; M.C.B.: concept and design, revising the article, final approval of the submitted version
Competing Interests: none of the authors report COIs
Grant Information: N/A
References
- Parry-Jones AR, Krishnamurthi R, Ziai WC, Shoamanesh A, Wu S, Martins SO, et al. World Stroke Organization (WSO): Global intracerebral hemorrhage factsheet 2025. Int J Stroke, 2025; 20(2): 145-150. doi: 10.1177/17474930241307876.
- Yang L, Zheng B, Gong Y. Global, regional and national burden of ischemic heart disease and its attributable risk factors from 1990 to 2021: a systematic analysis of the Global Burden of Disease study 2021. BMC Cardiovasc Disord,. 2025; 25(1): 625. doi: 10.1186/s12872-025-05022-x.
- Wang Y, Pan Y, Zhao X, Li H, Wang D, Johnston SC, et al. CHANCE Investigators. Clopidogrel With Aspirin in Acute Minor Stroke or Transient Ischemic Attack (CHANCE) Trial: One-Year Outcomes. Circulation, 2015; 132(1): 40-46. doi: 10.1161/CIRCULATIONAHA.114.014791.
- Johnston SC, Easton JD, Farrant M, Barsan W, Conwit RA, Elm JJ, et al. Clinical Research Collaboration, Neurological Emergencies Treatment Trials Network, and the POINT Investigators. Clopidogrel and Aspirin in Acute Ischemic Stroke and High-Risk TIA. N Engl J Med, 2018; 379(3): 215-225. doi: 10.1056/NEJMoa1800410.
- Pan Y, Elm JJ, Li H, Easton JD, Wang Y, Farrant M, et al. Outcomes Associated with Clopidogrel-Aspirin Use in Minor Stroke or Transient Ischemic Attack: A Pooled Analysis of Clopidogrel in High-Risk Patients with Acute Non-Disabling Cerebrovascular Events (CHANCE) and Platelet-Oriented Inhibition in New TIA and Minor Ischemic Stroke (POINT) Trials. JAMA Neurol, 2019; 76(12): 1466-1473. doi: 10.1001/jamaneurol.2019.2531.
- Johnston SC, Amarenco P, Denison H, Evans SR, Himmelmann A, James S, et al. THALES Investigators. Ticagrelor and Aspirin or Aspirin Alone in Acute Ischemic Stroke or TIA. N Engl J Med, 2020; 383(3): 207-217. doi: 10.1056/NEJMoa1916870.
- Wang Y, Meng X, Wang A, Xie X, Pan Y, Johnston SC, et al. CHANCE-2 Investigators. Ticagrelor versus Clopidogrel in CYP2C19Loss-of-Function Carriers with Stroke or TIA. N Engl J Med, 2021; 385(27): 2520-2530. doi: 10.1056/NEJMoa2111749.
- Gao Y, Chen W, Pan Y, Jing J, Wang C, Johnston SC, et al. INSPIRES Investigators. Dual Antiplatelet Treatment up to 72 Hours after Ischemic Stroke. N Engl J Med, 2023; 389(26): 2413-2424. doi: 10.1056/NEJMoa2309137.
- Chen HS, Cui Y, Wang XH, Ma YT, Han J, Duan YJ, et al. ATAMIS investigators. Clopidogrel Plus Aspirin vs Aspirin Alone in Patients with Acute Mild to Moderate Stroke: The ATAMIS Randomized Clinical Trial. JAMA Neurol, 2024; 81(5): 450-460. doi: 10.1001/jamaneurol.2024.0146.
- Chen HS, Cui Y, Zhou ZH, Zhang H, Wang LX, Wang WZ, et al. ARAMIS Investigators. Dual Antiplatelet Therapy vs Alteplase for Patients with Minor Nondisabling Acute Ischemic Stroke: The ARAMIS Randomized Clinical Trial. JAMA, 2023; 329(24): 2135-2144. doi: 10.1001/jama.2023.7827.
- Chimowitz MI, Lynn MJ, Howlett-Smith H, Stern BJ, Hertzberg VS, Frankel MR, et al. Warfarin-Aspirin Symptomatic Intracranial Disease Trial Investigators. Comparison of warfarin and aspirin for symptomatic intracranial arterial stenosis. N Engl J Med, 2005; 352(13): 1305-1316. doi: 10.1056/NEJMoa043033.
- Chimowitz MI, Lynn MJ, Derdeyn CP, Turan TN, Fiorella D, Lane BF, et al. SAMMPRIS Trial Investigators. Stenting versus aggressive medical therapy for intracranial arterial stenosis. N Engl J Med, 2011; 365(11): 993-1003. doi: 10.1056/NEJMoa1105335.
- Twine CP, Kakkos SK, Aboyans V, Baumgartner I, Behrendt CA, Bellmunt-Montoya S, et al. Document Reviewers; Koncar I, Fitridge R, Matsagkas M, Valgimigli M. Editor's Choice - European Society for Vascular Surgery (ESVS) 2023 Clinical Practice Guidelines on Antithrombotic Therapy for Vascular Diseases. Eur J Vasc Endovasc Surg, 2023; 65(5): 627-689. doi: 10.1016/j.ejvs.2023.03.042.
- Eckstein HH, Kühnl A, Berkefeld J, Dörfler A, Kopp I, Langhoff R, et al. German S3 guideline for diagnosis, treatment, and follow-up care for extracranial carotid stenosis, long version, short version, and guideline report, 2020.
- Zhang Y, Wang X, Gao Y, Chen W, Johnston SC, Amarenco P, et al. Dual Antiplatelet Treatment up to 72 Hours After Ischemic Stroke Stratified by Risk Profile: A Post Hoc Analysis. Stroke, 2025; 56(1): 46-55. doi: 10.1161/STROKEAHA.124.049246.
- Zhou Q, Gao Y, Chen W, Johnston SC, Amarenco P, Bath PM, et al. Efficacy and Safety of Dual Antiplatelet Treatment up to 72 Hours in Acute Ischemic Stroke Stratified by Glycemic Status. Ann Neurol. 2025 Jul;98(1):174-182. doi: 10.1002/ana.27207. Epub 2025 Feb 11. Erratum in: Ann Neurol, 2025; 97(6): 1226. doi: 10.1002/ana.27246.
- Wang Y, Pan Y, Wang X, Gao Y, Chen W, Johnston SC, et al. Relationship Between Neutrophil Count and 90-Day Outcomes and Effect of Dual Antiplatelet Therapy in Patients with Acute Ischemic Stroke or Transient Ischemic Attack: A Post Hoc Analysis of the INSPIRES Trial. J Am Heart Assoc, 2025; 14(14): e040492. doi: 10.1161/JAHA.124.040492.
- Xia Z, Gao Y, Chen W, Johnston SC, Amarenco P, Bath PM, et al. Dual Antiplatelet Therapy in Patients with Metabolic Syndrome After Mild Ischemic Stroke or Transient Ischemic Attack. J Am Heart Assoc, 2025; 14(15): e041449. doi: 10.1161/JAHA.125.041449.
- Liu T, Wang Y, Zhang K, Fan H, Li X, Wu X, et al. Evaluation of intracranial atherosclerotic stenosis burden, the use of dual antiplatelet therapy, and stroke recurrence in patients with minor stroke. J Thromb Haemost, 2025; 23(9): 2913-2925. doi: 10.1016/j.jtha.2025.04.034.
- De Matteis E, De Santis F, Foschi M, Romoli M, Tassinari T, Saia V, et al. READAPT Study Group. Real-World Dual Antiplatelet Therapy Use Exceeds Randomized Trials Boundaries with Possible Safety Issues in Patients with Large Artery Atherosclerosis-Insights From the READAPT Study. Eur J Neurol, 2025; 32(4): e70163. doi: 10.1111/ene.70163.
- Donners SJA, van Velzen TJ, Cheng SF, Gregson J, Hazewinkel AD, Pizzini FB, et al. ECST-2 investigators. Optimised medical therapy alone versus optimised medical therapy plus revascularisation for asymptomatic or low-to-intermediate risk symptomatic carotid stenosis (ECST-2): 2-year interim results of a multicentre randomised trial. Lancet Neurol, 2025; 24(5): 389-399. doi: 10.1016/S1474-4422(25)00107-3.

