Management of Scalp and forehead defects
Atul Philipose1,*, Milind A Mehta2, Rahul Kaushik3 and Mugdha Mehta4
1Assistant Professor, Department of Plastic Surgery, Christian Medical College, Vellore
2Professor, Department of Plastic Surgery, Narendra Modi Medical College, Ahmedabad
3Senior Resident, Department of Plastic Surgery, All India Institute of Medical Sciences, Bathinda
4Intern, B.J Medical College, Ahmedabad
Received Date: 22/09/2025; Published Date: 24/10/2025
*Corresponding author: Atul Philipose, Assistant Professor, Department of Plastic Surgery, Christian Medical College, Vellore
Abstract
Introduction: Scalp defects may result from various causes such as trauma, tumour excision, infections, or burns. Due to the scalp’s distinct anatomical characteristics—including limited elasticity, tight adherence to the calvarium, and a convex contour—reconstructive procedures pose both functional and aesthetic challenges.
Aim: To evaluate and compare scalp reconstruction techniques and assess outcomes in relation to defect size, aetiology, and anatomical site.
Materials and Methods: A prospective clinical study was carried out on 25 patients with scalp defects treated at a tertiary care centre between January 2018 and December 2022. Reconstruction technique used on the patients were primary closure, local advancement and rotation flaps, tissue expansion, or free tissue transfer. Surgical outcome, complication rates, healing time, and aesthetic satisfaction parameters were assessed.
Results: Trauma (45%) and post-tumour excision (35%) identified as the most common aetiologies. In 60% of cases local flaps were satisfactory. Medium to large or complex defects, especially those involving prior irradiation or infection, required tissue expanders (15%) or free flaps (10%). Minor complications reported in 7.5% of cases. Overall, 85% of patients reported good to excellent aesthetic and functional outcomes.
Conclusion: Scalp reconstruction requires a patient-specific, tailored, algorithmic approach. Transposition and local flaps remain the foundation for small to moderate defects, whereas for larger or complicated defects, tissue expansion and free flaps are recommended. A robust understanding of regional anatomy and reconstructive principles is essential to achieve optimal outcome.
Keywords: Scalp defect; Local flap; Tissue expansion; Free flap; Reconstructive surgery
Introduction
The management of scalp and forehead defects requires a comprehensive understanding of its’ complex anatomy and extensive vascular network. The scalp extends from the superior nuchal lines and occipital turbulences to the supraorbital foramen, consists of five distinct layers: skin, subcutaneous tissue, galea aponeurosis, loose areolar tissue, and pericranium [1]. The forehead and scalp have different musculature and hair-bearing characteristics, despite frequently being viewed as one single entity [2]. The region is highly vascularized, with abundant arterial supply and venous drainage including the valveless emissary veins, that increases the risk of intracranial spread of infection [3]. While lymphatic drainage has no nodal barriers, allowing direct flow to cervical nodes [4], innervation involves both cranial and cervical nerves [4]. So, careful consideration of these anatomical features is of utmost importance before planning an effective reconstructive strategy in this region.
Historically, the techniques of scalp reconstruction have evolved significantly over the centuries from ancient Egypt to more recent microsurgical procedures and tissue expansion methods. Few such milestones include Orticochea’s flap methods, Neumann’s pioneering tissue expansion, and successful total scalp replantation by Miller et al. in 1976 [5-7]. Scalp and forehead defects commonly result from trauma, burns, infections, tumours, or congenital anomalies [8-13]. As these regions pose unique anatomical and aesthetic challenges, effective management of scalp and forehead defects necessitates an individualized tailored approach, accounting for the size, depth, location of the defect, it’s aetiology, surrounding tissue quality, and patient comorbidities. Such as, the integrity of the pericranium is essential for graft survival and calvarial protection [14]. For the best functional and aesthetic results, "like with like" replacement is recommended, especially when it comes to hair-bearing scalp tissue [15-21]. However, operative success largely depends on achieving meticulous haemostasis, proper incision orientation, and careful galeal scoring to maximize tissue mobility [22-26].
Tissue expansion is a suitable technique for reconstructing large defects with excellent aesthetic integration, though it requires staged procedures and carries a risk of complications [27-30]. In cases of scalp avulsion, microsurgical replantation remains the preferred approach whenever feasible [31,32]. For smaller defects (<3 cm), primary closure is often feasible, especially in areas having tissue laxity, aided by wide undermining and galeal relaxation incisions if necessary [14]. In select non-cosmetically critical, shallow defects without bone exposure, healing by secondary intention may be acceptable, though the risk of alopecia makes this less ideal for the scalp [33].
Skin grafts, including split- and full-thickness types, offer quick and technically simple coverage for larger defects, provided a well-vascularized bed is present [34-36]. In cases lacking pericranium or vascularized tissue, dermal substitutes such as Integra or Alloderm can provide a scaffold for neodermis formation before grafting [37,38]. Additionally, negative pressure wound therapy (NPWT) can support granulation tissue formation and improve graft take, especially over calvarial surfaces [39-42].
For medium to large defects, a variety of local flaps including rotational, transposition, pinwheel, Orticochea, and Juri flaps etc. can be designed based on vascular networks and relaxed skin tension lines [26,43-48]. In larger or complex cases where local options are inadequate, tissue expansion may be used to generate additional hair-bearing scalp tissue, although it requires staged procedures and carries a moderate complication rate [27-30].
When local or regional flaps are unsuitable due to history of previous radiation, infection, or insufficient vascularity, free tissue transfer becomes necessary. Commonly used free flaps include the latissimus dorsi, serratus anterior, anterolateral thigh, and radial forearm flaps, each chosen based on defect size, location, and required pedicle length [49-51]. In cases where the calvarial bone is lost, reconstruction with autologous bone grafts (parietal or rib) or alloplastic materials (titanium mesh, methyl methacrylate, hydroxyapatite) is performed to protect intracranial contents and restore skull contour [52,53]. Presence of a stable soft tissue coverage remains essential to prevent future complications.
Postoperatively, careful patient positioning, graft protection, and use of drainage or bolster dressings are essential, especially for large flaps or reconstructions in high-tension areas. In microvascular free flap cases, pedicle protection and head elevation are crucial to preserve flap viability and minimize pressure-related complications [54,55].
Overall, successful reconstruction of scalp and forehead defects hinges on a deep understanding of regional anatomy, meticulous planning, selection of the most appropriate reconstructive modality, and individualized patient-centered care. In this study, we aimed to evaluate the epidemiology, etiology, type and distribution, management modalities, postoperative complications, and long-term outcomes associated with surgical treatment of scalp and forehead defects.
Materials and Methods
This prospective observational study was conducted over a period of 30-month from September 2018 to February 2021 in the Department of Plastic Surgery at our tertiary care institution. The patients treated here were either directly admitted to the Plastic Surgery Department or referred from other departments within the hospital or from outside facilities. Approval for the current study was obtained from the Institutional Ethics Committee.
1. Patient Selection and Data Collection
A total of 25 indoor patients with scalp or forehead defects treated surgically during the study period were included in this study. Written informed consent was obtained from all participants prior to enrolment in the study. Data collection was done using a predesigned proforma, which entailed variables such as age, sex, aetiology, anatomical site and size of the defect, reconstructive technique employed, and postoperative complications. A preselected team from plastic surgery department was in charge of Clinical assessment, surgical planning and management.
A.1. Initial Management and Resuscitation
In cases of trauma-related defects, initial management included a primary survey following the standard trauma protocols, with particular attention to airway, breathing, and circulation (ABCs). Once hemodynamic stability was ensured, detailed information regarding the type and cause of injury, time since trauma, primary wound care received, and relevant medical history was recorded. Emergency care included administration of tetanus toxoid, systemic antibiotics, analgesics, and appropriate wound debridement etc.
A.2. Investigations
Baseline investigations included complete hemogram, serum electrolytes level, renal function tests, liver function tests, fasting and postprandial blood glucose levels, urine routine and microscopy test, radiograph of chest and the wounded area etc. Patients over 40 years of age also underwent electrocardiography. Wound swabs were collected for culture and sensitivity testing. High definition Multislice computed tomography (CT) with three-dimensional reconstruction was performed (as and when indicated) to assess the extent of bony involvement and guide subsequent surgical planning.
2. Clinical and Diagnostic Evaluation
A thorough clinical examination was performed, with particular attention to comorbidities such as diabetes mellitus, coronary artery disease, smoking history, prior radiation therapy, immunosuppressive states etc. In suspected oncological cases, staging workup including biopsies, MRI, PET-CT, or bone scans was performed to assess for local invasion or metastatic disease.
B.1. Assessment of the Defect and Surrounding Tissue
Defects were classified based on size, shape, depth, and anatomical location for facilitating an algorithmic approach to reconstruction. Parietal defects offered greater mobility and were more amenable to primary closure, whereas occipital and temporal defects often required flap-based reconstruction due to limited laxity. Defects involving the calvarium underwent debridement of devitalized bone and were assessed for pericranial preservation, which was essential for graft adherence.
Quality of the surrounding tissue including thickness, elasticity, vascularity, and presence of any scarring from previous surgeries, burns, or radiation was carefully evaluated. In oncological cases, confirmation of negative margins was required before proceeding with definitive reconstruction. Hairline involvement was also considered when planning local flaps or tissue expansion.
B.2. Patient Considerations
Patient-specific factors such as overall health, level of function, compliance, and personal preference were key in surgical decision-making. Patients undergoing complex or staged procedures were evaluated for their ability to endure prolonged recovery and follow-up period, and possible cosmetic disfigurement. In oncological patients, the timing of surgery was adjusted accordingly with the chemotherapy or radiotherapy schedule.
Surgical Management and Postoperative Care
Surgical planning was individualized based on the characteristics of the defect and patient related factors. Reconstructive options included primary closure, healing by secondary intention, split- or full-thickness skin grafting, local or regional flaps, tissue expansion, and free tissue transfer. Multistage reconstructions were performed as per requirement.
Postoperative management included elevation of the head to reduce edema, use of bolster dressings for skin grafts, and protection of flap pedicles from pressure. Negative-pressure wound therapy was used in selected cases to reduce skin graft loss. In non-compliant patients, temporary halo devices were employed to prevent inadvertent flap compression.
Patients were monitored regularly in the outpatient department following discharge. The wound status, healing progression, associated complications, and patient satisfaction were recorded at each visit. Readmission, revision surgery, or additional wound care was advised as needed.
Data collected using the standardized proforma were compiled in Excel sheet for final analysis. Descriptive statistics including frequency and percentages were calculated for the quantitative Variables.
Results
A total of 25 patients with scalp and forehead defects were managed surgically between September 2018 and February 2021.
Demographic Profile:
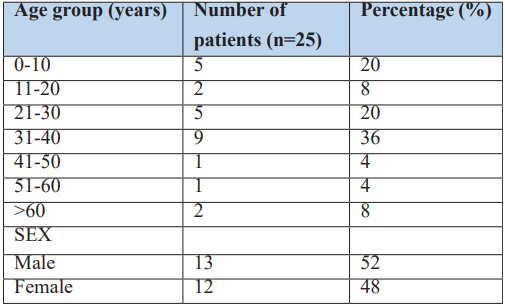
The age of the patients ranged from 5 to 67 years, with a mean age of 30.04 years. About 56% of the patients were in the third and fourth decades where a majority (36%) belonged to the 31- 40-year age group. Of the 25 patients, 13 (52%) were male and 12 (48%) were female, resulting in a near-equal gender distribution (male-to-female ratio of 1.08:1) (Table 1).
Table 1: Distribution of scalp and forehead defects according to patient’s age and gender.
Aetiology of Defects
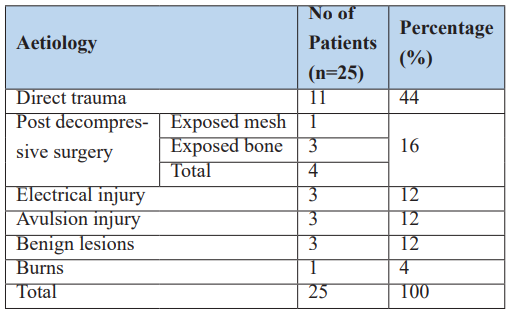
The most common cause of scalp and forehead defects was direct trauma, accounting for 44% (n=11) of cases. Other aetiologies included post-decompressive neurosurgical exposure (16%), electrical injuries (12%), avulsion injuries (12%), benign lesions (12%), and burns (4%) respectively (Table 2).
Table 2: Aetiology of the scalp and forehead defects.
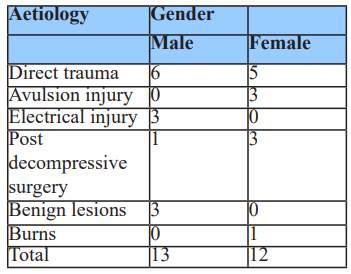
Among these cases, patients aged 11–40 years were most affected, accounting for 68% (n=17) of the total study groups, consistent with the high-risk nature of injuries seen in this working-age population (Table 3). Gender-based aetiology patterns indicated that avulsion injuries were exclusive to females, whereas electrical injuries occurred only in males (Table 4).
Table 3: Age group affected vs. Aetiology of scalp & forehead defects with gender distribution.
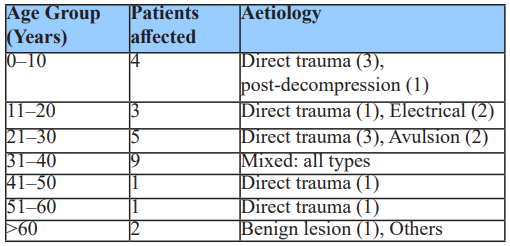
Table 4: Aetiology of scalp & forehead defects with gender distribution.
Site of Defects
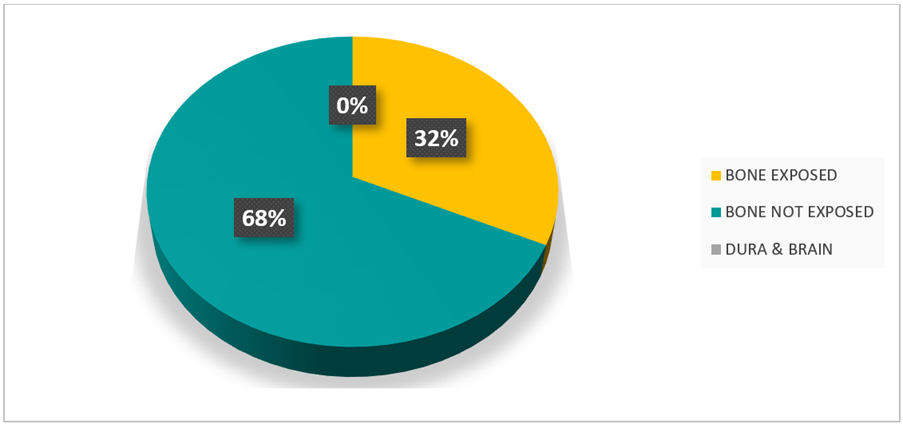
The frontal region was the most commonly affected site (20%), followed by the right and left parietal regions and right parieto-temporal areas (12% each) (Table 5). Associated injuries were present in 24% of patients, with head/neck injuries being the most common (12%), followed by skeletal injuries (8%), and abdominal/chest trauma (4%) (Table 6). Bone was exposed in 32% (n=8) of cases, while no patients presented with exposed dura or brain tissue (Table 7).
Table 5: Distribution of site of scalp and forehead defect.
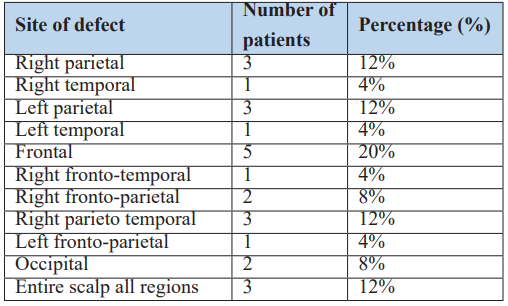
Table 6: Associated injuries with scalp and forehead defects.
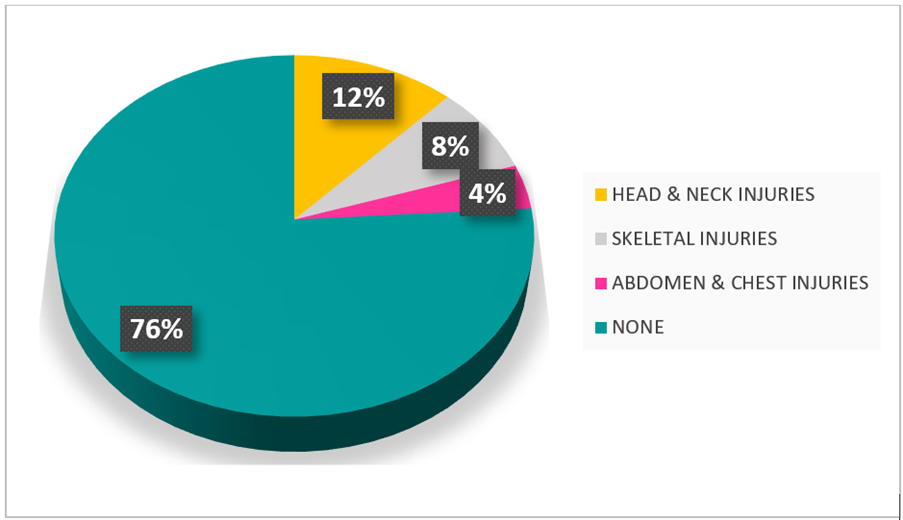
Table 7: Underlying structure exposed in the scalp and forehead wound.
Reconstructive Procedures and Postoperative Complications
Transposition flaps were preferred in moderate to large defects; STG was often employed when pericranium was intact. The choice of procedure was largely determined by the aetiology: direct trauma cases mostly required flaps, while avulsion injuries often needed grafting (Table 8).
Table 8: Distribution of flaps/procedures according to aetiology of defect.
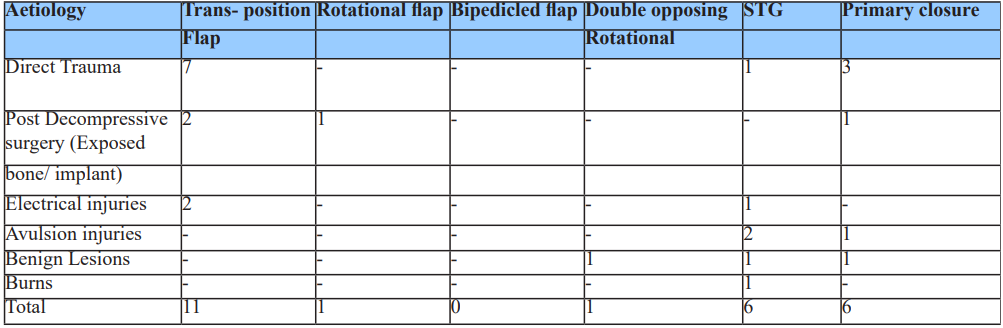
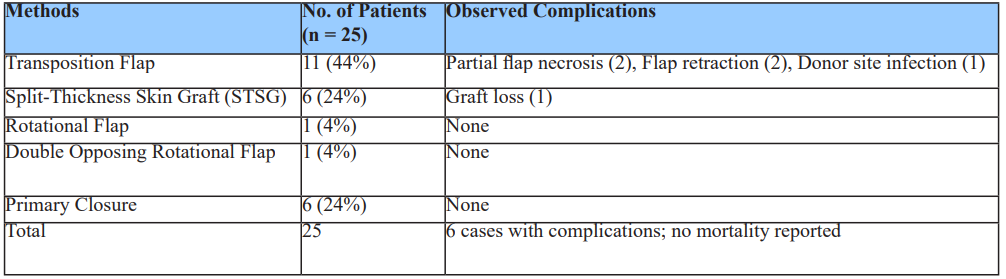
The most frequently used reconstruction method was the transposition flap (44%), followed by split-thickness skin grafting (24%) and primary closure (24%). Complications were observed in 20% of cases involving transposition flaps, including partial necrosis (n=2), flap retraction (n=2), and donor site infection (n=1). One case of partial graft loss was also reported (Table 9).
Table 9: Distribution of procedure performed and incidence of complications post flap/procedure in management of scalp & forehead defects.
Hospital Stay
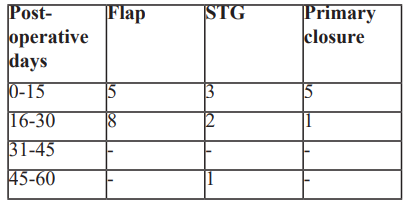
The average hospital stays varied by procedure, with primary closure patients typically discharged within 1–2 weeks, whereas flap or graft patients required longer hospitalization (Table 10).
Table 10: Distribution of cases according to the duration of post-operative hospital stay.
Discussion
Reconstruction of scalp and forehead defects remains a unique surgical challenge due to the complex anatomy of this region, functional significance, and high cosmetic visibility. The aetiology of such defects is diverse, encompassing trauma, oncologic resection, burns, electrical injuries, and congenital or iatrogenic causes. In the present study, trauma was the most frequent cause of scalp and forehead defects (44%), consistent with the findings of Memon et al. and Mateev et al., who also reported trauma as a dominant etiological factor among young, working individuals [55,56]. The higher number of cases in the 11–40-year age group (68%) reflects occupational hazards and greater risk of road traffic accidents in this demographic.
While many studies have reported a strong male predominance in scalp defect cases [55,56], our study observed an almost equal gender distribution, with a male-to-female ratio of 1.08:1. This deviation may be attributable to the increasing involvement of women in agricultural and industrial activities, particularly in rural settings. Avulsion injuries, observed exclusively in female patients in this study, have previously been associated with entanglement of long hair in rotary machinery [57].
Electrical injuries, although representing a smaller portion of the study population (12%), were notably severe. All affected patients were young males, with defects often involving full-thickness scalp loss and exposed calvarium, consistent with the previous study done by Adamo et al., who reported the reconstructive complexity and high morbidity associated with high-voltage electrical injuries of the scalp [58].
The predominance of defects found in the frontal and parietal regions in the current study aligns with the previous reports, suggests vulnerability due to anatomical exposure and thinner soft tissue coverage [59]. Calvarial bone exposure was noted in 32% of cases. In such cases, reliable coverage becomes paramount, as skin grafting alone is insufficient on bare bone without pericranial support. In these cases, outer table burring or vascularised flap coverage is often required to facilitate successful reconstruction [36,37].
Transposition flaps were preferred in moderate (5–25 cm²) to large defects (>25 cm²), offering a reliable option with acceptable aesthetic outcomes. This preference aligns with findings by Mario Iger et al. and Abdul Memon et al [55,60]. Local flaps were favoured due to their reliability, vascular safety, and technical simplicity, particularly in resource-limited settings. These findings are in agreement with prior work done by Orticochea [43] and Zayakova et al [42]. Skin grafting was effective in cases without pericranial compromise, although aesthetic outcomes and durability were suboptimal compared to local flaps. However, a 20% complication rate was observed in flap reconstructions, including partial necrosis, flap retraction, and donor site infection, consistent with reports by Abhyankar et al [46].
Split-thickness skin grafts were used primarily when the pericranium was intact. Though technically straightforward, they are associated with cosmetic limitations such as alopecia and poor contour match [35]. Additionally, graft take is unpredictable in irradiated or poorly vascularised beds [39,49], and long-term stability may be compromised.
Postoperative complication rates in this study were comparable to those reported in the literature [60-62], with 18.1% necrosis rate of transposition flaps, slightly higher than some previous studies, but manageable with conservative treatment or secondary procedures. Hospital stays varied according to the procedure type, with primary closure associated with the shortest duration and flap or graft cases requiring longer admissions. Institutional policies, including extended inpatient care under public healthcare schemes, likely contributed to the observed lengths of stay - an issue also reported by Bhaskaranand and Parikh [63].
This study highlights the importance of individualised reconstruction planning, defect characteristics, patient comorbidities, and regional resources. However, several limitations must be acknowledged. The relatively smaller sample size limited the generalisability of the findings. The study was conducted at a single tertiary care centre, potentially introducing referral bias and limiting the representation of cases from primary and secondary healthcare settings. Additionally, the absence of a control group limits the comparative evaluation of the efficacy of the reconstructive techniques.
Conclusion
Scalp and forehead defects present diverse reconstructive challenges. Local transpositional flaps remain the cornerstone of management in most cases, offering reliable coverage with acceptable complication rates. While outcomes were largely favourable, proper patient selection, refinement of techniques and careful postoperative monitoring remain essential for optimal results. Multidisciplinary collaboration and advancements in microsurgical techniques may further augment reconstructive outcomes in complex cases. Future multicentric studies with larger sample sizes and objective outcome measures including patient satisfaction and aesthetic scoring are recommended for further validation and refinement of reconstructive protocols for the complex scalp and forehead defects.
References
- Sinnatamby CS. Last’s Anatomy: Regional and Applied. 11th ed. Edinburgh: Churchill Livingstone, 2006.
- Standring S. Gray’s Anatomy: The Anatomical Basis of Clinical Practice. 40th ed. Edinburgh: Churchill Livingstone, 2008.
- Snell RS. Clinical Anatomy by Regions. 8th ed. Philadelphia: Lippincott Williams & Wilkins, 2007.
- Moore KL, Dalley AF. Clinically Oriented Anatomy. 6th ed. Philadelphia: Lippincott Williams & Wilkins, 2010.
- Orticochea M. A new rotation flap for the repair of large defects of the scalp and forehead. Br J Plast Surg, 1971; 24(2): 184–197.
- Neumann CG. The expansion of an area of skin by progressive distension of a subcutaneous balloon. Plast Reconstr Surg, 1957; 19(2): 124–130.
- Miller SH, Hartrampf CR, Gottlieb JR. Successful replantation of avulsed scalp. Plast Reconstr Surg, 1976; 57(3): 318–321.
- Abhyankar SV, Kulkarni A, Agarwal NK. Scalp reconstruction: An overview. Indian J Plast Surg, 2014; 47(2): 144–151.
- Memon AA, Khan S, Soomro YH. Reconstruction of scalp defects: An institutional experience. Pak J Surg, 2014; 30(3): 252–256.
- Mateev MA, Radev L, Iliev D. Scalp defect reconstruction: Our experience. J Craniofac Surg, 2017; 28(5): 1287–1291.
- Harashina T. Analysis of 200 cases of scalp reconstruction. Plast Reconstr Surg, 1980; 66(4): 542–546.
- Geddes CR, Tang M, Yang D, Morris SF. Anatomy of the scalp and its clinical applications. Br J Plast Surg, 2003; 56(7): 651–658.
- Mahipathy SR, Krishnan CU. Flap reconstruction for large scalp defects. Indian J Plast Surg, 2016; 49(3): 283–289.
- Glat PM, Longaker MT. Scalp reconstruction by tissue expansion. Clin Plast Surg, 2005; 32(3): 321–336.
- Juri J. Advancement and rotation flap for reconstruction of the scalp. Plast Reconstr Surg, 1975; 55(4): 456–460.
- Orticochea M. The design of local flaps for the repair of scalp defects. Br J Plast Surg, 1971; 24(2): 184–197.
- Georgescu AV, Capota IM, Ardeleanu V. Scalp reconstruction using local flaps. Ann Plast Surg, 2004; 53(4): 390–397.
- Thorne CH. Local flaps in head and neck reconstruction. In: Thorne CH, Chung KC, Gosain AK, Mehrara BJ, Rubin JP, Spear SL, editors. Grabb and Smith’s Plastic Surgery. 6th ed. Philadelphia: Lippincott Williams & Wilkins, 2007; p. 442–456.
- Hallock GG. Replantation of total scalp avulsion. J Reconstr Microsurg, 1992; 8(1): 29–32.
- Lin TS. Microsurgical replantation of avulsed scalp. Plast Reconstr Surg, 2004; 114(1): 101–104.
- Nout E, van der Wal MB, Vrouenraets BC, Hofer SO, Niessen FB. Galea scoring to increase mobility in scalp flaps. Plast Reconstr Surg, 2008; 122(2): 548–555.
- Sasaki GH. Tissue expansion of the scalp. Clin Plast Surg, 1987; 14(2): 367–385.
- Kroll SS, Schusterman MA. Complications of tissue expansion in head and neck reconstruction. Plast Reconstr Surg, 1990; 86(5): 892–897.
- Tanaka A, Tajima S. Complications of tissue expansion. Scand J Plast Reconstr Surg, 1991; 25(2): 191–195.
- Rogers NE, Marcus JR, Bentz ML. Outcomes of scalp reconstruction using tissue expanders. J Plast Reconstr Aesthet Surg, 2009; 62(10): 1279–1283.
- Vinh VQ, Ogawa R. Microsurgical replantation of the scalp: A review. Microsurgery, 2010; 30(5): 396–401.
- Agarwal CA, Liu PS, Gottlieb LJ, Anthony JP. Split-thickness skin grafts for scalp reconstruction: Long-term outcomes. J Craniofac Surg, 2008; 19(6): 1464–1467.
- Tolhurst DE, Carstens MH, Greco RJ, Furnas DW. Calvarial bone exposed in trauma and reconstructive surgery. Plast Reconstr Surg, 1991; 88(2): 295–302.
- Kroll SS, Reece GP, Robb GL. Split-thickness skin grafting to the calvarium without periosteum: 20-case review. Plast Reconstr Surg, 1998; 102(5): 1405–1409.
- Papp A, Lahtinen T, Uusaro A, Vuola J. Artificial dermis in scalp reconstruction. Burns, 2007; 33(2): 203–211.
- Wainwright DJ. Use of an acellular allograft dermal matrix in full-thickness burns. Burns, 1995; 21(4): 243–248.
- Orgill DP, Bayer LR. Update on negative-pressure wound therapy. Int Wound J, 2011; 8(1): 18–23.
- Banwell PE, Téot L. Topical negative pressure: The evolution of a novel therapy. J Wound Care, 2003; 12(1): 22–28.
- Morykwas MJ, Argenta LC, Shelton-Brown EI, McGuirt W. Vacuum-assisted closure: Basic animal studies. Ann Plast Surg, 1997; 38(6): 553–562.
- Kim PJ, Attinger CE, Oliver N, Garwood C, Evans KK, Steinberg JS, et al. Outcomes with NPWT in head and neck. Wound Repair Regen, 2014; 22(4): 488–498.
- Whitaker IS, Karoo RO, Spyrou GE, Fatah MF. Free flaps in scalp and forehead reconstruction: 15-year review. J Plast Reconstr Aesthet Surg, 2007; 60(6): 682–689.
- McCombe D, Brown T, Mah E. The “pinwheel” flap in scalp reconstruction. Plast Reconstr Surg, 2001; 107(6): 1355–1360.
- Mahadevan M, Suraj K, Ulaganathan G. Local flaps for scalp and forehead defects. Indian J Plast Surg, 2004; 37(2): 121–126.
- Abhyankar SV, Kulkarni A, Agarwal NK. Complications of scalp flap surgery. Indian J Plast Surg, 2013; 46(2): 425–432.
- Goertz O, Vogt P, Kolbenschlag J, Daigeler A, Harati K, Germann G, et al. Large scalp defects: Local and free flap closure. Scand J Plast Reconstr Surg Hand Surg, 2010; 44(4): 197–203.
- Wong CH, Wei FC, Lin CH, Chen HC, Chen IH. Free flap reconstruction in the scalp and calvarium: 10-year study. Head Neck, 2010; 32(12): 1602–1610.
- Zayakova Y, Uhrich S, Tzvetkov T, Atanasov B. Reconstruction of forehead and scalp defects with local flaps. J Craniofac Surg, 2009; 20(6): 2105–2110.
- Orticochea M. New rotation flaps for scalp and forehead. Plast Reconstr Surg, 1971; 47(2): 134–139.
- Beasley NJP, Gilbert RW. Microvascular free flap reconstruction of scalp. Head Neck, 2002; 24(5): 423–431.
- Di Benedetto G, Aquinati A, Pierangeli M, Scalise A, Bertani A. Free flaps for skull and scalp defects. Microsurgery, 2005; 25(1): 30–36.
- Erdmann D, Follmar KE, Debruijn M, Levin LS, Pap SA. Complex cranial defect reconstruction. Ann Plast Surg, 2000; 44(4): 385–391.
- Bauer BS, Few JW, Chavez CD, Galiano RD. Tissue expansion in pigmented nevi of pediatric forehead. Plast Reconstr Surg, 2001; 107(3): 668–675.
- Manson PN, Crawley WA, Yaremchuk MJ. Hydroxyapatite cranioplasty: 37-case review. Plast Reconstr Surg, 1999; 104(4): 983–991.
- Krishnan KG, Schackert G, Barth H. Free flap scalp reconstruction complications. Neurosurg Rev, 2002; 25(3): 141–144.
- Iger M, Swartz WM, Banis JC, Acland R. Vascularized flap repair of scalp defects. Plast Reconstr Surg, 1990; 86(6): 1108–1112.
- Adamo MA, Magnuson B, Heuer GG, Storm PB. Electrical injuries of the scalp: Pediatric reconstruction. Burns, 2008; 34(2): 223–228.
- Bhaskaranand K, Parikh SN. Prolonged inpatient stays in scalp reconstruction. Indian J Plast Surg, 2000; 33(1): 16–19.
- Yaremchuk MJ, Randolph MA, Weiland AJ. Long-term results in calvarial reconstruction. Plast Reconstr Surg, 1997; 99(5): 1295–1303.
- Erdmann D, Follmar KE, Levin LS, Friedman AH, Levin LS. Scalp defect closure using microsurgery. Clin Plast Surg, 2005; 32(3): 367–385.
- Antony AK, Chen WF, Kolokythas A, Weimer KA, Cohen MN. Versatility of radial forearm free flap in forehead reconstruction. Ann Plast Surg, 2011; 66(3): 260–265.
- Mücke T, Mitchell DA, Wolff KD. Microsurgical reconstruction of scalp defects. J Oral Maxillofac Surg, 2009; 67(8): 1646–1652.
- Cigna E, Barone-Adesi L, Lazzeri D, Malzone G, Tarallo M, Scuderi N. Anterolateral thigh flap for complex scalp reconstruction. J Craniofac Surg, 2012; 23(2): 511–515.
- Chen HC, Tang YB, Mardini S, Tsai BW, Tsai YT. Free flaps for reconstruction of scalp and skull defects. J Reconstr Microsurg, 1997; 13(8): 595–602.
- Georgescu AV, Capota IM, Ardeleanu V. Frontoparietal scalp wound repair with rotation flaps. J Plast Reconstr Aesthet Surg, 2006; 59(7): 761–769.
- Iger M, Swartz WM, Banis JC. Scalp repair with free tissue transfer. Plast Reconstr Surg, 1990; 86(6): 1108–1112.
- Yaremchuk MJ, Randolph MA, Weiland AJ. Complications in scalp reconstruction. Plast Reconstr Surg, 1997; 99(5): 1295–1303.
- Erdmann D, Follmar KE, Levin LS. Management of large scalp defects. Clin Plast Surg, 2005; 32(3): 367–385.
- Bhaskaranand K, Parikh SN. Public health implications of extended hospital stay in reconstruction. Indian J Plast Surg, 2001; 34(2): 95–100.

