Extravasation Injuries in Pediatric Patients: A Retrospective Study and Management Strategies
Chaimae Ben Driss1,2,*, Hamza Bensaghir1,2 and Nawfal Fejjal1,2
1Pediatric Plastic Surgery Unity, Children hospital of Rabat, Morocco
2Faculty of Medicine and Pharmacy, Mohammed V University, Rabat, Morocco
Received Date: 20/08/2025; Published Date: 12/09/2025
*Corresponding author: Chaimae Ben Driss, Pediatric Plastic Surgery Unity, Children hospital of Rabat, Morocco
Abstract
Introduction: Extravasation injuries are a serious complication in pediatric patients, particularly in intensive care. Delayed management can lead to severe tissue damage and long-term sequelae.
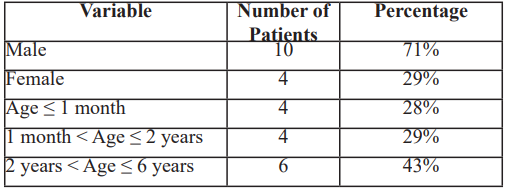
Methods: A 13-year retrospective review (2011–2024) identified 14 children (<15 years) requiring surgical intervention for extravasation injuries. The cohort included 10 males (71%) and 4 females (29%), with a mean age of 2.4 years. Data included demographics, causative agents, lesion severity, and outcomes.
Results: The most common site was the dorsal hand (10 patients, 72%). Hypertonic glucose caused 10 cases (72%), and 10 patients (71%) had full-thickness necrosis (Type IV). Surgical management included debridement (5 patients, 36%), skin grafts (5 patients, 36%), and amputations (2 patients, 14%). Early intervention (<48 hours) improved outcomes.
Conclusion: Extravasation injuries require prompt recognition and protocoled surgical care to minimize morbidity. Prevention strategies and multidisciplinary approaches are essential in high-risk settings.
Keywords: Pediatric extravasation; Vesicant injury; Hypertonic glucose; Surgical management; Prevention; Vascular access complications
Introduction
Extravasation is a frequent iatrogenic complication among neonates and children requiring intravenous therapy in intensive care and oncology settings [1]. It must be considered a diagnostic and therapeutic emergency because of its potential to cause significant tissue damage, sometimes surpassing the severity of the underlying illness that warranted hospitalization [2].
Two distinct terms are often used in the literature: infiltration and extravasation. Infiltration refers to the leakage of non-irritant solutions into surrounding tissues and is generally benign. In contrast, extravasation involves irritant or vesicant solutions leaking into subcutaneous tissues, leading to potentially severe complications such as skin necrosis, functional loss, and long-term aesthetic or psychological sequelae [3,4].
Extravasation complications are severe in approximately 25% of cases, resulting in longer hospitalizations, increased healthcare costs, higher morbidity, and sometimes mortality [5]. Nevertheless, there remains a significant lack of research and standardized management protocols for extravasation injuries, especially in neonates—a particularly vulnerable population [6].
Despite advances in intravenous therapy techniques, pediatric patients remain at elevated risk due to small-caliber veins, fragile skin, and prolonged infusion of irritant or vesicant drugs such as hypertonic glucose, chemotherapeutic agents, and certain antibiotics. Incidence rates vary widely in literature, with some reports suggesting figures between 0.1% and 6% of hospitalized children. These injuries often require complex surgical interventions and can lead to permanent disability if not treated promptly. Developing standardized preventive protocols and timely treatment strategies remains a global priority to mitigate functional impairment, aesthetic consequences, and psychological distress in affected children.
This study aims to raise awareness among healthcare teams regarding the impact of extravasation accidents and proposes structured management strategies to mitigate functional and aesthetic consequences. Additionally, it highlights the importance of prevention and standardized treatment protocols according to the nature of the infused solutions.
Despite advances in intravenous therapy techniques, pediatric patients remain at elevated risk due to small-caliber veins, fragile skin, and prolonged infusion of irritant or vesicant drugs such as hypertonic glucose, chemotherapeutic agents, and certain antibiotics [1,2]. Incidence rates vary widely in the literature, with some reports suggesting figures between 0.1% and 6% of hospitalized children [3,4]. These injuries often require complex surgical interventions and can lead to permanent disability if not treated promptly [5,6]. Developing standardized preventive protocols and timely treatment strategies remains a global priority to mitigate functional impairment, aesthetic consequences, and psychological distress in affected children [7].
Materials and Methods
Study Design and Setting
A retrospective, descriptive study was carried out in the Pediatric Plastic Surgery Unit of a tertiary care hospital, covering a 13-year period from June 2011 to December 2024. The study aimed to review all surgically managed extravasation injuries in patients under the age of 15 years.
Inclusion criteria included pediatric patients aged under 15 years who developed extravasation injuries requiring surgical management. Exclusion criteria were patients with extravasation injuries treated conservatively without surgery and those with incomplete or missing medical records.
Patient data were collected from hospital records and anonymized. The variables recorded included age, sex, department of origin, extravasation site, infusion product involved, lesion type, management strategy, and clinical outcomes. Clinical data were extracted using a standardized data collection form. Variables recorded included demographic characteristics (age, sex), department of origin, extravasation site, the nature of the infused solution, lesion severity, time elapsed between the event and initiation of treatment, type of surgical intervention performed, and clinical outcomes (including sequelae and complications).
Data collection was standardized using a predefined data sheet, and subsequent statistical analysis was performed using Microsoft Excel. Data were entered into Microsoft Excel for descriptive statistical analysis. Categorical variables (such as sex, site of extravasation, type of infused product, and severity grade) were expressed as frequencies and percentages. Continuous variables (such as age) were summarized as means and ranges.
Study Definitions
Lesion severity was classified according to the Williams Classification [7], which categorizes injuries into four grades: Grade I correspond to pain without erythema or swelling; Grade II indicates minor swelling without pallor; Grade III is characterized by significant swelling, pallor, and diminished warmth; and Grade IV represents extensive swelling with absent distal pulse and skin necrosis. Additionally, extravasation sites were grouped into three anatomical regions: the scalp, the dorsal hand, and the dorsal foot.
Management Protocol
All patients were evaluated by the surgical team upon diagnosis of severe extravasation injury. The decision for surgical intervention was based on clinical signs of tissue necrosis, ischemia, or functional impairment. Surgical options included debridement, full- or split-thickness skin grafting, and amputation in extreme cases. Postoperative care consisted of wound monitoring, physiotherapy for functional recovery, and scar management.
Results
Demographic and Clinical Characteristics
A total of 14 pediatric patients were included in the study. There was a notable male predominance, with 10 males (71%) and 4 females (29%), resulting in a sex ratio of 2.5 in favor of boys. The mean age of the cohort was 2.4 years, with extremes ranging from 1 month to 6 years. Distribution by age group showed that 4 patients (28%) were neonates (≤1 month), 4 patients (29%) were infants aged between 1 month and 2 years, and 6 patients (43%) were between 2 and 6 years (Table 1).
Table 1: Patient Demographics and Extravasation Characteristics.
Origin of Patients
Patients originated from various hospital departments, with the highest proportion coming from the Pediatric Intensive Care Unit (5 patients, 36%). Neonatology contributed 3 patients (21%), while the Hematology-Oncology Center also referred 3 cases (21%). Additionally, 2 patients (14%) were referred from external centers. These findings reflect the high prevalence of extravasation incidents in intensive care and oncology settings, where intravenous therapy is frequent and prolonged.
Site of Extravasation
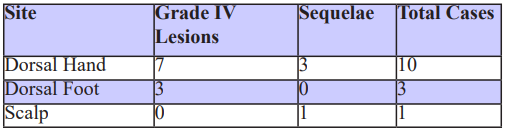
The dorsal hand was the most common site of extravasation, involved in 72% of cases (10 patients) .The dorsal surface of the foot was implicated in 21% of cases (3 patients) (Figure 1), while the scalp was involved in only one case (7%). Analysis of lesion severity according to location showed that dorsal hand extravasations accounted for most Grade IV tissue injuries (Table 2).
Table 2: Site and Severity of Lesions.
Severity of Lesions
According to the Williams Classification, grade IV necrosis occurred in 10 patients (71%) (Figure 2). Four patients (29%) presented with irreversible sequelae such as retraction bands or scarring alopecia (Figure 3).
Among patients with Grade IV lesions, immediate surgical management was essential. In contrast, patients presenting with sequelae required secondary reconstructive interventions.
Infused Products Involved
Hypertonic glucose solutions were the predominant agents causing extravasation (72%, 10 patients). Chemotherapy agents were responsible for 21% of cases (3 patients), and antibiotic solutions accounted for 7% (1 patient). The toxic potential of hyperosmolar and vesicant agents underscores the need for careful handling and infusion monitoring.
Time to Management
Only one patient (7%) received intervention within 48 hours of the extravasation incident. In contrast, 13 patients (93%) were managed after a delay exceeding 48 hours. Early management within 48 hours resulted in better clinical outcomes, with faster healing and fewer sequelae. Delayed diagnosis and intervention were associated with a higher incidence of tissue necrosis and long-term complications.
Management Strategies and Outcomes
Various surgical strategies were employed. Debridement with directed healing was performed in 5 patients (36%), while skin grafting was required in 5 patients (36%), divided between full-thickness grafts in 3 patients (22%) and split-thickness grafts in 2 patients (14%). Amputation was necessary in 2 patients (14%) due to extensive necrosis and compromised perfusion. Additional interventions included scar revision in 1 patient (7%) and fasciotomy with lavage in 1 patient (7%) (Figure 4). Despite these measures, 80% of patients experienced satisfactory healing without major sequelae, while 10% developed significant contracture requiring surgical revision and 10% showed unfavorable progression necessitating further intervention. These results reinforce the critical role of early, appropriate surgical management in optimizing patient outcomes (Table 3).
Table 3: Management Strategies.

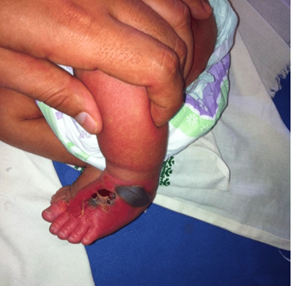
Figure 1: Infant in our department presenting with necrotic lesions following hypertonic glucose solution extravasation on the dorsum of the foot.
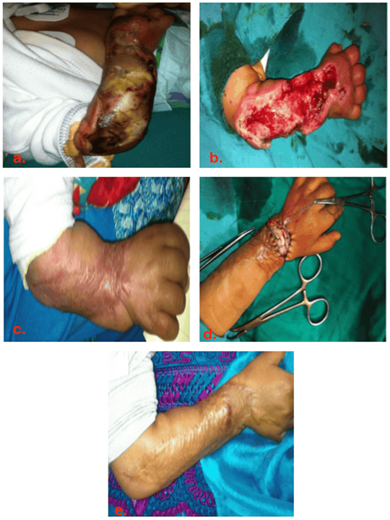
Figure 2: Management of a newborn presenting a type IV extravasation injury due to hypertonic glucose solution intravenous leakage.
- Type IV extravasation injury in the forearm
- Fasciotomy
- The scar after directed healing
- Debridment and full thickness skin graft
- Result after healing
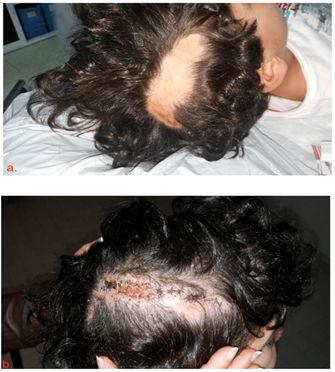
Figure 3: Child presenting cicatricial alopecia of the scalp following extravasation injury treated by reduction of the alopecia plaque.
- The alopecia spot before surgical management
- Result after the reduction of the alopecia spot
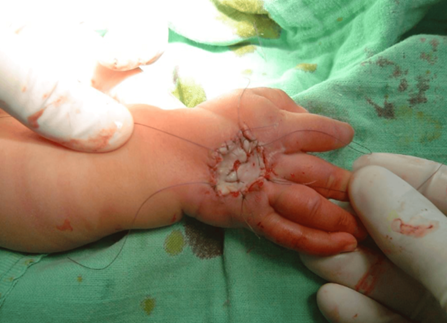
Figure 4: Child from our study who underwent full-thickness skin grafting following the release of a contractile band.
Discussion
Extravasation injuries represent a significant concern in pediatric patients receiving intravenous therapy. Our study, spanning 13 years, provides valuable insights into the epidemiology, management, and outcomes of severe extravasation injuries in a tertiary pediatric hospital. The results highlight crucial areas requiring improvement, including early detection, standardized management protocols, and prevention strategies.
In our cohort, a notable male predominance was observed (71%), consistent with several prior studies [1,2]. However, other investigations have reported a more balanced gender distribution [3]. No definitive biological rationale explains a gender-based difference in extravasation risk, and discrepancies across studies may reflect sampling variability rather than true epidemiologic trends.
The most affected age group in our study was children aged between 2 to 6 years (43%), differing from previous reports that identified neonates as the most vulnerable population [4,5]. Neonates have inherently fragile veins, low tissue resilience, and limited ability to communicate discomfort, placing them at heightened risk [6]. Our different age distribution could be attributed to the specific patient flow and selection bias in surgical referrals at our center, where more severe lesions necessitating surgery were prevalent among older infants and toddlers.
Anatomically, the dorsal hand emerged as the most frequent extravasation site (72%), a finding that mirrors observations in large pediatric cohorts [7,8]. The vulnerability of the dorsal hand relates to the thinness of subcutaneous tissues, frequent movement, and superficial vein positioning, all contributing to catheter instability [9]. The dorsal foot and scalp were also implicated, although to a lesser extent. Understanding the anatomical predilection for extravasation is vital in guiding the choice of vascular access sites and securement techniques.
The type of infused product significantly influenced the severity of extravasation injuries. In our series, hypertonic glucose solutions were responsible for 72% of cases, followed by chemotherapeutic agents (21%) and antibiotics (7%). Similar findings have been reported globally, with hyperosmolar fluids frequently implicated in severe injuries [10,11]. Hypertonic glucose's high osmolarity leads to cellular dehydration, protein denaturation, and subsequent tissue necrosis [12]. Chemotherapeutic agents, particularly anthracyclines, are known vesicants, and their extravasation can result in devastating soft tissue loss if not managed promptly [13].
Timely intervention is paramount in mitigating extravasation-related morbidity. Our study found that only one patient (7%) received intervention within 48 hours, whereas the majority experienced significant delays. Early detection and management are associated with improved outcomes, as demonstrated by several pediatric studies [14,15]. Delay in recognition leads to progression from infiltration to tissue necrosis, ultimately necessitating more aggressive surgical interventions, including debridement, grafting, and, in extreme cases, amputation.
Surgical intervention played a major role in our management strategy, particularly for Grade IV injuries. Directed healing after debridement was performed in 36% of cases, while skin grafting was required in 36% (full-thickness or split-thickness). Amputation was necessary in two patients (14%) who presented with extensive necrosis and vascular compromise. Our findings align with existing reports emphasizing the critical role of surgical management in advanced extravasation injuries [16,17].
The absence of antidotal therapies in our series represents a notable gap. Agents such as hyaluronidase, dexrazoxane, and dimethyl sulfoxide (DMSO) have been proven effective in limiting tissue damage in specific types of extravasations [18,19]. Hyaluronidase facilitates the dispersion of extravasated solutions and is particularly useful in cases involving non-cytotoxic hyperosmolar agents [20]. Dexrazoxane, an FDA-approved agent, is highly effective against anthracycline-induced tissue injury if administered promptly [21]. Future improvements in our center’s extravasation management could benefit from the incorporation of such pharmacological interventions.
Preventive strategies must be emphasized to reduce the incidence of extravasation. Recommended measures include careful selection of vascular access sites, using securement devices, limiting osmolarity and irritant concentrations in peripheral infusions, and frequent site monitoring [10,22]. Educational programs for nursing and medical staff are critical to improve vigilance and early recognition of signs of infiltration.
Overall, our study reaffirms the serious nature of extravasation injuries in pediatric populations and underscores the importance of early recognition, multidisciplinary management, and robust preventive strategies. Strengthening healthcare provider training, establishing standardized protocols, and ensuring access to antidotal therapies are pivotal steps toward reducing extravasation-related morbidity.
Despite these valuable findings, our study has limitations.It is limited by its retrospective design, which introduces potential selection and information biases. The small sample size and single-center setting limit generalizability. Moreover, only surgically managed cases were included, excluding milder injuries, which may distort the perceived severity profile. Future prospective, multicenter studies with larger cohorts are needed to better define risk factors and develop standardized prevention and management protocols.
Conclusion
Extravasation is a serious and preventable complication in pediatric intravenous therapy. Early recognition and timely, standardized management are critical in minimizing tissue damage and functional impairment. Surgical intervention remains essential for severe cases. Strengthening prevention protocols, staff training, and access to antidotal therapies will further enhance patient outcomes.
Contributions: All the authors contributed equally to the conception, acquisition, analysis, interpretation of data, and drafted and approved the manuscript.
Declaration of Conflicting Interests: No conflicts of interest.
Funding: None
Ethical Approval: No ethical approval is required for de-identified case reports and case series based on our institutional policies.
Consent: Written informed consent was obtained from the patient’s legally authorized representatives.
References
- Ghanem AM, Mansour A, Exton R, et al. Extravasation injuries in children: a concise review. Br J Plast Surg, 2006; 59(2): 109-120.
- Fonzo Christe C, Labarère J, Coudane H, et al. Pediatric extravasation injuries: a retrospective study. Pediatr Surg Int, 2010; 26(5): 505-510.
- Ching CB, Wong WY, Chung JO, et al. Management of extravasation injuries in the pediatric population. Pediatr Emerg Care, 2019; 35(2): 142-146.
- Paquette V, McGloin R, Northway T, et al. Intravenous extravasation injury in neonates: a systematic review. Adv Neonatal Care, 2011; 11(3): 123-129.
- Atay S, Sen S, Yildiz D, et al. Extravasation injuries in the neonatal intensive care unit: prevention and management. Neonatology, 2015; 107(3): 233-238.
- Hanrahan K, Lofgren K. Evidence-based pediatric IV extravasation protocol. J Pediatr Nurs, 2004; 19(3): 151-158.
- Wong J, Weber J. Extravasation injuries in children: etiology, prevention and management. Pediatr Emerg Care, 2011; 27(1): 52-58.
- Parron M, Jourdain M, et al. Pediatric extravasation injuries: anatomical considerations and management. J Plast Reconstr Aesthet Surg, 2012; 65(8): 1127-1133.
- Barton J, Wallace D, Woolson S, et al. Pathophysiology of hyperosmolar extravasation injuries. Ann Plast Surg, 2009; 63(4): 455-461.
- Schulmeister L. Vesicant extravasations: management and nursing care. Clin J Oncol Nurs, 2011; 15(4): 413-419.
- Gault DT. Extravasation injuries. Br J Plast Surg, 1993; 46(2): 91-96.
- Reynolds PM, MacLaren R, Bachand R, et al. Extravasation management: a review of the evidence. J Pharm Pract, 2014; 27(5): 476-482.
- Harrold K, Goulden N, Taylor M, et al. Early intervention for chemotherapy extravasation injuries. Cancer Nurs, 2007; 30(5): 348-354.
- Weinberg L, Peake B, Tan C, Nikfarjam M. Management of extravasation injuries: a review. J Clin Anesth, 2010; 22(8): 623-628.
- Boyar V. Managing IV extravasations. Nurs Crit Care, 2015; 10(5): 18-23.
- Langer SW. Dexrazoxane for the treatment of anthracycline extravasation. Cancer Chemother Pharmacol, 2014; 74(5): 867-871.
- Schmid H, Leu AJ, Stutz G, et al. Hyaluronidase treatment for extravasations: a retrospective analysis. Oncol Res Treat, 2018; 41(9): 526-532.
- Hadaway L. Infiltration and extravasation. Am J Nurs, 2007; 107(8): 64-72.
- Taddio A, Shah V, Katz J, et al. Prevention and management of extravasation injuries in neonates. Pediatr Clin North Am, 2013; 60(5): 1163-1174.
- Sawatzky-Dickson D, Bodnaryk K, Kirk V. Topical management of neonatal extravasation injuries. Adv Neonatal Care, 2006; 6(2): 82-95.
- Weiss RB, Issell BF. The anthracyclines: will we ever find a better doxorubicin? Semin Oncol, 1982; 9(3): 158-178.
- Schulmeister L. Management of vesicant extravasations. Semin Oncol Nurs, 2011; 27(2): 82-90.
- Infusion Nurses Society. Infusion therapy standards of practice. J Infus Nurs, 2016; 39(1S): S1-S159.

