An Unusual Threat: Cytomegalovirus-Triggered Hemophagocytic Lymphohistiocytosis in An Immunocompetent Adult
Marta García Gamero1, Marta Abengózar Mancera2,*, Juana Carretero Gómez3 and Araceli Vera Tomé4
1Medical Oncology Resident, Department of Medical Oncology, Badajoz University Hospital, Badajoz, Spain
2Intensive Care Medicine Resident, Department of Intensive Care Medicine, Badajoz University Hospital, Badajoz, Spain
3Department of Internal Medicine, University Institute for Biomedical Research of Extremadura (INUBE), Clinical Research Center of the Badajoz (CICAB), Badajoz University Hospital, Badajoz, Spain
4Department of Internal Medicine, Badajoz University Hospital, Badajoz, Spain
Received Date: 16/12/2025; Published Date: 23/02/2026
*Corresponding author: Marta Abengózar Mancera, Department of Internal Medicine, University Hospital Complex Badajoz, Badajoz, Avenida de Elvas, s/n, Badajoz 06080, Spain
Abstract
Hemophagocytic Lymphohistiocytosis (HLH) is a rare hyperinflammatory syndrome that can mimic more common infectious or inflammatory disorders, leading to delayed recognition. We report the case of a previously healthy woman in her 60s who presented fatigue, cytopenias, splenomegaly, hypertriglyceridemia and hyperferritinemia. Despite not fulfilling all HLH-2024 diagnostic criteria, clinical suspicion was raised and further investigations revealed active Cytomegalovirus (CMV) infection as the triggering factor. Targeted antiviral therapy led to progressive improvement without the need for cytotoxic HLH-specific treatment. This case illustrates that HLH may occur in immunocompetent adults without classical risk factors and that identification of a viral trigger can guide management and improve outcomes. It also highlights the limitations of current diagnostic criteria, the potential role of the H-Score in atypical or early presentations and the importance of multidisciplinary collaboration and structured follow-up to detect relapse and prevent long-term complications.
Keywords: Hemophagocytic lymphohistiocytosis; Cytomegalovirus; Hyperferritinemia; Mesenteric panniculitis; Hemophagocytosis
Introduction
Hemophagocytic Lymphohistiocytosis (HLH) is a rare but life-threatening hyperinflammatory syndrome, usually triggered by infections, malignancies, autoimmune diseases, immunodeficiencies or genetic defects that generate familial forms. Its nonspecific presentation and overlap with other inflammatory or infectious conditions delays its diagnosis and treatment. We report a case of HLH in a previously healthy adult woman without underlying chronic disease or immunodeficiency, highlighting that HLH can occur without classical risk factors and the importance of early recognition [1].
Case Report
A woman in her 60s, previously healthy and functionally independent, showed up at the emergency department with a one-week history of profound fatigue, generalized muscle and joint pain, dizziness and blurred vision. She denied fever or other systemic symptoms and reported a self-limiting episode of sore throat, cervical swelling and diarrhoea two months earlier, considered a potential trigger for her current hyperinflammatory syndrome. Her past medical history included well-controlled hypertension, dyslipidaemia and a tonsillectomy performed in childhood. She was a former smoker, consumed alcohol socially, and reported no relevant occupational or environmental exposures. There was no personal or family history of autoimmune disorders, malignancy or immunodeficiency.
On examination, she was hypotensive but otherwise hemodynamically stable. Abdominal palpation suggested hepatomegaly. Neurological examination and the rest of the physical examination were unremarkable.
She was admitted for further evaluation. During hospitalization, her clinical course included persistent fatigue and cytopenias. Given the combination of symptoms and systemic signs, HLH was suspected, prompting further laboratory and imaging investigations. In collaboration with Hematology Department, targeted viral studies were permorfed, including Cytomegalovirus (CMV) testing, and the diagnosis was supported. Bone marrow biopsy was discussed but deferred because of her stable clinical course and progressive improvement under antiviral therapy. The patient was discharged once she was stabilised.
Initial blood tests showed anaemia (haemoglobin of 90 g/L), neutropenia (0.92x109 neutrophils/L), elevated inflammatory markers, hypertriglyceridemia (3,51mmol/L) and high ferritin (1316 µg/L), suggesting a hyperinflammatory state. Liver and renal function were mildly deranged, while coagulation studies, autoimmune panels, hormonal assays and tumour markers were unremarkable (Table 1).
Computed tomography (CT) of the thorax, abdomen, and pelvis revealed splenomegaly, mesenteric panniculitis and small-volume ascites, raising suspicion for a systemic inflammation. Positron Emission Tomography–Computed Tomography (PET-CT) showed metabolically active lymph nodes and mesenteric fat that consists of inflammatory changes, that ruled out a lymphoproliferative syndrome (Figure 1).
These findings suggested a hyperinflammatory syndrome, such as HLH. Soluble interleukin-2 receptor/CD25 levels were elevated (630 U/mL) and viral studies, including CMV serology and polymerase chain reaction (7021 copies/mL), confirmed active CMV infection, which was identified as a plausible trigger and motivated the decision to initiate targeted antiviral therapy with valganciclovir.
Several alternative diagnoses were considered. Adrenal insufficiency was considered due to hypotension and fatigue but was excluded because of normal cortisol and adrenocorticotropic hormone levels. Sepsis was unlikely, given the absence of an active bacterial or fungal infection and extremely high ferritin levels. Splenomegaly and metabolic activity on PET-CT initially suggested a possible lymphoma, but imaging findings were interpreted as reactive and no masses were detected. Paroxysmal nocturnal haemoglobinuria was excluded by negative flow cytometry. The exclusion of these possibilities, combined with compatible laboratory and imaging findings, and the presence of a viral trigger, supported secondary HLH as the most likely diagnosis [2].
The patient’s condition improved progressively during hospitalization, with gradual resolution of cytopenias and normalization of inflammatory markers, causing initiation of antiviral therapy. She remained clinically stable and did not require additional invasive interventions.
One month after discharge, laboratory parameters remained normal and CMV PCR was negative. Our patient underwent routine monitoring, in line with current guidelines for follow-up of HLH. CT imaging showed persistent but partially improved mesenteric panniculitis, suggesting that, although CMV infection had been resolved, low-grade inflammation persisted. Clinically, she continued to experience mild fatigue but returned to her baseline functional status without relapse or complications.
Table 1: Comparative table of our patient´s analytical parameters at admission and after discharge.
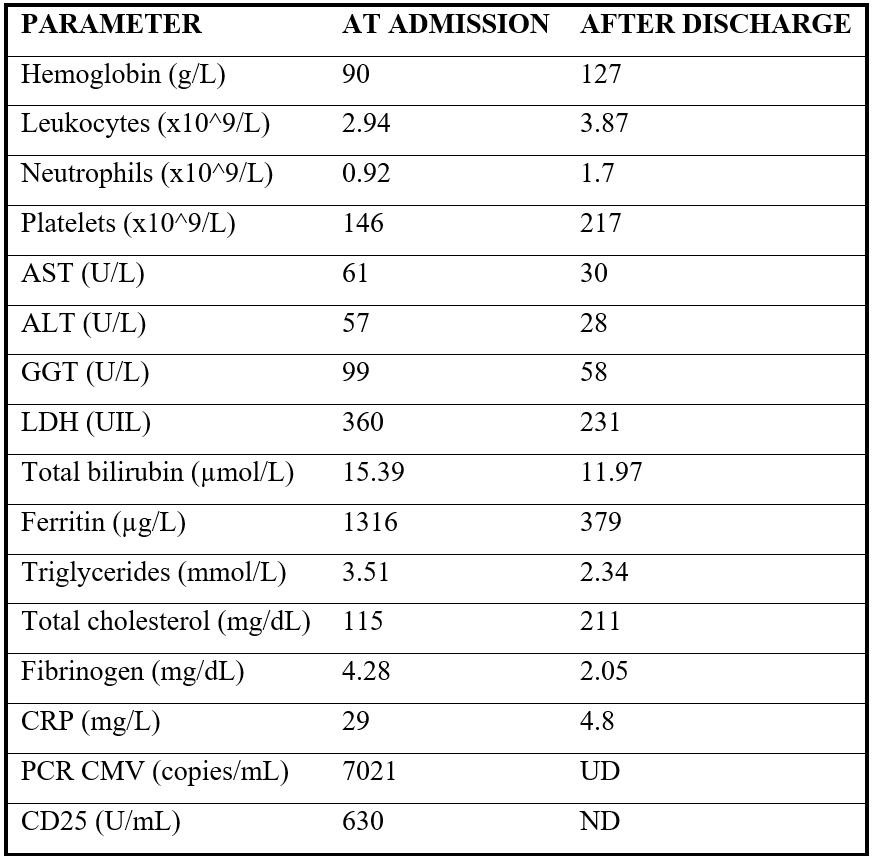
ALT: Alanine aminotransferase; AST: Aspartate aminotransferase; CMV: cytomegalovirus; CPR: C-reactive protein; GGT: gamma-glutamyl transferase; PCR: polymerase chain reaction.
Table 2: Hemophagocytic lymphohistiocytosis criteria 2024 (HLH-24) and which of them does our patient fulfill.

Table 3: HLH triggers.

EBV: Epstein Barr virus; HIV: Human immunodeficiency virus; SARS-CoV2: Severe acute respiratory syndrome coronavirus 2.
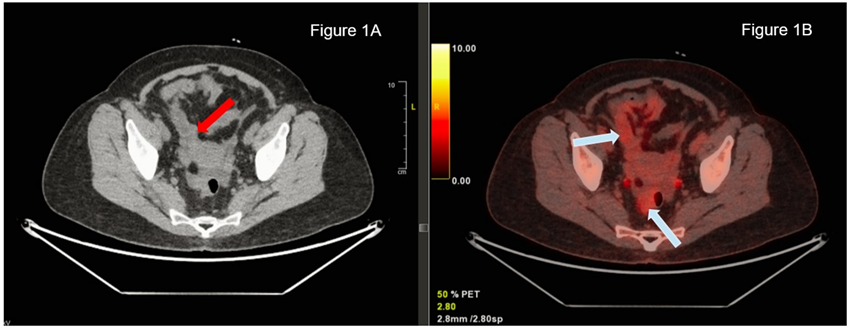
Figure 1: Mesenteric panniculitis.
Figure 1A: CT scan: Nonspecific inflammation involving the adipose tissue of the mesenter.
Figure 1B: PET-CT: Nonspecific increased metabolism (FDG uptake) due to inflammation.
Discussion
HLH is a rare, life-threatening hyperinflammatory syndrome caused by uncontrolled activation of macrophages and cytotoxic T lymphocytes, leading to excessive cytokine release and multiorgan dysfunction. While primary HLH is associated with genetic defects and typically presents in childhood, secondary HLH is increasingly recognized in adults, usually triggered by infections, malignancies, autoimmune disorders or immunosuppression. Prompt treatment of HLH is critical for successful outcomes, but the greatest barrier to a successful outcome is often a delayed diagnosis due to its rarity, variable clinical presentation and lack of specificity of clinical and laboratory findings [1].
Adult HLH usually manifests with persistent fever, hepatosplenomegaly, cytopenias, transaminase elevation, coagulopathy and markedly elevated ferritin levels. Neurological symptoms may appear in up to one-third of cases, adding further diagnostic complexity. Imaging, although sometimes is nonspecific, it often reveals hepatosplenomegaly or lymphadenopathy. Because these findings are shared with several other acute conditions, HLH requires a combination of clinical suspicion and laboratory results rather than reliance on any single abnormality [3].
Diagnosis of non-familial HLH relies on the HLH-2024 criteria, with ≥5 fulfilled items required of the following: fever, splenomegaly, cytopenias in at least two lineages, hypofibrinogenemia or hypertriglyceridemia, hyperferritinemia, haemophagocytosis in bone marrow or other tissues and elevated sCD25 (Table 2). However, these criteria were originally designed for paediatric populations with more florid presentations and which may not fully capture the subtle or evolving forms of HLH seen in adults. In this context, the H-Score offers an alternative tool incorporating variables and threshold values more applicable to adult patients. Nevertheless, both systems have limitations and clinical judgement remains essential, particularly when HLH is suspected but the patient does not fulfil complete criteria [1,4].
Our patient exemplifies this scenario. She fulfilled three of HLH-2024 criteria (splenomegaly, hypertriglyceridemia and hyperferritinemia), along with cytopenias and elevated sCD25. Although she did not meet the five-criteria requirement, the constellation of findings, clinical trajectory and absence of alternative explanations strongly suggested an HLH-like hyperinflammatory syndrome. The unique features of our case highlights how strict adherence to classical criteria may delay diagnosis in adults and supports the need for flexible diagnostic strategies, especially in the context of identifiable triggers [4,7].
Triggers of HLH are crucial to identify because treating them may lessen the severity of HLH and, in some cases, enable avoidance of more cytotoxic HLH-specific therapy. The most important triggers include infections such as Epstein-Barr Virus (EBV) and hematologic malignancies like lymphomas. Autoimmune disorders and immunodeficiencies may also contribute to the underlying pathophysiology of the disease. The development of CMV-induced HLH in an immunocompetent adult, as in our case, is therefore unusual and clinically relevant. A full list of triggers can be found in the accompanying table (Table 3) [5].
Treatment of adult HLH must be individualized and depends primarily on severity, organ involvement and the identification of an underlying trigger. When the patient is clinically stable and a treatable trigger such as infection is identified, targeted therapy alone may be sufficient (antibiotic or antiviral therapy). In contrast, unstable patients or those with rapidly progressive disease may require immediate initiation of HLH-directed therapy, typically following the HLH-94 protocol based on dexamethasone and etoposide. Hematopoietic Cell Transplantation (HCT) is reserved for CNS involvement, refractory or genetic cases. Supportive care is critical and includes management of cytopenias, coagulopathy, blood pressure and prevention of secondary infections. Targeted therapies, such as emapalumab, an IFN-γ inhibitor, are increasingly considered for relapsed or refractory HLH [3].
Prognosis of HLH depends on early recognition, the underlying cause and response to treatment. Once our patient was under treatment of valganciclovir, CMV viremia effectively suppressed and led to partial recovery of infection without the need for corticosteroids or immunosuppressive therapy. This favourable response underscores the importance of identifying infectious triggers early and supports an approach where treatment is escalated only if clinical deterioration or failure to respond occurs. The benign evolution and absence of organ failure justified a conservative strategy and reflect the better prognosis generally associated with infection-mediated HLH as compared to malignancy-associated HLH, which carries a significantly worse outcome [6,7].
Long-term follow-up is essential to monitor for recurrence, late organ dysfunction or complications from therapy. Most relapses occur within the first year of the initial episode. Recommended follow-up includes serial assessment of hematologic parameters, inflammatory markers, liver and renal function and monitoring for potential neurologic sequelae. Improvements in survival are expected to come from increased disease recognition with earlier diagnosis, reductions in HCT-associated morbidity and disease-specific immunotherapies [6].
In conclusion, this course of events illustrates how early identification of a triggering factor can inform management decisions and improve outcomes and underscores the importance of maintaining a high index of suspicion for HLH even in patients without pre-existing immunodeficiency or malignancy. Finally, our case highlights the need for continued refinement of diagnostic approaches and exploration of emerging, less toxic therapeutic strategies for virus-associated HLH [8].
Conclusion
HLH can occur in immunocompetent adults without classical risk factors and should be considered in patients with cytopenias, splenomegaly, hyperferritinemia and other signs of systemic inflammation.
Early recognition of potential triggers, particularly viral infections such as CMV, is essential, as targeted treatment may control the hyperinflammatory state and prevent the need for cytotoxic HLH-specific therapy.
Strict fulfilment of HLH-2024 diagnostic criteria may not always be present; alternative diagnostic tools such as the H-Score can support the diagnosis in early stages of the disease or atypical presentations.
Prompt multidisciplinary collaboration and careful follow-up are crucial to monitor disease resolution, detect relapse and minimise long-term complications.
Authorship Criteria
The following authors were responsible for drafting of the text, sourcing and editing of clinical images, investigation results, drawing original diagrams and algorithms, and critical revision for important intellectual content:
Marta García Gamero, Marta Abengózar Mancera, Juana Carretero Gómez, Araceli Vera Tomé
The following authors gave final approval of the manuscript:
Marta García Gamero, Marta Abengózar Mancera, Juana Carretero Gómez, Araceli Vera Tomé
Conflict of Interests: The authors declare that they have no conflicts of interest regarding the publication of this manuscript or any of the institutions mentioned herein.
Grant Information: The authors received no specific funding for this work
Acknowledgements: The authors wish to acknowledge the support of their respective institutions.
References
- Henter JI. Hemophagocytic Lymphohistiocytosis. N Engl J Med, 2025; 392: 584. doi:10.1056/NEJMra2314005.
- Holloway A, Ahmed S, Manson JJ. Does your unwell patient have haemophagocytic lymphohistiocytosis? Br J Hosp Med (Lond), 2024; 85: 1-12. doi:10.12968/hmed.2023.0394.
- Bergsten E, Horne A, Aricò M, Astigarraga I, Egeler RM, Filipovich AH, et al. Confirmed efficacy of etoposide and dexamethasone in HLH treatment: long-term results of the cooperative HLH-2004 study. Blood, 2017; 130: 2728-2738. doi:10.1182/blood-2017-05-784320.
- Lachmann G, Heeren P, Schuster FS, Nyvlt P, Spies C, Feinkohl I, et al. Multicenter validation of secondary hemophagocytic lymphohistiocytosis diagnostic criteria. J Intern Med, 2025; 297: 312-327. doi:10.1111/joim.20065.
- Ramos-Casals M, Brito-Zerón P, López-Guillermo A, Khamashta MA, Bosch X. Adult haemophagocytic syndrome. Lancet, 2014; 383: 1503-1516. doi:10.1016/S0140-6736(13)61048-X.
- Otrock ZK, Eby CS. Clinical characteristics, prognostic factors, and outcomes of adult patients with hemophagocytic lymphohistiocytosis. Am J Hematol, 2015; 90: 220–224. doi:10.1002/ajh.23911.
- Chevalier K, Schmidt J, Coppo P, Galicier L, Noël N, Lambotte O. Hemophagocytic lymphohistiocytosis associated with cytomegalovirus infection: 5 cases and a systematic review of the literature. Clin Infect Dis, 2023; 76: 351-358. doi:10.1093/cid/ciac649.
- Rolsdorph LÅ, Mosevoll KA, Reikvam H. Cytomegalovirus induced hemophagocytic lymphohistiocytosis: diagnostic and treatment challenges for the future. Expert Rev Hematol, 2022; 15: 667-670. doi:10.1080/17474086.2022.2100341.

