Chylothorax Doesn't Lie: The Forgotten Clue to Lymphoma
González González P1, Carretero Gómez J1,2,3,*, Casilda Sánchez M1, Arévalo-Lorido JC1,2,4
1Internal Medicine Department, University Hospital Complex of Badajoz, Avda de Elvas, S/N. 06085, Badajoz, Spain
2INUBE: University Institute of Biosanitary Research of Extremadura
3CICAB: Clinical research center in the Badajoz area.
4FUNDESALUD: Foundation for the Training and Research of Health Professionals of Extremadura
Received Date: 08/12/2025; Published Date: 20/02/2026
*Corresponding author: Juana Carretero Gómez, Department of Internal Medicine, University Institute for Biomedical Research of Extremadura (INUBE), Clinical Research Center of the Badajoz (CICAB), Badajoz University Hospital. Badajoz, Spain
ORCID: https://orcid.org/0000-0002-7754-6090
Abstract
Chylothorax occurs when the thoracic duct ruptures or is damaged, causing chyle to leak into the pleural space. It is classified etiologically as traumatic or non-traumatic; lymphoma accounts for 70% of cases in the latter group. Diagnosis is made through a combination of clinical evaluation, imaging studies, and pleural fluid analysis via thoracentesis to determine triglyceride and cholesterol levels.
Complications include malnutrition, immunosuppression, and respiratory compromise. Treatment will be conservative or aggressive depending on the patient's clinical condition and should be approached in a multidisciplinary manner.
This case highlights the importance of considering lymphoproliferative causes in the presence of chylous pleural effusion, even when the clinical presentation is nonspecific.
Keywords: Chylothorax; Lymphoma; Nutritional supplementation
Background
Chylothorax is a clinical condition characterized by bilateral pleural effusion secondary to injury or obstruction of the thoracic duct. While the etiologies are diverse, Lymphoproliferative Syndromes (LPS) account for 70% of non-traumatic cases. This case report presents a 63-year-old man with bilateral chylothorax as the initial manifestation of an PLS due to follicular lymphoma. The clinical and diagnostic findings are described, as well as the comprehensive therapeutic management, highlighting the importance of a multidisciplinary approach.
From a pathophysiological standpoint, chyle is composed of triglyceride-rich lymph absorbed from the gastrointestinal tract, along with lymphocytes and other immunological components. Its accumulation in the pleural cavity progressively alters respiratory mechanics and leads to significant metabolic and nutritional complications. Although chylothorax is classically associated with chest trauma or surgical procedures, in the absence of these factors, suspicion should be directed toward hematologic malignancies, especially low- or high-grade lymphomas. In fact, in many patients, as in the case presented, chylous effusion constitutes the first clinical clue to an underlying, undiagnosed process.
Early detection is crucial, as delayed recognition can lead to respiratory decompensation, secondary immunodeficiency, and a decline in overall health. Integrating imaging studies, biochemical analysis of pleural fluid, and systemic evaluation allows not only for confirming the diagnosis but also for identifying the underlying cause and planning the most appropriate therapeutic approach. In this context, a multidisciplinary team—involving Internal Medicine, Pulmonology, Radiology, and Histopathology—is key to achieving timely diagnosis and effective treatment, especially in atypical presentations such as bilateral or insidious onset.
Case Presentation
A 63-year-old male with a history of hypertension, dyslipidemia, GOLD C chronic obstructive pulmonary disease (COPD), and ICD-VR was referred from Cardiology to the Emergency Department after an echocardiogram showed a non-dilated inferior vena cava and a bilateral pleural effusion, predominantly on the left, with no apparent cardiac cause. The patient reported progressive dyspnea, a dry cough, and a 14 kg weight loss over the past year, without orthopnea, edema, or decreased urine output. On arrival, his baseline oxygen saturation was 91–92%. A chest X-ray confirmed the pleural effusion (Figure 1), which is why he was admitted to Internal Medicine for study.
On physical examination, the patient was stable, alert, and oriented, with a tendency toward hypotension. Bilateral pulmonary hypophonia was observed, extending to the mid-lung fields, without edema or signs of deep vein thrombosis. A complete blood count was ordered, and a diagnostic thoracentesis was performed.
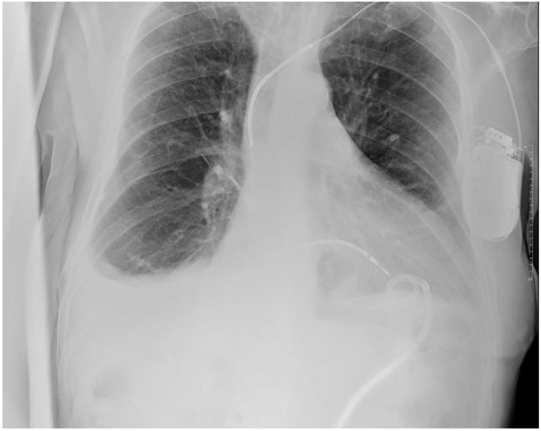
Figure 1: Chest radiography in anteroposterior projection. There is cardiomegaly and bilateral pleural effusion without condensation images.
Investigations and Treatment
Blood tests showed normal blood count and coagulation; biochemistry with total protein of 6.4 g/dL and albumin of 3.8 g/dL; normal renal function, ions and liver profile; CRP of 5 mg/L; NT-proBNP of 15 pg/mL and troponin T of 21 ng/L. The study of autoimmunity, immunoglobulins, thyroid and iron profile, and QuantiFERON were negative.
Thoracentesis yielded milky pleural fluid (Figure 2), with 85% mononuclear cells, protein of 5.0 g/dL, LDH of 171 IU/L, glucose of 109 mg/dL, cholesterol of 53 mg/dL, and triglycerides of 1060 mg/dL. Chylomicrons were identified after centrifugation, and the microbiological study was negative. Light's criteria, along with the lipid characteristics, confirmed a bilateral chylothorax compatible with a possible lymphoproliferative syndrome.
The PET-CT scan showed supra- and infradiaphragmatic lymphadenopathy, hypermetabolic thyroid lesions, and a left axillary lymph node with significant uptake (Figure 3). Colonoscopy revealed only diverticulosis. Lymphoscintigraphy of the upper and lower limbs was normal (Figure 4). Cytometry of the pleural fluid showed a predominance of T lymphocytes, and the peripheral blood smear showed no abnormalities.
Breast surgery identified a palpable left axillary lymph node, which was biopsied. Pathology confirmed high-grade follicular lymphoma, and chemotherapy was initiated. During hospitalization, the patient developed bacteremia due to S. epidermidis, treated with vancomycin, and subsequently a deep vein thrombosis in the right leg.
The management of chylothorax included drainage by repeated thoracentesis, nutritional support with digestive rest and parenteral nutrition, followed by a low-fat diet rich in medium-chain triglycerides, plus octreotide to reduce pleural output.
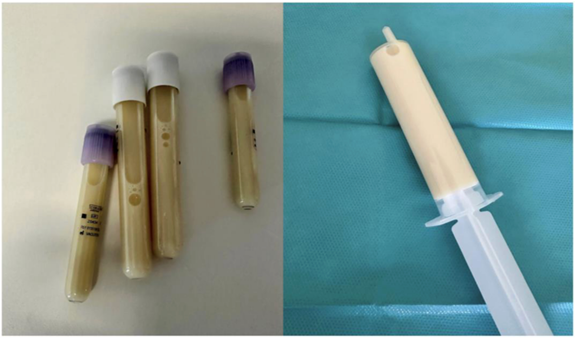
Figure 2: Tubes of milky pleural fluid obtained after thoracentesis.
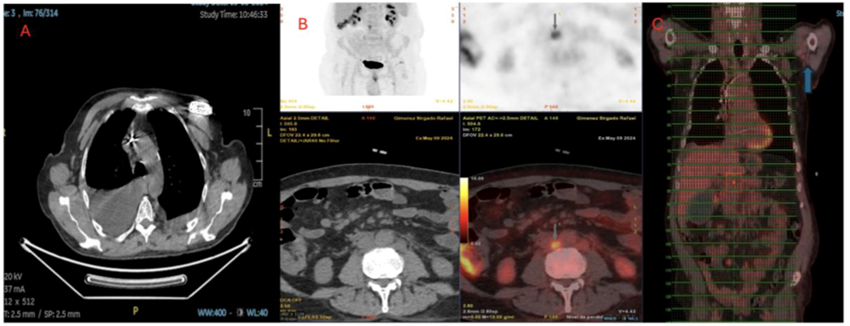
Figure 3: PET-CT. A) Axial view of the lung window reveals the presence of a larger and more extensive pleural effusion on the right side, with no metabolic activity. B) Axial view of the abdomen window reveals several lymph nodes, notable for their size and metabolism, located in the inter-aortocaval area. C) Significant metabolic uptake at the level of the left axillary lymph node.
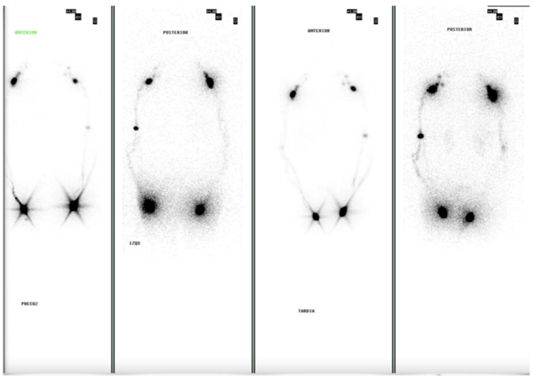
Figure 4: Lymphoscintigraphy. Physiological distribution with early tracer migration and evidence of deep lymphatic vessels with absence of subcutaneous migration.
Differential Diagnosis
The differential diagnosis of chylothorax includes several clinical entities that can present with pleural effusions of similar appearance or with biochemical characteristics that can cause confusion. One of these is pseudochylothorax, a chronic pleural effusion usually associated with tuberculosis or rheumatoid arthritis, which takes on a cloudy or yellowish appearance due to its high cholesterol content. Unlike true chylothorax, pseudochylothorax presents with low triglyceride levels, elevated cholesterol, and an absence of chylomicrons, allowing for its analytical differentiation.
Another relevant entity is pleural empyema, which may present with cloudy or even whitish fluid, mimicking chyle. However, the presence of a low pH, decreased glucose, very high LDH, and microbiological isolation clearly point to an infectious origin. Pleural effusion secondary to extreme hypertriglyceridemia should also be considered, an infrequent but possible situation in patients with very high serum triglycerides. In these cases, the fluid may be milky, although without evidence of chylomicrons and with pleural lipid concentrations not compatible with chylothorax, ruling out thoracic duct injury.
It is also necessary to differentiate chylothorax from malignant non-chylous pleural effusion. Several neoplasms, such as lung carcinoma or mesothelioma, produce lymphocytic exudative effusions that may appear cloudy. However, they lack the lipid criteria diagnostic of chylothorax and usually show malignant cells in cytological studies or specific abnormalities in flow cytometry. Finally, certain transudative pleural effusions, such as those due to heart failure or cirrhosis, may show an opalescent appearance in their initial stages. However, the application of Light's criteria, along with the low concentrations of pleural lipids, allows these diagnoses to be easily ruled out. Overall, the integration of clinical, biochemical, and imaging findings is essential to establish an accurate diagnosis and guide appropriate management.
Outcome and Follow-Up
Following the initiation of chemotherapy and conservative measures for chylothorax, the patient showed progressive clinical improvement, with a decrease in pleural effusion and improved ventilation. Radiographic evolution was favorable, without the need for further invasive interventions. Once stabilized, the patient was discharged with nutritional recommendations, joint follow-up by Internal Medicine, Pulmonology, and Hematology, and close monitoring for recurrence.
Initial outpatient visits revealed no new episodes of significant effusion, and the patient remained stable from a respiratory and systemic standpoint. Furthermore, a gradual recovery of nutritional status and improved exercise tolerance were observed, allowing for a progressive reduction in hospital visits. The patient's adherence to dietary recommendations and oncological treatment contributed significantly to their stability. No new infectious events or thrombotic complications were recorded during immediate follow-up, and the overall outcome was satisfactory, highlighting the importance of comprehensive and coordinated management among the various specialties involved.
Discussion
Chylothorax is a clinical condition caused by an injury or blockage of the thoracic duct, leading to the accumulation of chyle (lymphatic fluid from the gastrointestinal tract, especially the small intestine) in the pleural cavity.
In terms of pathophysiology, chylothorax can be classified as traumatic or non-traumatic. Traumatic chylothorax is the most common, with an incidence of up to 4%, and iatrogenic causes during surgery are the most frequent. Non-traumatic chylothorax includes various diagnoses, and neoplasms, especially lymphoma, account for 70% of cases [1].
Chylothorax typically presents with symptoms similar to those of any pleural effusion, depending on the etiology and the rate of chyle accumulation. In the early stages, patients are usually asymptomatic due to the low volume of chyle. Common symptoms include cough, fatigue, weight loss, and night sweats, especially if associated with a malignancy. Chest pain and fever are uncommon, as the chyle does not irritate the pleural surface. In chronic cases, electrolyte imbalances and nutritional deficiencies predominate [2].
The diagnosis of chylothorax is established when triglycerides in the pleural aspirate exceed 110 mg/dl and cholesterol is less than 200 mg/dl, or when the cholesterol/triglyceride ratio is < 1. However, in 15% of cases, triglycerides are less than 110 mg/dl and in 3% they are less than 50 mg/dl. In addition, Light's criteria allow the pleural effusion to be classified as transudate or exudate, the latter being the most frequent (75% of cases) [3].
The treatment of chylothorax should be individualized and is divided into three categories: treatment of the underlying cause (such as corticosteroids for sarcoidosis, diuretics for heart failure, or chemotherapy/radiotherapy for neoplasms, as in our patient), conservative treatment with a nutritional focus and medications such as somatostatin or octotropic agents, and nutritional therapy. The latter includes initial bowel rest and parenteral nutrition, followed by a lipid-lowering diet rich in easily absorbed medium-chain triglycerides without the need for pancreatic enzymes.
The surgical option, which was not necessary in our patient, is considered in cases where, despite conservative measures, a leak greater than 1.5 L/day persists or drainage persists after 2 weeks [3,4].
The originality of our case lies in the unusual bilateral presentation of the chylothorax. Typically, the exudates are lymphocytic, unilateral, and only 1 in 10 have a milky appearance. Although the current mortality rate of non-traumatic chylothorax is less than 40%, when it is associated with neoplasia and is bilateral, it is considered to have a worse prognosis [5].
In conclusion, the interest in this case lies not only in its unusual presentation but also in the diagnostic challenges and associated morbidity and mortality. The clinical and epidemiological history, as well as the presence of chylothorax, suggested the likely existence of lymphoma, a diagnosis difficult to reach due to the complementary tests performed. However, a detailed physical examination and access to a group of lymph nodes, along with immunohistochemical studies, were key to the diagnosis.
Learning Points
1. Non-traumatic chylothorax should raise suspicion of a lymphoproliferative syndrome, with lymphoma being the most frequent cause (≈70%), especially in the presence of lymphocytic pleural effusion and without an obvious cardiopulmonary etiology.
2. The presence of triglycerides >110 mg/dL and the identification of chylomicrons in the pleural fluid constitute the most reliable diagnostic criteria, even when the appearance of the effusion is not clearly milky.
3. Management requires a multidisciplinary approach, combining etiological control (in this case, chemotherapy for lymphoma), specialized nutritional support, pleural drainage, and antisecretory drugs such as octreotide.
4. A thorough physical examination and access to accessible peripheral lymph nodes are essential when imaging tests do not allow for a safe biopsy, avoiding diagnostic delays in high-impact pathologies such as lymphoma.
References
- Bhatnagar M, Fisher A, Ramsaroop S, Carter A, Pippard B. Chylothorax: pathophysiology, diagnosis and treatment: a comprehensive review. J Thorac Dis, 2024; 16: 1645-1661. doi: 10.21037/jtd-23-1636.
- Agrawal A, Chaddha U, Shojaee S, Nadolski G, Liberman M, Lee YCG, et al. Multidisciplinary management of adult patients with chylothorax: a consensus statement. Eur Respir J, 2024; 64: 2400470. doi: 10.1183/13993003.00470-2024.
- Agrawal A, Chaddha U, Shojaee S. Multidisciplinary management of adult patients with chylothorax: Consensus statement. European Respiratory Journal, 2024; 2: 400-470. DOI: https://doi.org/10.1183/1399300300470-2024
- Janjetovic S, Janning M, Daukeva L, Bokemeyer C, Fiedler W. Chylothorax in a patient with Hodgkin lymphoma: a case report and literature review. Tumori, 2013; 99: e96-9. doi: 10.1177/030089161309900324.
- Cortés Télles A, Sandoval R. An unusual presentation of follicular lymphoma. Arch Bronconeumol, 2011; 47: 56-57. English, Spanish. doi: 10.1016/j.arbres.2010.07.008.

