Posterior Reversible Encephalopathy Syndrome Linked to the Coexistence of Three Etiologies: First Case
Abdoulahy Diallo1,*, Yacouba Dembele2 and Mohamadou Niang1
1Service de Maladies Infectieuses et Tropicales, Centre Hospitalier de Mayotte, Mamoudzou, Mayotte
2Service de Radiologie, Centre Hospitalier de Mayotte, Mamoudzou, Mayotte
Received Date: 16/12/2025; Published Date: 23/01/2026
*Corresponding author: Abdoulahy Diallo, Service de Maladies Infectieuses et Tropicales, Centre Hospitalier de Mayotte, Mamoudzou, Mayotte
Abstract
Background: Posterior reversible encephalopathy syndrome (PRES) is a neurotoxic state that occurs secondary to the inability of the posterior circulation to autoregulate in response to acute changes in blood pressure.
Case presentation: Here, we’re probably describing a first case in a 10-year-old patient, varicella vaccine up to date, who has developed clinical and radiological characteristics of PRES related to steroid-resistant nephrotic syndrome treated with tacrolimus and progressing to nosocomial varicella encephalitis. Controlled brain MRI 6 months later showed regression of lesions after treatment with rituximab.
Conclusion: PRES should always be considered in pediatric patients with nephrotic syndrome who develop an unexpected episode of neurological signs, especially if they have systemic hypertension and are on prolonged immunosuppressive therapy. The treatment strategy concerns early diagnosis and general measures to correct the underlying cause of PRES.
Keywords: Child, Steroid-resistant Nephrotic Syndrome, Tacrolimus, Chickenpox Encephalitis Nosocomial, Posterior reversible encephalopathy syndrome (PRES)
Abbreviation: Ac: Antibody; ADC: Apparent Diffusion Coefficient; CSF: Cephalo-Spinal Fluid; DWI: Diffusion Imaging; EEG: Electroencephalography; HIV: Human Immunodeficiency Virus; IgG: Immunoglobulin G; IgM: Immunoglobulin M; IV: Intravenous; IEC: Conversion Enzyme Inhibitors; MRI: Magnetic Resonance Imaging; PRES: Posterior Reversible Encephalopathy Syndrome; Pu/Cu: Urinary protein to urinary Creatinine ratio; PCR: Polymerase Chain Reaction; VZV: Varicella Zoster Virus
Introduction
Posterior Reversible Encephalopathy Syndrome (PRES), also known as Reversible Posterior Leukoencephalopathy Syndrome (RPLS), is a neurotoxic state that occurs secondary to the inability of the posterior circulation to autoregulate in response to acute changes in blood pressure. Hyperperfusion with resultant disruption of the blood-brain barrier results in vasogenic edema, usually without infarction, most commonly in the parieto-occipital regions [1]. The Reversible Posterior Encephalopathy Syndrome (PRES) can develop in association with a wide range of clinical conditions including a steroid-resistant nephrotic syndrome, especially along with hypertension [2], immune-modifying (tacrolimus) [3,4], Varicella Encephalitis [5].
Here, we’re probably describing a first case in a 10-year-old patient, varicella vaccine up to date, who has developed clinical and radiological characteristics of PRES related to steroid-resistant nephrotic syndrome treated with tacrolimus and progressing to nosocomial varicella encephalitis.
Case Report
A 10-year-old girl, followed in her home country for nephrotic syndrome treated with oral corticosteroids and thiazides without remission, was admitted to our pediatric unit for severe generalized body edema, confirmed pure nephrotic syndrome with Pu/Cu ratio 320 mg/mmol and hypoalbuminemia at 20 g/L. His blood pressure was 129/75mmHg and heart rate was 86 beats/min. A renal ultrasound showed an appearance of bilateral nephritis focus. The etiological balance of nephrotic syndrome came back negative.
Treat with Enalapril (before the anti-proteinuria action of the IEC) for his high blood pressure with good blood pressure remission. Oral corticosteroids of cortancyl (60 mg/day) and methylprednisolone bolus (560 mg/day) over a one-month period were initiated but without biological improvement (persistence of proteinuria).
The diagnosis of steroid-resistant nephrotic syndrome was made. Treatment with tacrolimus 6 mg per day was initiated with improvement of his general condition.
On the fifth day on corticosteroids and immunosuppressants appeared a febrile peak at 38.9°C with predominant abdominal pain on the left flank and a vesicular rash on the entire skin surface, very pruritic with neurological degradation (vomiting, aphasia, impaired alertness, followed by crisis).
Transferred to the Pediatric Intensive Care Unit with tacrolimus discontinued in the event of PRES syndrome. She received one ampoule of benzodiazepines (diazepam) intrarectally, clonazepam IV 0.03 mg/kg and levetiracetam 20 mg/kg/day. Absence of recurrence of crisis in the aftermath.
An electroencephalography (EEG) found a broad and slow background rhythm for the age of the delta band. No variation of the plot as a function of waking states. No paroxysmal anomaly. No seizure. A brain scan (Figure 1) showed a supra tentorial leptomengitis without abscess or brain complication.
Lumbar puncture revealed leukocytes at 2/mm3 and proteinorachie 0.24 g/L, hyperglycorachie at 6.18 mol/L, direct examination and culture were negative. VZV PCR was positive in CSF and serology of varicella zoster virus infections (VZV) Ac anti-VZV IgG Positive (1,449 mUI/ml) Ac anti-VZV IgM Positive [2,8].
The diagnosis of nosocomial VZV encephalitis in an epidemic situation in the service was confirmed and treated with Aciclovir IV 60 mg/kg/d for 21 days. Resumption with increased corticosteroid therapy at 60 mg/m2 before stopping Tacrolimus.
At J5 back from resuscitation, she showed a decrease in visual acuity, eye exam was normal. A magnetic resonance cerebral angiography was performed showing right lateral and temporo-frontal parieto-occipital lesions consistent with reversible posterior leukoencephalopathy (Figure 2-1).
After a nephro-pediatric opinion, it was decided to continue corticosteroids and a dose of rituximab at 375 mg/m2 (if Absence of proteinuria and Ig anti-VZV positive). At the end of his hospitalization, blood pressure normalized with a good biological clinico evolution. Brain magnetic resonance imaging 6 months later to show a regression of lesions (Figure 2 - 2).
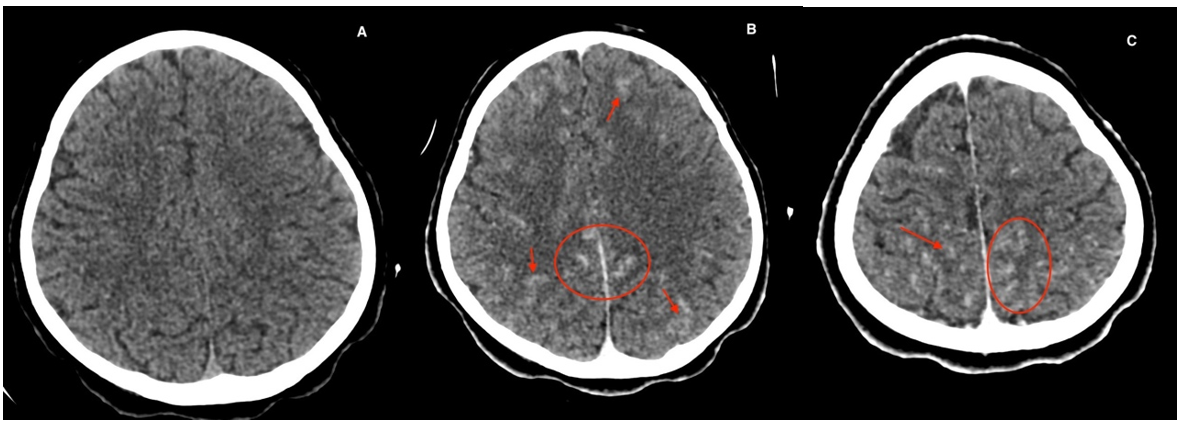
Figure 1: Brain scan performed without (a) and after injection (b and c) of iodized product, showing a bilateral gyriform contrast enhancement (red arrows and circles) in favor of supratentorial leptomeningitis without abscess or encephalic complication.
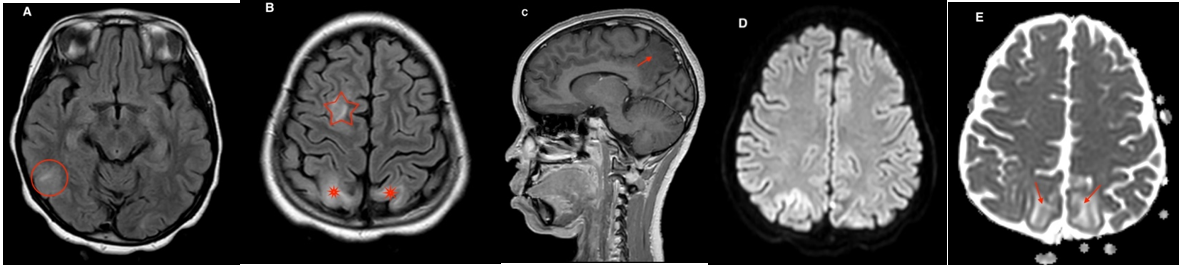
Figure 2 – 1: A brain MRI in axial FLAIR (A and B), in sagittal T1 FFE with gadolinium injection (C) and DWI with ADC (E and F) found right temporal, and bilateral parietal and occipital lesions in hyper intense FLAIR (red circle and stars), without restriction on ADC mapping (red arrows), and showing no enhancement in T1 after injection (red arrow).
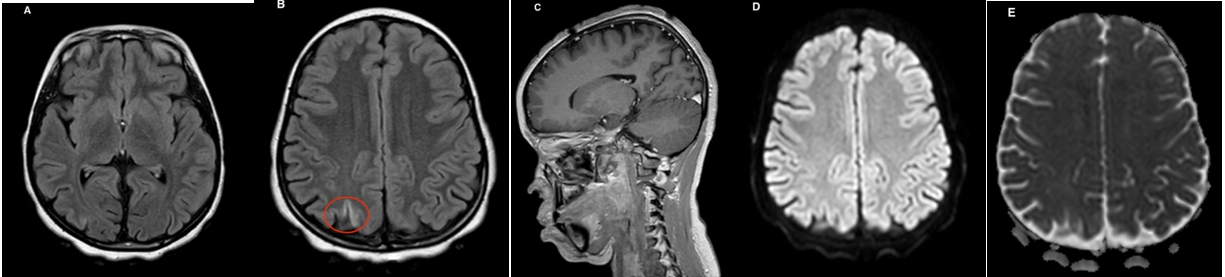
Figure 2 – 2: A brain MRI control at 6 months in axial FLAIR (A and B), DWI with ADC mapping (D and E) and sagittal T1 FFE after injection of gadolinium (C) shows a clear regression of the parieto-occipital lesions, and persistence of a small right parietal hyper intense in FLAIR (red circle).
Discussion
We probably report a single case involving three etiologies related to PRES that are the systemic disorders (Steroid-resistant nephrotic syndrome especially along with hypertension), the immune-modifying drugs (Tacrolimus) and the chickenpox Encephalitis nosocomial in a 10-year-old girl who is up to date with her varicella vaccine.
A review of the literature reported a case of steroid-resistant nephrotic syndrome, particularly with hypertension contributes prominently to the etiopathogenesis and development of PRES [9] concluded that these two entities should be considered risk factors for the development of PRES in children. Awareness must be crucial for timely diagnosis and treatment, and thus to minimize the risk of permanent neurological deficits. In addition, the case suggested that immunosuppressive agents could be used with caution in steroid-resistant nephrotic syndrome with PRES when patients present with severe edema and extreme hypoalbuminemia as in our 10-year-old patient [10].
A first hypothesis of a case of VZV vasculopathy associated with PRES in an HIV patient was raised by Sarah C. Sasson et al in 2013 [11] and also another first hypothesis of a case of varicella encephalitis associated with PRES without usual risk factor (immunodepression, epidemics...) was raised by Huzaifa Dawood et al in 2021 [12] and recommended conducting epidemiological studies to better understand the pathophysiological mechanisms involved, as early identification leads to better patient outcomes.
Neuroimaging has been considered to be central to the diagnosis of PRES [13].
In our case, at J5 back from resuscitation, she showed a decrease in visual acuity, eye exam was normal. A magnetic resonance cerebral angiography was performed showing right lateral and temporo-frontal parieto-occipital lesions consistent with reversible posterior leukoencephalopathy.
According to the literature, despite the involvement of the occipital region often during a PRES, only 39% of patients have visual symptoms. These disorders include reduced visual acuity, diplopia, visual field deficits, cortical blindness, abnormal color vision and visual hallucinations. [14,15]
Various case series have described imaging abnormalities in the parieto-occipital region (in 65 to 99% of cases), frontal region (in 54 to 88%), temporal region (in 68%). [16,17]
MRI monitoring of brain injuries is crucial, not only to confirm the diagnosis of PRES, but also to assess the effectiveness of treatment.
Conclusion
PRES should always be considered in pediatric patients with nephrotic syndrome who develop an unexpected episode of neurological signs, especially if they have systemic hypertension and are on prolonged immunosuppressive therapy. Infections can be a trigger for PRES.
The MRI scatter sequence is the best diagnostic tool, and lesions appear as diffuse posterior hyperintensities of white matter in T2, FLAIR sequences and hyper intense in scatter, with a high ADC coefficient. The treatment strategy concerns early diagnosis and general measures to correct the underlying cause of PRES.
Conflict of interest: The authors declare that they have no conflict of interest.
Ethical approval: The authors certify that they comply with the Principles of Ethical Publishing.
Authors’ contributions:
AD and YD were involved in the design of the study, data collection, supervision, data processing, image cleaning, analysis and interpretation, and manuscript writing.
MN reviewed the manuscript and submitted it.
All authors made an important contribution and approved the final version of the manuscript.
References
- Gaillard F, Sharma R, Jaber H, et al. Posterior reversible encephalopathy syndrome. Reference article, Radiopaedia.org, 2024. https://doi.org/10.53347/rID-1915.
- Thavamani A, Umapathi KK, Puliyel M, Super D, Allareddy V, Ghori A. Epidemiology, comorbidities, and outcomes of posterior reversible encephalopathy syndrome in children in the United States. Pediatr Neurol, 2020; 103: 21–26. doi: 10.1016/j.pediatrneurol.2019.07.007.
- Gera DN, Patil SB, Iyer A, Kute VB, et al. Posterior reversible encephalopathy syndrome in children with kidney disease. Indian J Nephrol, 2014; 24(1): 28-34. https://doi.org/10.4103 /0971-4065.125053.
- Childhood posterior reversible encephalopathy syndrome: clinicoradiological characteristics, managements, and outcome. Chen TH. Front Pediatr, 2020; 8. doi: 10.3389/fped.2020.00585.
- Yu Zhang, Jianhua Zhou, Yu Chen. Posterior reversible encephalopathy syndrome in a child with steroid-resistant nephrotic syndrome: a case report and review of literature. Int J Clin Exp Pathol, 2014; 7(7): 4433-4437
- Granata G, Greco A, Iannella G, et al. Posterior reversible encephalopathy syndrome— insight into pathogenesis, clinical variants and treatment approaches. Autoimmun Rev, 2015; 14: 830-836.
- Largeau B, Boels D, Victorri-Vigneau C, Cohen C, Salmon Gandonnière C, Ehrmann S. Posterior reversible encephalopathy syndrome in clinical toxicology: a systematic review of published case reports. Front Neurol, 2019; 10: 1420.
- Dawood H, Nasir S, Ahmed M, et al. Posterior Reversible Encephalopathy Syndrome Secondary to Varicella Encephalitis. Cureus, 2021; 13(1): e12484. DOI 10.7759/cureus.12484.
- Hinchey J, Chaves C, Appignani B, et al. A reversible posterior leukoencephalopathy syndrome. N Engl J Med, 1996; 334: 494–500.
- Yu Zhang, Jianhua Zhou, Yu Chen. Posterior reversible encephalopathy syndrome in a child with steroid-resistant nephrotic syndrome: a case report and review of literature. Int J Clin Exp Pathol, 2014; 7(7): 4433-4437.
- Sarah C Sasson et al. Posterior reversible encephalopathy syndrome (PRES) in an HIV-1 infected patient with disseminated varicella zoster virus: a case report. BMC Infectious Diseases, 2013; 13: 396.
- Dawood H, Nasir S, Ahmed M, et al. Posterior Reversible Encephalopathy Syndrome Secondary to Varicella Encephalitis. Cureus, 2021; 13(1): e12484. DOI 10.7759/cureus.12484.
- Hinchey J, Chaves C, Appignani B, et al. A reversible posterior leukoencephalopathy syndrome. N Engl J Med, 1996; 334: 494-500.
- Lee VH, Wijdicks EFM, Manno EM, et al. Clinical spectrum of reversible posterior leukoencephalopathy syndrome. Arch Neurol, 2008; 65: 205–120.
- Legriel S, Schraub O, Azoulay E, et al. Determinants of recovery from severe posterior reversible encephalopathy syndrome. PLoS One, 2012; 7: e44534.
- Fugate JE, Claassen DO, Cloft HJ, Kallmes DF, Kozak OS, Rabinstein AA. Posterior reversible encephalopathy syndrome: associated clinical and radiologic findings. Mayo Clin Proc, 2010; 85: 427-432.
- Covarrubias DJ, Luetmer PH, Campeau NG. Posterior reversible encephalopathy syndrome: prognostic utility of quantitative diffusion-weighted MR images. AJNR Am J Neuroradiol, 2002; 23: 1038-1048.

