Marine-Lenhart Syndrome : A Rare Presentation of Thyroid Disease
Ibrahima Amadou Dembélé1,*, Stéphane Loique Djeugoue1,5, Kaly Keïta1, Drissa Traore3, Adama Sinayoko1, Mamadou Cissoko1, Ibrahima Sagara8, Jordane Anaïs MK Fianyoh1, Ismaël Salami2, Florent Mbakilayi Tshitenga6, Soumare Assitan1, Samba Camara1, Hamsatou Sadou Diallo1, Christian Aubrey K Fondjo1, Clémence Sefako Kpokou1, Landouré Sekou1, Aoua Diarra1, Yacouba Koné1, Oumou Dembélé1, Nouhoum Koné1, Moussa Sangaré1, Romuald Nounga Nyanké1, Costa K Biakudia7, Mamadou Mallé1, Amadou Koné2, Sy Djibril1,4, Djénébou Traoré1,4, Didier Mukeba Tshialala5, Soukho Assétou Kaya1,4, Dembélé Mamadou1,4 and Hamar Alassane Traore1,4
1Internal Medicine Department, University Hospital Center Point G, Bamako, Mali
2Endocrinology and Metabolism Department of the Mali University Hospital, Bamako, Mali
3Surgery Department B, University Hospital Center Point G, Bamako, Mali
4Faculty of Medicine and Odontostomatology, University of Sciences, Techniques and Technologies of Bamako, Mali
5Faculty of Medicine, Pharmacy and Public Health, University of Mbuji-Mayi, Mbuji-Mayi, Democratic Republic of Congo
6Internal Medicine Department, Faculty of Medicine, Official University of Mbuji-Mayi, Mbuji-Mayi, Democratic Republic of Congo
7Pediatrics Department, Monkolé Hospital, Kinshasa, Democratic Republic of Congo
8Clinique Almed, Bamako, Mali
Received Date: 27/08/2025; Published Date: 07/10/2025
*Corresponding author: Ibrahima Amadou Dembélé, Internal Medicine, University Hospital Center Point G, Bamako, Mali
Abstract
Background : Marine-Lenhart syndrome (MLS) is now understood to be a combination of Graves’ disease and autonomously functioning thyroid nodule(s) (AFTNs).
Clinical Case : This article describes the case of a 14-year-old patient in whom we diagnosed Graves' disease and isthmic thyroid nodules suggestive of Marine Lehnart syndrome. It also reviews the existing literature on the diagnosis and treatment of the disease.
Conclusion : Marine-Lenhart syndrome is a rare entity of Graves' disease, coexisting with autonomous hyper- or hypofunctional nodules on thyroid scintigraphy. The diagnostic and therapeutic difficulty in our context lies in the lack of scintigraphy to classify the pathology and to search for cold nodules at risk of malignancy and poor prognosis for patients.
Keywords : Graves’ disease, thyroid nodule, Marine-Lenhart syndrome, Endocrinology, Internal Medicine
Introduction
Hyperthyroidism is a medical condition that results from an overactive thyroid gland. Graves’ Disease (GD) is the most common cause of hyperthyroidism [1]. This autoimmune disorder is characterized by the presence of autoantibodies that activate the thyrotropin receptors in thyroid cells [1]. This activation leads to an increase in the synthesis and release of thyroid hormones [3]. Marine-Lenhart syndrome (MLS) is the coexistence of Graves disease with autonomous functioning thyroid nodule(s) (AFTN) such as toxic adenoma or toxic multinodular goiter ; the two diseases may coexist or may be present at different times in the same patient, which can occur in up to 35% of patients and are functioning adenomas in 0.8–2.7% of cases [1,3,6]. A thyroid nodule is a localized hypertrophy, occurring within the thyroid gland [1]. This is a rare condition, and the reported prevalence is approximately 0.26% to 4.1% [3]. Patients with MLS have a lower remission rate with oral antithyroid drugs, requiring definitive management therapies with radioactive iodine or surgery [5]. MLS has been primarily described in adult female subjects; and very few cases of this manifestation in a male subject have been described; thus, we report one of the rare cases of a young patient with Marine-Lenhart syndrome.
Observation
This was a young patient of 14 years of age, of Malian nationality, with only biological hyperthyroidism, rhinosinusitis and a notion of food allergy as medical history ; and who had been transferred to us for etiological research and further treatment. He complained at the entrance of palpitations and unquantified weight loss.
General examination revealed a patient who was conscious, ambulatory, independent, and afebrile to touch; his Karnofsky score was 90%. His sitting blood pressure in the left arm was 159/79 mmHg ; his heart rate was 142 bpm ; his respiratory rate was 20 cycles per minute ; his left axillary temperature was 38.9°C, his SpO2 was 99% on room air ; and his capillary blood glucose was not taken g/L. His measurements at admission were as follows: weight 52 kg, height 163 cm, and body mass index 19.57 kg/m².
On examination of the devices, bilateral exophthalmos, an antero-cervical swelling mobile on swallowing, smooth surface, firm consistency presenting a systolic murmur on auscultation with a cervical perimeter at 42cm, concluding with a WHO stage 2 goiter; the heart sounds are audible, rapid at 142bpm and regular. The other devices were carefully examined and did not present any abnormalities.
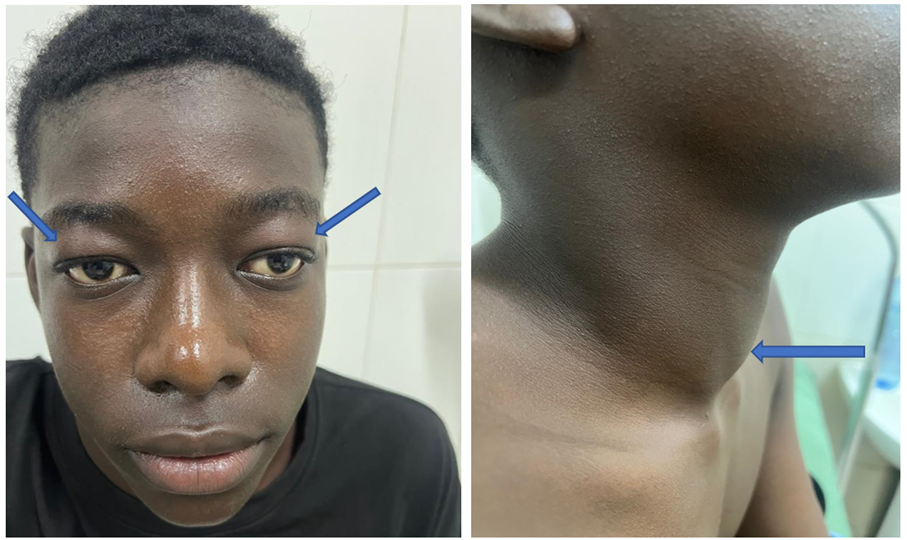
Figure 1: Pictures of the patient's face (Bilateral proptosis) and neck (Neck swelling related to WHO grade 2 goiter).
The paraclinical evaluation showed:
- Mild anemia, microcytic hypochromic with a hemoglobin level of 12,8 g/dL, mean corpuscular volume of 72.9 fL, MCHC of 30.5 g/dL.
- White blood cells at 7300/mm3, PNN : 5840/mm3, Lymphocytes : 950/mm3ALAT of 46 IU/L and ASAT of 41 IU/L
- TSHus at 0,0000 µiu/L and FT4 superior at 70 pmol/L
- Immunological assessment : Anti-TSH stimulating receptor Ab positive at 25.47 UI (N<3.10) ; Negative anti-TPO Ac
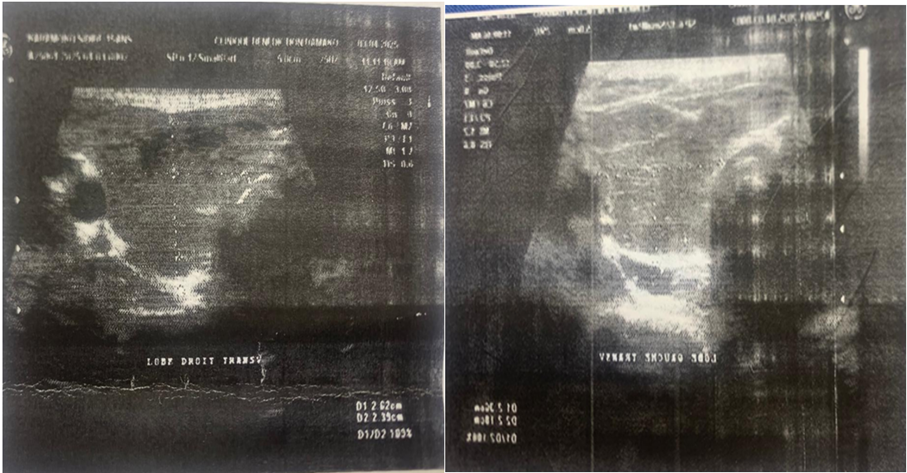
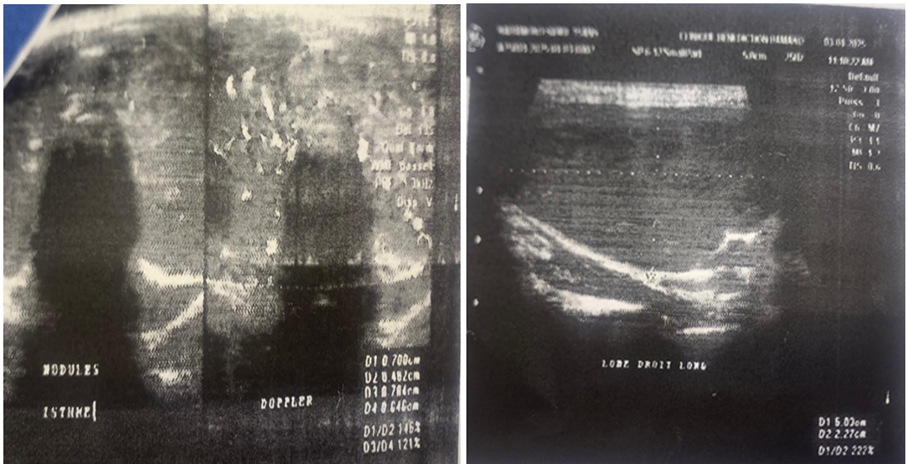
Figure 2: Thyroid ultrasound of the patient (As a clue by the arrows, 2 very vascularized isthmic nodules ; on a heterogeneous and hypervascularized goiter).
Cervical ultrasound (Figure 2) revealed the following :
- An enlarged right lobe, echogenic, heterogeneous, and measuring 50x26x21mm
- An enlarged left lobe, measuring 50x26x23mm, echogenic and heterogeneous
- Isthmic hypertrophy (11mm thick), grossly echogenic, heterogeneous, and presenting two nodules measuring 07x06mm and 07x04mm
- No cervical lymphadenopathy, and the jugulocarotid bundles appeared normal
- On color Doppler, diffuse and perinodular hypervascularization of the thyroid gland related to thyroid overactivity
A thyroid scintigraphy was requested but not performed.
We have retained the diagnosis of Marine-Lehnart Syndrome associating Graves' disease and toxic thyroid nodules.
Treatment consisted of cessation of all physical activity and bed rest at home, propranolol 40 mg morning and evening after meals, and carbimazole 40 mg in the morning after breakfast. Blood count and transaminase levels were monitored every 10 to 15 days for the first 2 months of treatment. The outcome was favorable after 2 months of treatment, marked by an absence of complaints, a slight regression of bilateral exophthalmos, and a normalization of heart rate. On the paraclinical level, TSHus at 0.000 mIU/l, FT4 at 12.55 pmol/l, hemoglobin level at 12.8 g/l, white blood cells at 4900/mm3 with neutrophil polynuclear cells at 1860/mm3, normal transaminases (ALAT : 36 IU/l, ASAT : 39 IU/l). Carbimazole was reduced to 20 mg per day (maintenance treatment) and propranolol was stopped. A thyroid hormone assessment was performed a month later, still demonstrating euthyroidism. The patient underwent a total thyroidectomy (Figure 3) with thyroid and nodular biopsy; and the patient was supplemented with Levothyroxine 12.5 micrograms per day.
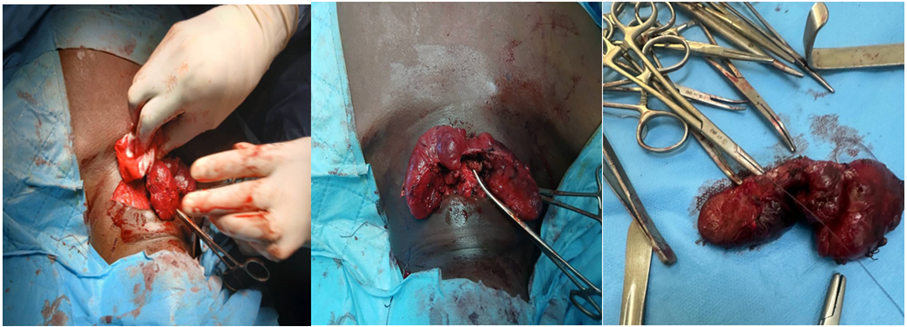
Figure 3: Enlarged thyroid of patient with isthmic nodules.
The histopathological examination of the thyroid biopsy specimens and thyroid nodules revealed the following (Figure 4). The thyroid tissue fragments showed follicles arranged in nodules, separated by connective tissue septa, without any atypical features. The follicular epithelium was regular and cuboidal. In addition, there were follicles lined with cuboidal epithelium exhibiting pseudo-papillary projections, along with fibrosis, congestion, and a lymphocytic inflammatory infiltrate, all without any signs of malignancy. The colloid was pale with resorption vacuoles at the periphery. In conclusion, this is a case of nodular goiter associated with Graves' disease, consistent with the Marine-Lenhart syndrome. He is regularly monitored by the General Surgery and Internal Medicine teams.
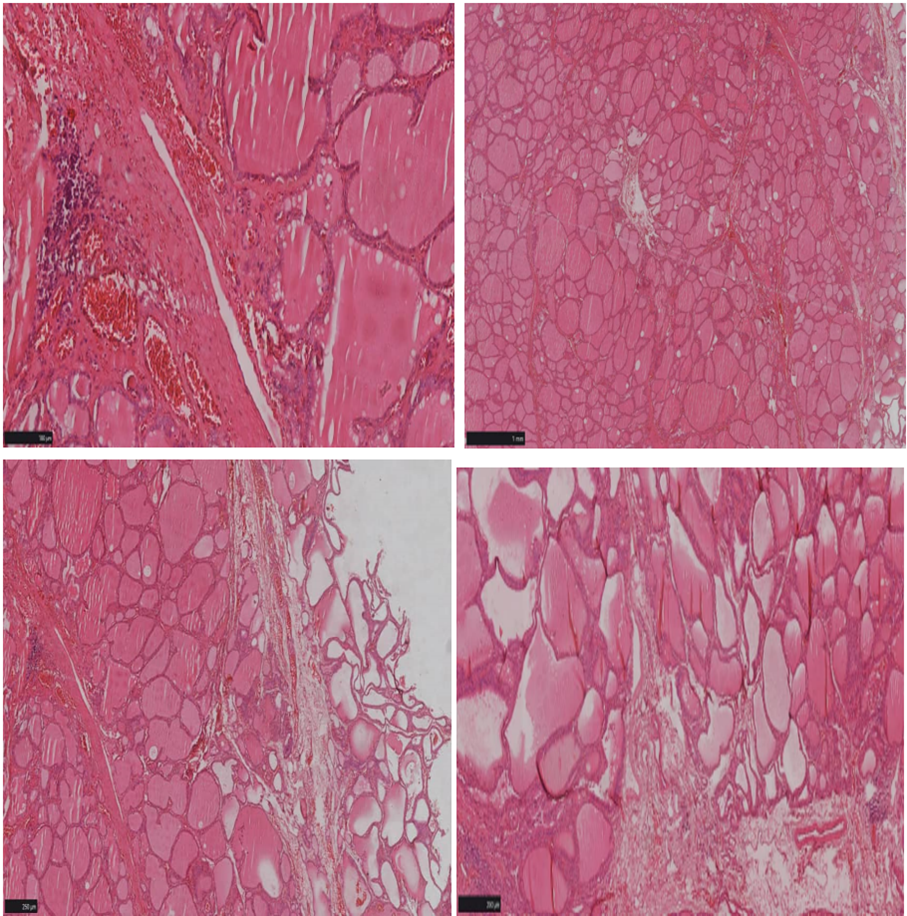
Figure 4: Histological examination of the thyroid gland.
Discussion
Doctors David Marine and Carl H. Lenhart in Ohio, United States, 110 years ago, published a series of cases with exophthalmic goiter (corresponding to GD), finding hyper-uptake nodules in eight of them, which is why it was considered a rare and interesting variant of this disease [15]. David Charkes in 1972, first coined the term “MLS” after studying 10 patients (2.7%) of functioning nodules in a series of 375 patients with GD and was the first to introduce the eponymous “Marine-Lenhart syndrome.” This condition is estimated to occur in 0.8%-2.7% of patients with GD [4,5, 14, 15]. He also proposed specific diagnostic criteria for this syndrome. According to these criteria, the diagnosis of MLS includes thyroid function tests indicative of hyperthyroidism along with positive serological tests, increased radioiodine uptake displaying either hot or cool nodules, evidence of thyroid nodularity on ultrasound, and a thyroid biopsy showing hyperplastic lesions or follicular adenoma [4,5,14,15]. These features collectively distinguish MLS from other similar disorders, such as toxic multinodular goiter or Plummer’s disease, which are important differentials for this condition.
Danno found during his study the rate of MLS cases was 0.26% among all patients with Graves’ disease (22/8,343), the median age was 57 (range 20–81) years in the MLS group [7]. Most of the cases described in the literature are in adults or elderly people, most often female; but very rarely in an adolescent male. [4,7,12,13].
Within the pathophysiological aspects, a unifying hypothesis has been proposed, which suggests that in patients with hyperfunctioning diffuse goiters, the preferential development of diffuse or nodular follicular hyperplasia may depend on the intrinsic function, concentration, and higher selective affinity of receptorstimulating immunoglobulins of TSH that allow the development of TA [5]. It is not clear if the hyperfunctioning nodules represent a form of localized - autoimmune Graves’ disease or if they could be an acquired and localized mutation in the TSH receptor gene, producing a constitutive activation of the TSH receptor, which leads to the development of a toxic adenoma [8]. The activity of toxic thyroid nodules can be enhanced by stimulators such as TSH or antibodies against the TSH receptor.
MLS is categorized into three subtypes based on scintigraphy patterns [5].
- Type 1 is characterized by high uptake in both the thyroid and a single autonomous functioning nodule.
- Type 2 exhibits increased uptake in the thyroid along with several hot nodules.
- Type 3 shares similarities with Types 1 and 2 but additionally includes cold nodules
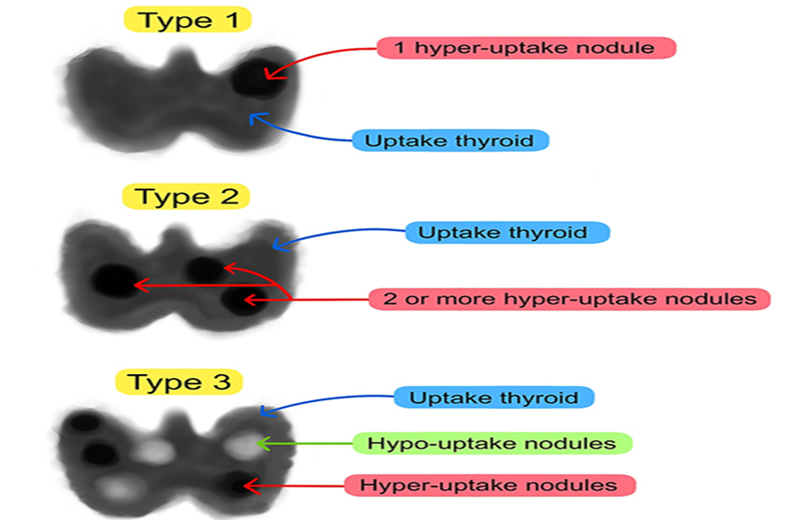
Figure 5: Scintigraphic classification of Marine Lenhart Syndrome [5].
In the case presented, a thyroid scintigraphy was requested but not performed.
Currently, there is no standardized management protocol for MLS [8]. Anti-thyroid drugs (ATDs), while commonly used, may not always restore normal thyroid function due to potentially missed active nodules, as observed in our case. Radioactive iodine ablation emerges as a viable option, particularly because of the increased tracer uptake demonstrated in scintigraphy. However, patients might require higher doses due to relative resistance to radioiodine, although this approach does offer the potential for a permanent resolution of the disease [7,8]. The hyperthyroidism management guidelines and the nuclear medicine guidelines consider patients with MLS as candidates for iodotherapy, in the absence of contraindications for its use [5]. Alternatively, surgical intervention can definitively resolve the issue of multiples nodules but comes with its own risks, including permanent hypothyroidism, potential iatrogenic hypoparathyroidism, and damage to the recurrent laryngeal nerve [8]. In the study of Danno, ATDs constituted a significantly higher proportion at 44.1% (8/18) in the MLS group [7]. In our case, the patient received as a synthetic anti-thyroid drug, carbimazole in an attack dose then in a maintenance dose associated with a beta-blocker, notably bisoprolol ; and therapeutic education of the patient and his family. The outcome was favorable and a total thyroidectomy is planned after achieving euthyroidism in the patient.
Conclusion
Marine-Lenhart syndrome is a rare entity of Graves' disease, coexisting with autonomous hyper- or hypofunctional nodules on thyroid scintigraphy. This entity requires special attention because the pathology can present a serious picture that may require total ablation of the thyroid gland. The diagnostic and therapeutic difficulty in our context lies in the lack of scintigraphy to classify the pathology and to search for cold nodules at risk of malignancy and poor prognosis for patients.
Additionnal Information
Declaration of patient consent : Written informed consent was obtained from the patient’s family to publish this report in accordance with patient consent policies. In this report, the patient and his family consented to the publication of his images and other clinical information in the journal. He understands that his name and initials will not be published and that every effort will be made to conceal his identity, but that anonymity cannot be guaranteed.
Authors’ Contributions : All authors participated in the evaluation and follow-up of the patient, in the writing and correction of the case report. All the authors of the manuscript have read and agreed to its content.
Competing interests : The authors declare no conflict of interest.
Funding : No funding was required in the preparation of this manuscript
References
- Jean-Louis Wémeau. Les Maladies de la thyroïde. 2e édition, Paris: Elsevier Masson SAS, 2022; p.331.
- Collège des enseignants d'endocrinologie, diabète et maladies métaboliques. Endocrinologie, diabétologie et maladies métaboliques. 5e édition, Paris : Elsevier Masson, 2021; p.498.
- Oyiyechukwu Onwudiwe, Huong Nguyen. Marine-Lenhart Syndrome : A Rare Presentation of Thyroid Disease. AACE Clinical Case Rep, 2023; 9: 23–24.
- Jha V, Das L, Baruah MM, Sood A, Bhadada SK. Type 1 Marine-Lenhart syndrome : An uncommon cause of thyrotoxicosis. IJEM Case Rep, 2024; 2: 130-133.
- Builes Barrera C, Castaño P, Herrera Revollo P, et al. Type 2 Marine-Lenhart Syndrome : An Uncommon Cause of Thyrotoxicosis. Cureus, 2021; 13(12): e20558.
- Ribeiro FCM, Ruas LA, Júnior AL, de Sousa AA, Araújo AF, Mwambire J, et al. Graves’ Disease and Marine Lenhart Syndrome : A Rare Clinical Presentation. Health, 2019; 11: 1169-1176.
- Danno H. Prevalence and Treatment Outcomes of Marine Lenhart Syndrome in Japan. Eur Thyroid J, 2021; 10(6): 461-467.
- Bryan A, Ramos MGM. Marine-Lenhart Syndrome about a Case. Medi Clin Case Rep J, 2024; 2(1): 205-207.
- Danielle Neuman, Russ Kuker, Francesco Vendrame. Marine-Lenhart Syndrome: Case Report, Diagnosis, and Management. Case Reports in Endocrinology, 2018: Article ID 3268010: 4 pages.
- Janki Patel, Nishok Karthikeyan. Diagnosis And Management Of A Rare Case Of Marine-Lenhart Syndrome. J Endocrine Soc, 2022; 6(1).
- Swayamsidha Mangaraj, VaIbhav Pathak, rukma Narkar, arun kumar Choudhury, anoj kumar balIarsnha. Marine-Lenhart Syndrome- A Curious Case of Thyrotoxicosis. Journal of Clinical and Diagnostic Research, 2020; 14(1): OD09-OD11.
- Chkikar, Sofia. “Marine-Lenhart syndrome : The of Nuclear Medicine in Diagnosis and Management.” J Nucl Med Radiat Ther, 2024; 15: 584.
- Agrawal K, Patro PS, Meher BR, Gnanasegaran G. Prevalence of Marine-Lenhart syndrome on 99mTc-thyroid scintigraphy and response to radioiodine : A single institutional retrospective study. World J Nucl Med, 2021; 20: 369-373.
- Charkes ND. “Graves’ disease with functioning nodules (Marine-Lenhart syndrome).,” Journal of Nuclear Medicine, 1972; 13(12): pp. 885–892.
- Marine D, Lenhart CH. “Pathological anatomy of exophthalmic goiter: The anatomical and physiological relations of the thyroid gland to the disease; the treatment,” JAMA Internal Medicine, 1911; VIII(3): pp. 265–316.

