Unveiling the Uncommon: Uterine PEComa
Chaviaropoulou A1,#,*, Argyriadis A2,#, Kalfoutzos K3, Suh M4, Braun S5 and Jackisch C6
1Varisano Klinikum Hoechst GmbH, Department of Gynecology and Obstetrics, Frankfurt-Hoechst, Germany
2Sana Klinikum Offenbach GmbH, Department of Gynecology and Obstetrics, Offenbach, Germany
3Paracelsus Medical University, Department of Gynecology, Nuremberg, Germany
4Klinikum Hanau, Department of Gynecology and Obstetrics, Hanau, Germany
5Sana Klinikum Offenbach GmbH, Institute of Pathology, Offenbach, Germany
6OncoNet Rhein-Main e. V.
#Contributed equally
Received Date: 15/07/2025; Published Date: 18/08/2025
*Corresponding author: Anastasia Chaviaropoulou, Department of Obstetrics and Gynecology Varisano Klinikum Hoechst GmbH Gotenstraße 6-8, 65929 Frankfurt Germany
Abstract
Perivascular Epithelioid Tumors (PEComas) constitute a rare subgroup of mesenchymal tumors originating from perivascular epithelioid cells. The spectrum of PEComas encompasses various types, including angiomyolipomas, lymphangiomas, clear cell "sugar" tumors of the lung, and PEComa not otherwise specified. The determination of malignancy potential relies heavily on immunohistological characteristics rather than the tumor's origin, exhibiting a broad range of reported malignancy grades. Gynecological tract PEComas primarily manifest in sites such as the uterus, cervix, vagina, adnexa, broad ligament and vulva, with the uterus emerging as the most frequently reported location. This report presents two cases of uterine PEComas, highlighting the histopathological classification of this rare tumors. In both cases the patients were referred to our center and underwent primary surgery under the initial suspicion of symptomatic uterine fibroids, based on the clinical examination and imaging. Histopathological evaluation of the surgical specimens revealed the presence of PEComas, and further analysis classified them as benign in the first and malignant in the second case. Distinguishing among mesenchymal neoplasms, including PEComas, fibroids, endometrial stromal sarcomas, and leiomyosarcomas, poses diagnostic challenges. Therefore, meticulous examination of morphological and immunohistochemical features assumes paramount importance in accurate diagnosis and subsequent management.
Keywords: PEComa; Perivascular; Epithelioid; Mesenchymal; Rare tumor
Introduction
Perivascular epithelioid cell tumors (PEComas) are a rare group of mesenchymal tumors that show proliferation of perivascular epithelioid cells, the nature of which remains a matter of controversy. Degeneration of perivascular structures raise questions, whether these tumors derive from vessel walls, or a separate population of muscle cells [1]. These tumors are composed of epithelioid cells with clear, granular, or eosinophilic cytoplasm, which are arranged in nests, or sheets with little intervening connective tissue and a low mitotic rate. They present variable immunoreactivity with melanocytic and myogenic antigens and confirmatory positivity is noted for smooth muscle antigens like SMA, Desmin, Caldesmon and for the melanocytic antigens HMB 45 and Melan A [2].
The first publication on PEComa goes back to 1995 and according to the data available in the English language, fewer than 100 cases have been reported until 2022, which emphasizes the scarcity of knowledge about this entertainment and the challenges encountered during its diagnosis and therapeutic approaches. Uterine involvement is described in about one-third of the reported cases which places the uterus the second most common involved site after the peritoneum [3].
In the past, the diagnosis of PEComas has been difficult mainly if presenting as heterogeneous usually well circumscribed lesions on MRI resembling leiomyomas, leiomyosarcomas or sarcomas. Symptoms that bring patients to the clinic are usually tummy ache and/or spotting. Histopathological examination can confirm the diagnosis only when associated with surgical excision. The ability to recognize these tumors before surgery has remained difficult because of their non-specific imaging features, making it challenging to design optimal surgical approaches and treatments.
Although the knowledge of these neoplasms increases, common guidelines for diagnosis and treatment remain a concern, due to the low incidence of uterine PEComas. To the best of our knowledge, the current cases add to the existing literature by describing two cases of uterine PEComa with different clinicopathological features with histopathological and immunohistochemical analysis considered crucial in diagnosis and management of the disease. Moreover, this report underlines the need for enhanced clinicopathological recognition of this rare tumor entity to establish the diagnosis of uterine mass.
Case Presentation
Case 1
The first case involves a 32-year-old nulligravida, virgo intacta, presenting with abdominal pain and bloating. Laboratory analysis revealed asymptomatic anemia, while her medical history was non- contributory aside from a familial history of breast cancer in her mother post-50 years of age. Clinical examination revealed an enlarged uterus, protruding above the umbilicus, with resistance noted upon speculum adjustment and leftward displacement of the cervix. Transabdominal sonography demonstrated multiple fibroids predominantly on the anterior uterine wall, the largest measuring >10 cm with central necrosis at the fundal region.
Further imaging via MRI delineated a 16 x 10 x 13 cm enlarged uterus with a 12 cm lesion displaying signal intensity akin to myometrial tissue, consistent with a uterine leiomyoma. Two additional smaller leiomyomas were identified in the lower uterine segment, with no evidence of infiltrative growth. The larger leiomyoma exhibited signs of necrosis. No pathological lymphadenopathy or adjacent organ infiltration was observed.
Based on these findings, a diagnosis of uterine leiomyomatosis was established, subsequently managed operatively due to the patient's age and desire for future fertility. Laparotomy was performed for myomectomy without intra- or postoperative complications. Tissue specimens were sent for histopathological examination.
The initial histopathological examination revealed a 685g, 12cm-long leiomyoma with an accompanying 0.5 cm epithelial tumor detected at the periphery. Further immunohistochemical analysis demonstrated positivity for HMB-45, cytokeratin, AE 1/AE 3, calretinin, and weak positivity for smooth muscle actin (SMA), while testing negative for cytokeratin 5, D2-40, CD34, Melan A, and S100. The proliferative index (Ki-67) within the tumor tissue was determined to be up to 5%, with weak, inconclusive positivity for PAX8. These findings confirmed the diagnosis of a subserosal, hyalinized epithelioid tumor consistent with a poorly sclerosed PEComa. The excision was performed with a minimum safety distance to the serosa of 0.2mm, ensuring complete removal of the tumor. Given the tumor's size (< 5 cm), absence of nuclear atypia, and low proliferation rate, the PEComa was classified as benign in nature. The patient was discharged on the seventh postoperative day without requiring further systemic therapy. Contraceptive measures were advised for a minimum duration of one year.
Case 2
The second case pertains to a 54-year-old obese woman with a history of hypertension and two pregnancies, who presented at our clinic with an initially unclarified, asymptomatic, sonographic suspicious cystic mass measuring 6.6 x 6.4 cm in the left lower abdomen. The patient was referred to our center following a diagnostic laparoscopy at another clinic, which was prematurely terminated due to significant adhesions between the mass and the left pelvic wall, likely attributed to prior surgeries including emergency cesarean section and conventional appendectomy, rendering the findings challenging to interpret. Preoperative tumor markers (CEA, CA 125 and HE 4) were within normal limits. The preoperative MRI showed a cystic lesion at the right pelvic wall measuring approximately 3.1 cm. The differential diagnosis included ovarian tumor or cystic lymph node and there were no evidence of suspicious tumor changes. Τhe patient underwent primary laparoscopic intervention. Intraoperative assessment revealed a 7 cm mass in the left lower abdominal wall with evident vascularization, adhering to the left uterus, pelvic wall, and sigmoid colon, suggestive of probable uterine origin. Initially suspected as a uterine leiomyoma, an unsuccessful attempt at laparoscopic excision was made due to profuse bleeding and concerning findings, necessitating conversion to conventional laparotomy. Despite extensive adhesiolysis, the suspicious mass remained unresectable. Rapid pathological evaluation revealed atypical cells, warranting consideration of malignancy, prompting the decision to proceed with total hysterectomy and bilateral adnexectomy. The patient was discharged without complications on the fifth postoperative day.
The histological examination revealed a 7.5 cm diameter mesenchymal tumor located in the left parametrial region, displaying extensive superficial capsular defects infiltrating the left pelvic wall. A subsequent pathological analysis identified a high-grade TFE3-associated PEComa. Immunohistochemical analysis of the specimen demonstrated subserosal tumor positivity for HMB-45, cytokeratin, AE 1/AE 3, and calretinin, with weak positivity for smooth muscle actin (SMA). Conversely, the tumor tested negative for Cytokeratin 5, D2-40, CD34, Melan A, and S100. The proliferation rate (Ki-67) within the tumor tissue was observed to be up to 5%, with weak, inconclusive positivity for PAX8.
The uterine PEComa exhibited a high-grade morphological presentation characterized by the absence of infiltrative growth patterns. Assessment revealed a mitotic rate of 3 per 50 high-power fields (HPF), devoid of necrotic areas and vascular infiltration. These observations collectively fulfill three malignancy criteria [2], thereby warranting classification as malignant according to both overarching and gynecologically tailored diagnostic standards.
Following the surgical intervention, postoperative bone scintigraphy was conducted to evaluate potential osseous metastasis, yielding negative findings. Subsequently, hormone receptor determination was undertaken to establish an additional therapeutic avenue in the event of disease recurrence. Results indicated a moderate degree of positivity for both estrogen receptor and progesterone receptor. After presentation at our interdisciplinary tumor conference, a consensus decision was reached to continue with intensive follow-up care without any form of adjuvant treatment.
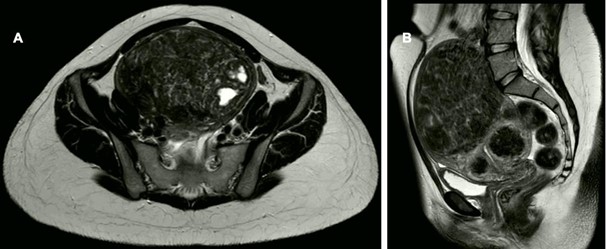
Figure 1: Radiologic findings in the 1st case. Large uterine fibroid (uterus myomatosus) without signs of infiltration. No pathological lymph nodes. Bilateral cystic ovaries.
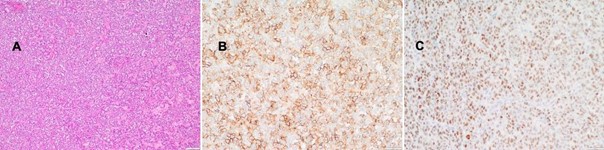
Figure 2: Histopathologic finding in the 1st case A Diffuse proliferation of epitheloid cells with clear cytoplasm and hyaline stroma; HE staining, magnification x40. B The tumor cells show heterogenous (membranous) staining with anti-CD56; magnification x200. C A large fraction of the PEComa cells are positive for anti-estrogen receptor (nuclear staining); note the mild nuclear atypia; magnification x200.
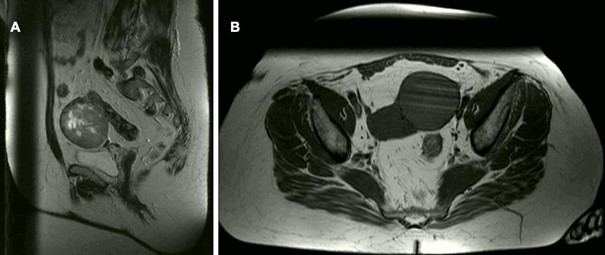
Figure 3: Radiologic findings in the 2nd case. Cystic lesion at the right pelvic wall measuring approximately 3.1 cm, differential diagnosis includes ovarian tumor or cystic lymph node. Follow-up recommended. Otherwise, no evidence of suspicious tumor changes.
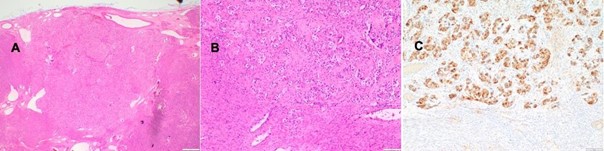
Figure 4: Histopathologic findings in the 2nd case A Overview with numerous large PEComa cells displaying clear cytoplasm; HE staining, magnification x20. B Nests of PEComa cells within a fibroblast-rich and hyaline stroma; HE staining, magnification x100. C As in this case, PEComa cells typically show diffuse and strong positivity for anti-HMB45; magnification x100.
Discussion
Uterine Perivascular Epithelioid Cell Tumors (PEComas) represent a distinctive yet infrequent pathological entity, posing ongoing diagnostic and therapeutic challenges for medical practitioners. Predominantly, these cases have been identified post-hysterectomy, initially presumed benign. Preoperative radiological discernment proves arduous owing to the absence of defining PEComa- specific radiographic markers, thereby complicating differentiation from benign uterine leiomyomas or malignant sarcomas, compounded by the rarity of such occurrences.
Clinically, uterine PEComas often mimic the presentation of benign leiomyomas, with patients commonly presenting with vaginal bleeding, nonspecific abdominal discomfort, or, less frequently, palpable abdominal masses, occasionally remaining asymptomatic. Radiologically, these tumors typically exhibit characteristics akin to other benign uterine lesions. Ultrasonography typically reveals solid masses with heterogeneous echotexture devoid of cystic regions or notable vascularity on Doppler examination.
A retrospective study conducted by Tan et al. [4] scrutinized 32 PEComa cases post-pathological diagnosis, correlating findings with preoperative CT or MRI scans. Dynamic imaging modalities revealed tumors predominantly of low density or hypointensity on T1-weighted imaging, contrasting with hyperintensity on T2, with some presenting isodensity with fat tissue. Typically, tumors manifested well-defined margins and exhibited uniform shapes, ranging in diameter from 1.5 to 18 cm. Most notably, these lesions demonstrated heterogeneous and significant enhancement during arterial and venous phases, often appearing slightly hyperdense on delayed CT imaging, albeit with some instances (6 out of 32 cases) showcasing delayed enhancement. Despite the distinct imaging characteristics elucidated in the literature, distinguishing PEComas from other uterine neoplasms, both benign and malignant, remains a formidable task, exacerbated by the rarity of these tumors, further complicating preoperative diagnostic endeavors.
Controversy persists in academic literature about the necessity of histological verification of uterine PEComas prior to surgery. Although successfully attempting to distinguish these neoplasms from other pathologic entities like leiomyosarcomas by using preoperative biopsies and immunohistochemical stainings, the diagnostic difficulties remain. In addition, the occurrence of uterine PEComas is relatively rare; thus, the cost-effectiveness concerns regarding routine biopsies in uterine processes should be considered. Therefore, an assessment of the diagnostic approach regarding both diagnostic performance and applicability is needed for the clinical care of such sparse malignancies.
One of the major obstacles in the diagnosis of uterine PEComas involves determination of their malignant potential. Different scoring models have been used to categorize the features of these tumors according to their class. Folpe et al. described a scoring system of benign, UMP, and malignant nuclear atypia which are deemed worrisome histologically. These defined features include tumor diameter/size ≥ 5cm, infiltrating border, high nuclear grade cellularity, mitosis rate per 50 HPF and necrosis or vascular invasion [2].
Uterine PEComas are typically sharply delimited lesions on imaging that might represent other benign or malignant lesions of the uterus including leiomyomas or sarcomas. Anderson et al. [5] opined that despite MRI and CT imaging, no imaging characteristic is unique to PEComa. As with most tumors, histopathological examination is the gold standard investigation demonstrating epithelioid cells with a clear to eosinophilic cytoplasm and a characteristic immunophenotype that comprises HMB-45 and SMA [6]. Nevertheless, according to Dubé et al. [7], various marker patterns may result in false diagnoses and, therefore, it becomes necessary to perform multiple tests as well as multiple biopsies whenever possible in complicated cases.
PEComas can be clinically indolent and have localized manifestations of Sturge-Weber syndrome or are aggressively malignant with metastatic capabilities. Historical osteology risk factors, including those reported by Folpe et al. [2], stem from test elements including tumor size, MIB-1, and necrosis. However, the existing data indicate that these criteria do not fully address the heterogeneity of PEComas. For example, Sanfilippo et al. [8] showed that there were cases with low risk factors and metastases in some patients hence underlining some of the existing models’ drawbacks.
Newer studies are trying to improve the methods of risk assessment. Recent data suggest that molecular markers including TSC1/TSC2 mutations should be included in prognostic models because of their correlation with the activation of the mTOR pathway. Lyle et al. [9] observed that TFE3-altered PEComas may be a molecularly different subtype regarding its prognosis. Subsequently, there is a call to continue similar research so as to confirm the conclusions made above and apply molecular analytics into formal clinical practice.
Schoolmeester et al. [10], using the Folpe criteria, applied the scoring system to 16 gynecological tract PEComas and suggested a new scoring system with a threshold of four or more features suggestive of malignancy. These changes allowed an increased discrimination in terms of positive and negative predictive values for likelihood of subsequent malignant behavior but caused no change in the overall sensitivity of the tests. On the other hand, Bleeker et al. [11] recommended a more stringent risk stratification system by retaining size ≥ 5 cm as well as high mitotic activity, because these were the only two histopathological characteristics that in their opinion, were predictive of recurrence.
A retrospective analysis compared the performance of the original Folpe criteria with a modified version it, in which a tumor with a single ‘suspicious’ feature such as a maximum of size more than 5 cm, infiltrative borders, lymphovascular invasion or mitotic rate exceeding 1/50 HPF would be classified as benign. Indeed, the modified Folpe criteria exhibited higher sensitivities and negative predictive values than the Schoolmeester criteria. Nonetheless, the tumors defined as malignant by the strict criteria of the Schoolmeester classification were found to have a tendency for early recurrence. Based on these observations, until more extensive cohorts are available, the modified Folpe criteria should be first used for tumor classification [1].
In our above-mentioned case of malignant PEComa the tumor was described as TFE3-altered. In particular, during the last several years, about twenty cases of TFE3 translocation related PEComas have been described in various anatomic locations including renal, bladder, colonic, pelvic soft tissue, ovarian, vaginal, and uterine PEComas. Notably, activation of the mTOR pathway may not always be a contributory factor in these specific tumors. This observation has major relevance for the patient’s suitability for clinical trials of mTOR pathway inhibitors [1].
In cases like the current one where a more aggressive biology is expected, particularly when systemic treatment is required, mutation analysis for prospective mTOR changes is warranted. In general, such broad molecular profiling not only helps to individualize treatment and enhance the outcome of therapy of TFE3-altered PEComas, but also can be useful for successful preliminary diagnostic tests.
The treatment of uterine PEComas also continues to be a therapeutic challenge in both approaches. Lack of sufficient cases makes it still challenging to develop a clear definition of an acceptable standard of care in patients with gynecological PEComas. Currently, surgical resection remains the sole option for the management of this disorder. In any case, it is predominantly performed as a total hysterectomy, with or without BSO, which has larger support in practice. For cases of PEComas that show spread or localization in the uterine cervix or malignant potential, radical hysterectomy with bilateral salpingo- oophorectomy should be considered [12]. This additional surgery is expected to help remove more suspicious tissue or tumor volume because of higher disease prevalence which may lead to local recurrence or metastasis. However, the best therapeutic strategy for managing uterine PEComas remains a question mark that calls for interdisciplinary cooperation and the enhancement of existing therapeutic strategies. Transabdominal hysterectomy is by far the most often adopted procedure. Some surgical techniques, including myomectomy, have been described in this setting; however, Fang et al. [13] suggested that fertility-sparing approaches should be avoided if the tumor exhibits high-risk criteria because of the possible recurrence.
mTOR inhibitors that include sirolimus and everolimus have been used for the treatment of PEComa with TSC mutations [8]. These targeted agents work on the fact that the existence of the tumor is dependent on the mTOR signaling pathway. However, the role of mTOR inhibitors in TFE3-altered PEComas is still questionable because such tumors may not be as dependent upon mTOR signaling as mTOR inhibitors were initially believed to be. In some patients with high-grade or metastatic lesions, Adriamycin, ifosfamide and carboplatin have been used in different schedules, and efficacy varies widely [14].
Radiotherapy has also been used for local control in malignancies that are unsuitable for resection, although the utility is questionable. Similarly, Seidman et al. stated that the extent of radio sensitivity in PEComas is not absolute that is why the use of that modality should be patient specific depending on the features of the tumor and the preferences of the patient [6].
Progress in the field of molecular pathology increased our knowledge about the PEComas. Indeed, the identification of TSC1/TSC2 gene mutation and TFE3 rearrangement not only improves diagnostic accuracy but also provides therapeutic targets [8]. However, due to the low incidence of uterine PEComas, the applicability of these data is questionable, which underlines the imperativeness of cooperation.
Wang et al. [14] indicated that further registries should be encouraged to focus on the clinical, pathological, and molecular profile of PEComas. They could help create guidelines that draw from research results and advance clinical investigations concerning new therapeutic instruments.
Conclusion
In summary, we describe our two cases of uterine PEComa showing different clinicopathological features and different biological behaviors. Histopathological examination without immunohistochemical testing yielded correct diagnosis only after hysterectomy, after which the examined tissue was positive for both smooth muscle and melanocytic markers after immunohistochemistry testing. It is therefore important that a battery of morphological techniques and MS-IHC analysis be conducted in the determination of PEComas from other mesenchymal tumors. A positive preoperative diagnosis of such a tumor, however, is still considered to be out of reach in most cases. In addition, there is a need to detail out therapeutic approaches and protocols faithfully. Based on our experience with the present cases, PEComa should be considered in the differential diagnosis of uterine mass, even though it is rare. Thus, there is a need to increase the clinicians’ index of suspicion for PEComa in the differential diagnosis of uterine neoplasms.
Declarations: The authors declare no competing interests.
References
- Conlon N, Soslow RA, Murali R. Perivascular epithelioid tumours (PEComas) of the gynaecological tract. J Clin Pathol, 2015; 68(6): 418-426. doi: 1136/jclinpath-2015-202945.
- Folpe AL, Mentzel T, Lehr HA, Fisher C, Balzer BL, Weiss SW. Perivascular epithelioid cell neoplasms of soft tissue and gynecologic origin: a clinicopathologic study of 26 cases and review of the literature. Am J Surg Pathol, 2005; 29: 1558–1575.
- Padmehr R, Arasteh S, Aminimoghaddam S, Rahbari A, Bohloli M, Mir Eskandari SM, et al. PEComa-A Rare Uterine Neoplasm: A Case Report. J Reprod Infertil, 2022; 23(3): 224-227. doi: 10.18502/jri.v23i3.10015.
- Tan Y, Zhang H, Xiao E-H. Perivascular epithelioid cell tumour: Dynamic CT, MRI and clinicopathological characteristics - Analysis of 32 cases and review of the literature. Clinical Radiology, 2013; 68(6): Pages 555-561. https://doi.org/10.1016/j.crad.2012.10.021.
- Anderson ME, Clemens SM, Gursel B. Imaging features of uterine PEComas: Correlation with pathological findings in 12 cases. European Radiology, 2023; 33(4): 2508-2517. https://doi.org/10.1007/s00330-023-09762-1.
- Seidman JD, Hornick JL, Nucci MR. PEComas of the female genital tract: A clinicopathological study of 27 cases with emphasis on diagnostic Modern Pathology, 2020; 33(8): 1570-1582. https://doi.org/10.1038/s41379-020-0502-z.
- Dubé V, Mahadevan D, Khanna The role of imaging and immunohistochemistry in the diagnosis of rare uterine neoplasms. Clinical Radiology, 2019; 74(7): 544-550. https://doi.org/10.1016/j.crad.2019.04.005.
- Sanfilippo R, Bertulli R, Marrari A, Casali PG. Tuberous sclerosis complex mutation-associated PEComas: Advances in mTOR-targeted therapies. Cancer Treatment Reviews, 2016; 50(1): 1-9. https://doi.org/10.1016/j.ctrv.2016.07.002.
- Lyle MR, Rivera LB, Padera TP. Histopathologic and molecular insights into uterine American Journal of Pathology, 2017;187(4): 809-817. https://doi.org/10.1016/j.ajpath.2017.01.014
- Schoolmeester JK, Howitt BE, Hirsch MS, et al. Perivascular epithelioid cell neoplasm (PEComa) of the gynecologic tract: clinicopathologic and immunohistochemical characterization of 16 cases. Am J Surg Pathol, 2014; 38: 176-188.
- Bleeker JS, Quevedo JF, Folpe AL. “Malignant” perivascular epithelioid cell neoplasm: risk stratification and treatment Sarcoma, 2012; 2012: 541626.
- Kudela E, Biringer K, Kasajova P, Nachajova M, Adamkov M. Perivascular epithelioid cell tumors of the uterine cervix. Pathol Res Pract, 2016; 212(8): 667–671.
- Fang SH, Zhou SH, Wang X. Five cases of uterine perivascular epithelioid cell tumors (PEComas) and review of the literature. Archives of Gynecology and Obstetrics, 2018; 298(5):
1025- 1033. https://doi.org/10.1007/s00404-018-4853-6. - Wang J, Wang D, Zhang H. Prognostic factors and treatment outcomes in uterine PEComas: A review of the literature. Journal of Obstetrics and Gynaecology Research, 2021; 47(9): 3243- 3251. https://doi.org/10.1111/jog.14915.

