Biventricular Thrombi in Juvenile Lupus Cardiomyopathy: An Exceptional Association
Zaynab Laoufi*, Rokya Fellat and Nadia Fellat
Cardiology A department, Ibn Sina university hospital, Faculty of Medicine and Pharmacy, Mohammed V University, Rabat, Morocco
Received Date: 24/06/2025; Published Date: 24/07/2025
*Corresponding author: Zaynab Laoufi, Cardiology A department, Ibn Sina university hospital, Faculty of Medicine and Pharmacy, Mohammed V University, Rabat, Morocco
Abstract
Biventricular thrombi are uncommon in cases of Systemic Lupus Erythematosus (SLE) and represent one of the most severe cardiac manifestations. They result from lupus myocarditis, that’s more severe in juvenile cases. We present the clinical case of a 19-year-old female treated for juvenile SLE since the age of six, complicated by lupus nephropathy. The patient was admitted due to severe dyspnea. Thoracic computed tomography angiography revealed no signs of pulmonary embolism but identified three masses in both the left and right ventricle. Echocardiography, showed non dilated left ventricle with global hypokinesia, severe systolic dysfunction and Left Ventricular Ejection Fraction (LVEF) at 38 %. Two thrombi were identified at the apex of the left ventricle, and an additional one detected at the lateral wall of the right ventricle. Anticoagulant therapy was promptly initiated.
Introduction
Lupus cardiomyopathy is an uncommon but serious manifestation of Systemic Lupus Erythematosus (SLE), often associated with significant morbidity and mortality. Its clinical presentation can vary widely, ranging from asymptomatic ventricular dysfunction to severe heart failure. Among its rare complications is the formation of intracardiac thrombi, which significantly worsens the prognosis by increasing the risk of embolic events and hemodynamic compromise. In juvenile forms, cardiac involvement often occurs at an early stage of the disease and is typically more aggressive, requiring prompt recognition and intensive immunosuppressive and supportive therapies. The occurrence of biventricular thrombi in this context is extremely rare, with only a few cases reported in the literature, making it a challenging diagnostic and therapeutic entity.
Clinical Case
A 19-year-old female patient, treated for Juvenile Lupus complicated of Lupus nephritis class IV , was admitted with fever and shortness of breath. Clinical examination revealed an orthopneic patient with lower limb edema and bilateral crackles at lung bases. Electrocardiogram showed negative T waves in the septo-apical and inferior leads. Laboratory findings revealed impaired renal function, normochromic normocytic anemia, and normal coagulation parameters with low titer of antiphospholipid antibodies. Thoracic computed tomography angiography ruled out pulmonary embolism and detected three well-defined masses regular contours (Figure 1).
A cardiac ultrasound showed reduced contractility of the left ventricle, with left ventricular ejection fraction (LVEF) of 38% , global longitudinal strain (GLS) of -12% (Figure 2), along with significant spontaneous contrast and dilated left atrium. Additionally, two masses were identified at the apex of the left ventricle (LV) (Figure 3), and a third, larger mass was attached to the lateral wall of the right ventricle (RV) (Figure 4). The appearance of these masses was highly suggestive of thrombus, thus anticoagulation treatment was initiated.
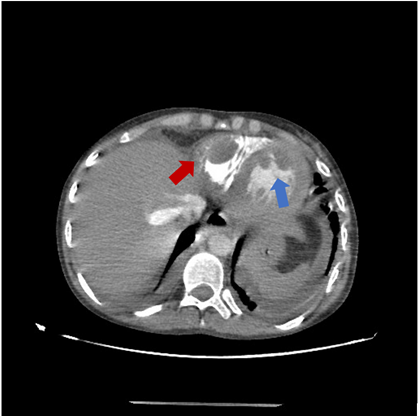
Figure 1: Thoracic angiographic scan showing 2 masses at the apex of the left ventricle (blue arrow) and a third one at the lateral wall of the right ventricle (red arrow).
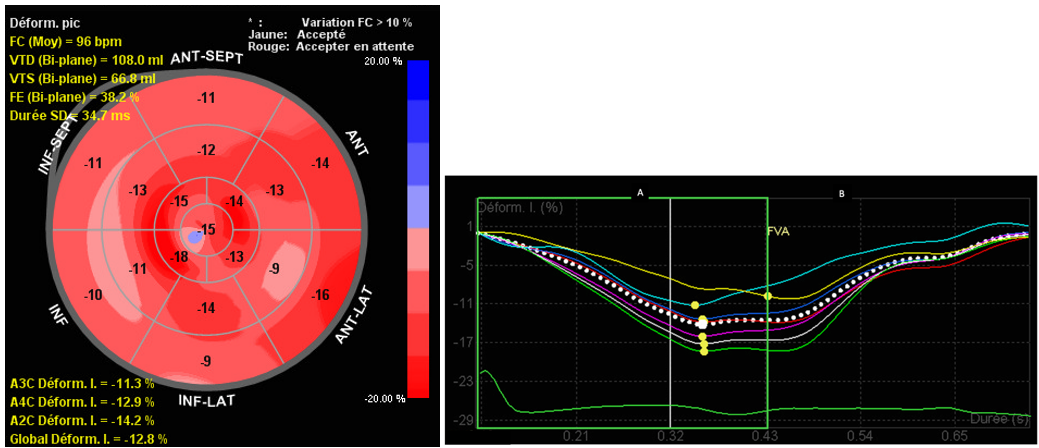
Figure 2: Altered GLS calculated from the apical long axis, four-chamber and two-chamber view.
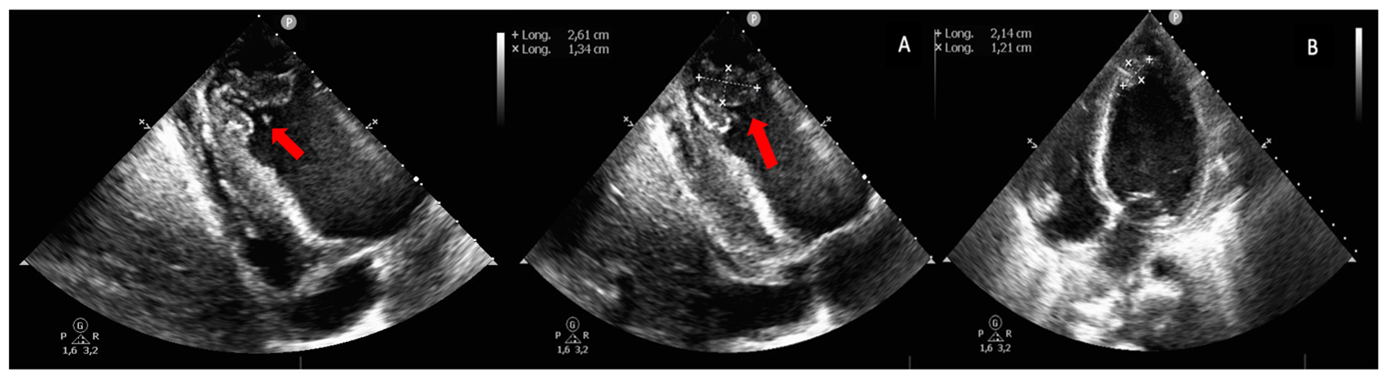
Figure 3: TTE views showing two thrombi at the apex of the left ventricle (red arrow).
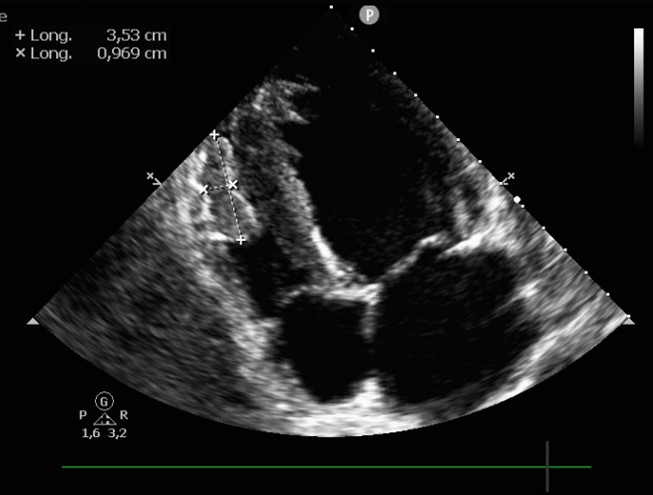
Figure 4: Four-chamber view showing a thrombus at the lateral wall of the right ventricle.
Discussion
Biventricular thrombi in cases of SLE are exceptional [1] ; in fact, only one case has been reported to date, and no cases have been described with the juvenile form. Patients with SLE present a higher thrombotic risk than the general population [2] .
Thrombotic manifestations can result from early atherosclerosis or from the severity of lupus activity. In this context, a Canadian prospective study showed [3] that patients with severe lupus activity were at a high risk of thrombotic events. This could be one of the mechanisms responsible for thrombosis in our patient, as she presents with juvenile SLE, which is known to be a more severe form, and also she presents with severe nephropathy, which is a sign of high lupus activity.
The thrombotic risk is even higher when SLE is associated with antiphospholipid syndrome [4] , but in the case of the juvenile form, this association is less common. Since our patient presented with a low titer of antiphospholipid antibodies, the implications of these are eliminated.
Another possible mechanism of thrombosis in our patient is myocarditis. Myocardial involvement in adult SLE is rare (9% of patients); it can range from a fatal fulminant form to a long-lasting asymptomatic form [5]. In young patients, this involvement is more common and severe. In cases of severe lupus activity, myocardial damage occurs at an early stage of the disease and is usually associated with lupus nephritis [6]. Some of the main factors implicated are coronary atherosclerosis, hypertensive cardiomyopathy, and side effects of specific treatments[7]. Diagnosis is based on clinical examination, biochemical markers, cardiac imaging, and ultimately endomyocardial biopsy. In our patient, echocardiography confirmed myocardial involvement with altered GLS and reduced LVEF. Additionally, globally non-systematized hypokinesis of the left ventricle was highly suggestive of lupus myocarditis.
The reduction in LVEF represents a risk factor for LV and RV thrombosis, as kinetic abnormalities lead to stasis and ultimately to the formation of intracavitary thrombi [8] . LV thrombosis is associated with high mortality, with a risk of systemic embolism. RV thrombosis is usually associated with pulmonary embolism and can result from deep vein thrombosis. The occurrence of both LV and RV thrombosis is rare due to their distinct mechanisms, although heart failure can be a common mechanism [9].
In our patient, the diagnosis of thrombus was made using echocardiography. Cardiac Magnetic Resonance (CMR) could have been used to confirm the thrombotic nature of the masses described, but the appearance of the masses was highly suggestive of the diagnosis, in addition to the significant spontaneous intracavitary contrast, representing a prethrombotic state. However, CMR would have been useful for better analysis of myocardial involvement.
In cases of lupus cardiomyopathy, steroids and heart failure treatment can significantly improve the LVEF [10]. Our patient received optimal anticoagulation therapy for her thrombi, in addition to heart failure conventional treatment. Specific lupus treatment was also initiated to improve her kidney function and, subsequently, her cardiac function.
Conclusion
Although patients with systemic lupus erythematosus (SLE) are known to have an increased thrombotic risk due to factors such as antiphospholipid antibodies and systemic inflammation, the occurrence of biventricular thrombi remains rare. When present, these thrombi are often associated with underlying lupus cardiomyopathy. Early recognition and comprehensive management are crucial, as these intracardiac thrombi may resolve with optimal anticoagulation, immunosuppressive therapy, and heart failure treatment. In cases of juvenile SLE with cardiac involvement, a multidisciplinary approach is crucial to improving cardiac function and overall prognosis.
References
- Li Yanmei, Wei Wei. Thrombi in Both Ventricles in a Systemic Lupus Erythematosus Patient. JCR: Journal of Clinical Rheumatology, 2022; 28(5). DOI: 10.1097/RHU.0000000000001867.
- Mok CC. Accelerated atherosclerosis, arterial thromboembolism, and preventive strategies in systemic lupus erythematosus. Scand J Rheumatol, 2006; p. 35: 85-95.
- Chang ER, Pineau CA, Bernatsky S, et al. Risk for incident arterial or venous vascular events varies over the course of systemic lupus erythematosus, 2006.
- Brouwer JL, Bijl M, Veeger NJ, Kluin-Nelemans HC, van der Meer et J. The contribution of inherited and acquired thrombophilic defects, alone or combined with antiphospholipid antibodies, to venous and arterial thromboembolism in patients with systemic lupus erythematosus. Blood, 2004; 104: 143-148.
- Moder KG, Miller TD, Tazelaar HD. Cardiac involvement in systemic lupus erythematosus. Mayo Clin. Proc., 1999; 74(no3): p. 275‑284. doi: 10.4065/74.3.275.
- Du Toit R, Herbst PG, van Rensburg A, et al. Clinical features and outcome of lupus myocarditis in the Western Cape, South Africa. Lupus, 2017; 26: 38–47.
- Martin A Shearn. The heart in systemic lupus erythematosus, American Heart Journal, 1959; 58(3): 1959.
- McCarthy C, Murphy S, Venkateswaran RV, et al. Left ventricular thrombus: contemporary etiologies, treatment strategies, and outcomes. J Am Coll Cardiol, 2019; 73: 2007–2009.
- Liu X, Lei S, Huang H, Wu Y, Hu Z-X. Resolution of huge thrombi in bilateral ventricles caused by severe lupus cardiomyopathy. Lupus, 2021; 30(5): 840-844. doi:10.1177/0961203321989808.
- Gottenberg JE, Roux S, Assayag P, Clerc D, Mariette X. Specific cardiomyopathy in lupus patients: report of three cases. Joint bone spine, 2004; 71(1): 66–69. https://doi.org/10.1016/S1297-319X(03)00101-5.

