IgG4 Related Pancreatitis with Concomitant Renal Involvement
Murad Jamal*, Muhammad Mudasir, Arsalan Pervaiz, Khadeeja Anwar and Muhammed Aasim Yususf
Shaukat Khanum memorial cancer hospital & research center (SKMCH&RC), Pakistan
Received Date: 02/06/2025; Published Date: 14/07/2025
*Corresponding author: Dr. Murad Jamal, Shaukat Khanum memorial cancer hospital & research center (SKMCH&RC), Pakistan
Abstract
Background: Immunoglobulin G4-related disease (IgG4-RD) is a systemic immune-mediated condition characterized by the infiltration of IgG4-positive plasma cells, often leading to fibrosis and organ dysfunction. While pancreatic involvement is well established, renal manifestations—collectively termed IgG4-related kidney disease (IgG4-RKD)—are less frequently reported but carry significant clinical implications.
Case Presentation: We report a case of a 50-year-old male with type 1 autoimmune pancreatitis who developed IgG4-RKD. The patient initially presented with obstructive jaundice and was found to have a pancreatic head mass, which resolved completely following corticosteroid therapy. Subsequent relapse upon steroid tapering was associated with liver and renal lesions. Elevated serum IgG4 levels and characteristic radiologic findings confirmed IgG4-RKD. The patient responded well to reintroduction of corticosteroids, with full radiologic resolution of renal lesions and clinical stabilization on maintenance therapy.
Conclusion: This case highlights the diagnostic and therapeutic challenges of IgG4-RD with multi-organ involvement, emphasizing the need for ongoing monitoring and individualized management strategies. Early identification of renal involvement and prompt treatment can prevent irreversible damage and improve long-term outcomes.
Keywords: IgG4-related disease (IgG4-RD); IgG4-related kidney disease (IgG4-RKD); Autoimmune pancreatitis; Tubulointerstitial nephritis; Corticosteroid therapy; Multi-organ involvement; Relapse; Immunoglobulin G4; Renal hypodensities; Immune-mediated disorders
Introduction
Immunoglobulin G4-related disease (IgG4-RD) is an immune-mediated disorder characterized by the infiltration of IgG4-producing plasma cells, leading to fibrosis in affected organs [1-2]. Initially recognized in the early 2000s, it was first identified in the pancreas as lymphoplasmacytic sclerosing pancreatitis, also known as Type 1 autoimmune pancreatitis [3]. IgG4-RD can affect multiple organs, including the endocrine, nervous, gastrointestinal, genitourinary, and vascular systems [4-6]. Commonly, the condition presents with a gradual development of a mass or diffuse organ enlargement.
Association between Type 1 autoimmune pancreatitis and renal dysfunction is well established [7]. The kidneys are frequently involved in IgG4-RD, with tubulointerstitial nephritis (TIN) being a common renal manifestation [8-9]. Although glomerular involvement is less frequent, membranous glomerulonephritis is the primary glomerular issue linked to the disease. Additionally, conditions like retroperitoneal fibrosis can secondarily affect the kidneys. The broad spectrum of renal manifestations has led to the term IgG4-related kidney disease (IgG4-RKD) [10]. In 2011, Kawano et al. proposed the first diagnostic criteria for IgG4-RKD, which were later updated by Saeki et al. in 2020 [10-12]. The diagnosis typically requires renal biopsy, alongside clinical, laboratory, and imaging findings.
Case Presentation
We report the case of a 50-year-old male who presented with left flank pain, jaundice, and significant weight loss over the past three months. His medical history included diabetes and a recent endoscopic retrograde cholangiopancreatography (ERCP) for a distal common bile duct (CBD) stricture with plastic stent placement. Physical examination was mostly unremarkable except for jaundice. Initial lab results showed normal complete blood count (CBC) and urea & electrolytes (U&E). Liver function tests (LFTs) were abnormal, with an obstructive pattern (ALT 121 U/L, AST 116 U/L, GGT 32 U/L, total bilirubin 6.26 mg/dL, alkaline phosphatase 285 U/L). CA19-9 was also elevated. A CT scan revealed a 34 mm x 28 mm mass in the head of the pancreas with a CBD stent in place. (Figure 1).
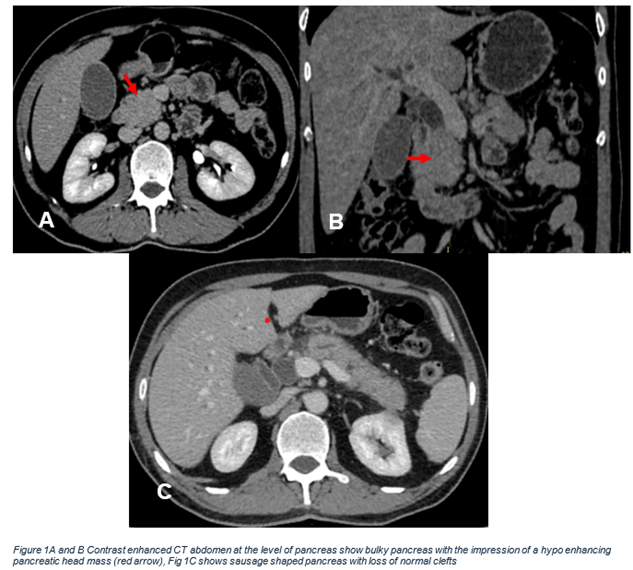
Figure 1A and B: Contrast enhanced CT abdomen at the level of pancreas show bulky pancreas with the impression of a hypo enhancing pancreatic head mass (red arrow),
Figure 1C: Shows sausage shaped pancreas with loss of normal clefts.
Endoscopic ultrasound (EUS) and fine needle aspiration (FNA) were performed, which showed benign ductal cells and some histiocytes. IgG4 levels were checked to rule out autoimmune pancreatitis, which was elevated at 502.6 mg/dL. The patient was started on prednisone 40 mg, and a follow-up CT scan after six weeks showed complete resolution of the pancreatic mass (Figure 2).
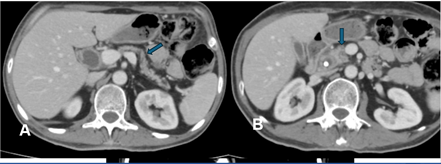
Figure 2: Follow up CT abdomen with contrast shows interval normal appearance of pancreas with clefts (blue arrow Figure 2A) and no mass at pancreatic head (blue arrow Figure 2B).
IgG4 levels decreased to 229 mg/dL, and the steroids were gradually tapered. However, the patient developed right-sided hypochondriac pain after tapering, and an MRI revealed subtle hypodense lesions in the liver. These lesions resolved with further treatment. Steroids were then stopped after 1 year of treatment, but the patient later developed jaundice again. Follow-up labs showed worsening liver enzymes, and a repeat CT scan revealed new kidney abnormalities, in the form of multiple hypodensities in both kidneys. Which are typical radiological findings for IgG4 related kidney disease. Which led to the diagnosis of IgG4-related kidney disease (Figure 3).
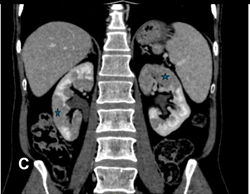
Figure 3: Shows interval appearance of multiple hypodense renal lesions (blue stars).
The patient was restarted on steroids at 20 mg daily and was kept on regular follow up. After 3 months of steroids repeat scan was done which shows complete resolution of renal hypodensities. Patient is now on maintenance dose of steroids and is doing well.
Discussion
IgG4-RD is a systemic autoimmune disorder that can affect multiple organs, including the pancreas, liver, and kidneys. The most common renal manifestation is tubulointerstitial nephritis. [8-9]. Although less frequent, membranous glomerulonephropathy can also occur. Characteristic biopsy findings include infiltration by IgG4-positive mononuclear cells and storiform fibrosis [2]. A study from Sweden showed that approximately 27% of patients with autoimmune pancreatitis also had IgG4-RKD, with a male predominance [13]. This suggests a strong association between pancreatic and renal manifestations of IgG4-RD, with some patients developing both conditions simultaneously. The pathophysiology of this overlap may involve systemic immune dysregulation, leading to the infiltration of multiple organs by IgG4-positive plasma cells.
Corticosteroids are the first-line treatment for IgG4-RD, with most patients showing rapid and favorable responses [14-17]. However, relapses can occur in up to 30% of cases, and alternative treatments such as rituximab, mycophenolate, azathioprine, and bortezomib are being explored, although evidence is still limited [18-19]. A more tailored, individualized approach to treatment is essential, particularly for patients with comorbidities such as diabetes, who may be more vulnerable to the adverse effects of steroids. Ongoing clinical trials and studies will help define the optimal therapeutic strategies for managing IgG4-RD, especially in cases with significant renal involvement.
While glucocorticoids can effectively manage IgG4-RD, reports have indicated that despite intensive corticosteroid therapy, some patients with IgG4-RKD require maintenance hemodialysis or even renal transplantation (Tanaka et al., 2014) [20]. This underscores the importance of early diagnosis and timely treatment to prevent irreversible renal damage. The advent of steroid-sparing agents holds promise for reducing the need for renal replacement therapy in such cases. Early diagnosis and treatment are critical for improving renal outcomes. Maintenance therapies like azathioprine, mycophenolate, and rituximab may help reduce the need for steroids. Ongoing clinical trials and studies will help define the optimal therapeutic strategies for managing IgG4-RD, especially in cases with significant renal involvement.
Conclusion
Patients with one manifestation of IgG4 disease may develop other related conditions, including kidney involvement. Early recognition of renal manifestations in IgG4-RD is essential for prompt diagnosis and treatment, improving patient outcomes. Due to the potential side effects of high-dose steroids, further research into steroid-sparing therapies is crucial for more effective management of the disease.
References
- Bledsoe JR, Della-Torre E, Rovati L, Deshpande V. IgG4- related disease: review of the histopathologic features, differential diagnosis, and therapeutic approach. APMIS, 2018; 126: 459-476. doi:10.1111/apm.12845
- Stone JH, Zen Y, Deshpande V. “IgG4-related disease,” New England Journal of Medicine, 2012; 366(6): pp. 539–551.
- Hamano H, Kawa S, Horiuchi A, et al. “High serum IgG4 concentrations in patients with sclerosing pancreatitis,” New England Journal of Medicine, 2001; 344(10): pp. 732–738, 2001.
- Himi T, Takano K, Yamamoto M, Naishiro Y, Takahashi H. “A novel concept of Mikulicz’s disease as IgG4-related disease,” Auris Nasus Larynx, 2012; 39(1): pp. 9–17.
- Mikulicz J. ¨Uber eine Eigenartige Symmetrische Erkrankungder Tr¨anen und Mundspeicheldr¨usen, Beitr Chir Fortsch Gewidmet *eodor Billroth, Stuttgart, Germany, 1892.
- K¨uttner H. “¨Uber entz¨undliche tumoren der submaxill¨arspeicheldr ¨use,” Beitr Klin Chir, 1896; 15: pp. 815–834.
- Uchiyama-Tanaka Y, Mori Y, Kimura T, et al. “Acute tubulointerstitial nephritis associated with autoimmunerelated pancreatitis,” American Journal of Kidney Diseases, 2004; 43(3): pp. e13.1–e13.8.
- Saeki T, Nishi S, Imai N, et al. “Clinicopathological characteristics of patients with IgG4-related tubulointerstitial nephritis,” Kidney International, 2010; 78(10): pp. 1016–1023.
- Backhus J, Neumann C, Perkhofer L, et al. “A follow-up study of a European IgG4-related disease cohort treated with rituximab,” Journal of Clinical Medicine, 2021; 10(6): p. 1329.
- Saeki T, Imai N, Ito T, Yamazaki H, Nishi S. “Membranous nephropathy associated with IgG4-related systemic disease and without autoimmune pancreatitis,” Clinical Nephrology, 2009; 71(02): pp. 173–178.
- Watson SJ, Jenkins DA, Bellamy CO. “Nephropathy in IgG4-related system3w2wic disease,” American Journal of Surgical Pathology, 2006; 30(11): pp. 1472–1477.
- Kawano M, Saeki T, Nakashima H, et al. “Proposal fordiagnostic criteria for IgG4-related kidney disease,” Clinical and Experimental Nephrology, 2011; 15(5): pp. 615–626.
- Vujasinovic M, Mucelli RP, Valente R, Verbeke CS, Haas SL, Lohr JM. Kidney involvement in patients with type 1 autoimmune pancreatitis. J Clin Med, 2019; 8: 258. doi:10.3390/ jcm8020258.
- Saeki T, Kawano M, Mizushima I, et al. “The clinical course of patients with IgG4-related kidney disease,” Kidney International, 2013; 84(4): pp. 826–833.
- Kamisawa T, Okazaki K. “Diagnosis and treatment of IgG4-related disease,” Current Topics in Microbiology and Immunology, 2017; 401: pp. 19–33.
- Khosroshahi A, Stone JH. “Treatment approaches to IgG4-related systemic disease,” Current Opinion in Rheumatology, 2011; 23(1): pp. 67–71.
- Brito-Zer´on P, Kostov B, Bosch X, Acar-Denizli N, Ramos-Casals M, Stone JH. “Therapeutic approach to IgG4-related disease: a systematic review,” Medicine, 2016; 95(26): Article ID e4002.
- Hara S, Kanno T, Suzuki K. Rituximab for the treatment of IgG4-related disease. Annals of Internal Medicine, 2015; 162(2): 68-76.
- Khan ML, Colby TV, Viggiano RW, Fonseca R. Treatment with bortezomib of a patient having hyper IgG4 disease. Clin Lymphoma Myeloma Leuk, 2010; 10: 217-219. doi:10.3816/ 2010.n.034.
- Tanaka Y, Goto H, Suzuki K. IgG4-related kidney disease: A review of the literature and clinical experience. Clinical and Experimental Nephrology, 2014; 18(6): 907-914.

