Efficacy of 177Lutetium-PMSA-617 treatment in a Metastatic Castration-Resistant Prostate Cancer Patient
Omar Alonso Nuñez1,2 and Gerardo Gabriel dos Santos Loureiro1,2,*
1Uruguayan Centre of Molecular Imaging (CUDIM). Avenida Doctor Americo Ricaldoni 2010; Montevideo 11600, Uruguay
2Nuclear Medicine and Molecular Imaging Centre, Hospital de Clínicas, Universidad de la República, Avenida Italia s/n; Montevideo 11600, Uruguay
Received Date: 05/06/2025; Published Date: 03/07/2025
*Corresponding author: Prof. Adj. Gerardo Gabriel dos Santos Loureiro, MD, PhD, Uruguayan Centre of Molecular Imaging (CUDIM). Avenida Doctor Americo Ricaldoni 2010; Montevideo 11600, Uruguay; Nuclear Medicine and Molecular Imaging Centre, Hospital de Clínicas, Universidad de la República, Avenida Italia s/n; Montevideo 11600, Uruguay
Abstract
The 177Lutetium-PSMA-617 is a new radioligand therapy used in patients with hormone-resistant metastatic prostate cancer (HRMPC). We present the case of a patient with HRMPC who, having exhausted all his therapeutic options, underwent radioligand therapy receiving a total of 5 doses of 177Lutetium-PMSA-617, with an almost complete response and and regression of the symptoms.
Keywords: Hormone-resistant metastatic prostate cancer; Theragnosis-Diagnotherapy; 68Ga-PSMA-11; 177Lutetium-PSMA-617; PET/CT positron emission tomography
Introduction
Approximately 10-20% of prostate cancer patients have advanced disease at the time of diagnosis, and more than 40% will eventually progress to hormone-resistant metastatic prostate cancer, defined as disease progression despite pharmacological treatment or surgical castration [1].
In patients with advanced disease, systemic treatment is the cornerstone of therapy, with androgen deprivation therapy being a fundamental component followed by chemotherapy if the disease progresses [2].
For these cases, antiandrogens such as Enzalutamide and Abiraterone, as well as taxanes (Docetaxel, Cabazitaxel), are approved. However, the response to these treatments is often temporary, leaving many patients with symptomatic advanced disease and few therapeutic options [3].
Prostate-specific membrane antigen (PSMA) is found in large amounts on prostate cells and significantly increases in prostate cancer, which makes it a great target for treating advanced PC. Radioligand therapy using 177Lu-PSMA-617 is a specialized treatment for metastatic PC.
These findings verified that Lu-PSMA is a secure treatment choice for patients with metastatic prostate cancer and presents minimal side effects. Approximately 70% of patients experience a favorable reaction to the treatment, reflected by a decrease in PSA levels.
Case Report
58-year-old patient with a history of intensity-modulated radiation therapy for prostate adenocarcinoma Gleason 4+3=7 in eight samples from both lobes in July 2015, with perineural invasion (biopsy dated 02/10/2015). Initial PSA was 38.41 ng/ml. No findings of metastasis in bone scintigraphy from March 6, 2015. Good initial response to treatment. Subsequently, PSA levels rose in March 2017 (32.59 ng/ml), diagnosing multiple bone metastases in the spine, ribs, right clavicle, and sacrum. He began complete androgen blockade treatment with Leuprolide acetate 22.5 mg every 90 days and Bicalutamide 50 mg/day. This was supplemented with Zoledronic acid 4 mg every 30 days. Good initial progression with symptomatic improvement and decrease in PSA values to 0.23 ng/ml. Later, he presented with pain in the right clavicle, dorsolumbar spine, and sacroiliac region. PSA levels rose to 56.61 ng/ml, with testosterone levels of 0.40 ng/ml.
In the CT scan of the thorax, abdomen, and pelvis, no visceral or lymph node secondary lesions are observed. There is blastic involvement in multiple dorso-lumbar vertebral bodies, the right clavicle, and both iliac bones. The alkaline phosphatase level is 471 U/L. Given the patient's age, hormonal-resistant disease stage, and the presence of only multiple symptomatic bone metastases (pain in the spine, clavicle, and sacroiliac joint that hinders mobility), with the patient being in good general condition and actively working, treatment with 177Lutetium is indicated as treatment in patients with hormone-resistant prostate cancer. A total of 5 doses are administered, spaced 6 weeks apart with an average dose. A PET/CT with 68Ga-PSMA is performed after each dose, along with monitoring in the gamma camera.
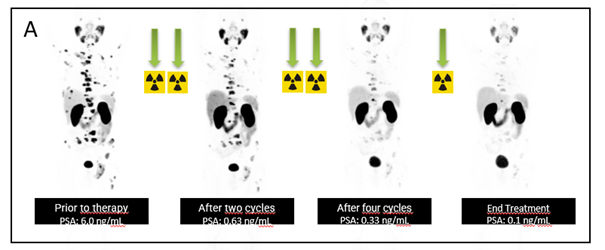
Figure 1: A: Sequential PET studies with 68Ga-PSMA-11 1-hour post-injection (MIP) in a patient with MHRPC. Note the continuous decline in PSA and the partial response in the PET images with 68Ga-PSMA PET after five cycles.

Figure 2: 59-year-old patient with PSMA-avid and bone metastases on pre-therapy [18F]-AlF-PSMA-11 PET/CT (A), and on 177Lu-PSMA-617 scintigraphy after 1st PSMA-Radioligand-Therapy (B). Note that the uptake in lesions and normal organs is very similar for the two radiopharmaceuticals.
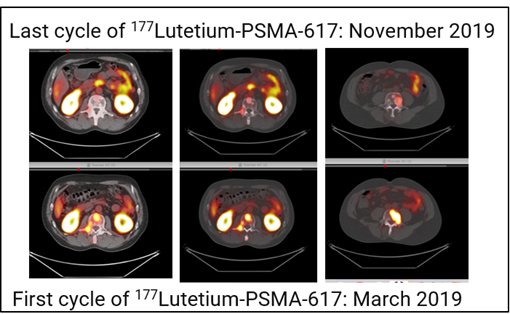
Figure 3: Axial 68Ga-PSMA PET/CT images showing regression of bone lesion uptake at the end of treatment.
Discussion
Prostate cancer is the second most common malignant tumor in men and one of the most common malignant diseases in the world [2].
On the other hand, HRMPC represents a clinical challenge in which new therapeutic tools are needed beyond chemotherapy with taxanes or hormonal treatment with abiraterone or enzalutamide. Indeed, in many cases, these therapies have proven to be relatively ineffective in controlling disease progression. For this reason, new therapeutic strategies capable of controlling the evolution of the disease at this stage are urgently needed [4].
The application of this THERAGNOSIS strategy in patients with prostate cancer was first described by Weineisen et al. who treated two patients with CPMHR using 177Lu-PSMA guided by PET/CT images with 68Ga-PSMA [5].
On March 23, 2022, the FDA approved Pluvicto ([Lu-177] lutetium-vipivotide tetraxetan, Advanced Accelerator Applications, Novartis) for the treatment of adult patients with positive metastatic prostate-specific membrane antigen (PSMA) with compound malignant hormone receptor [5].
The efficacy was evaluated in VISION (NCT03511664), a randomized (2:1), multicenter, open-label trial that assessed Pluvicto plus best standard of care (BSoC) (n=551) or BSoC alone (n=280) in men with progressive PSMA-positive MHRPC.
177Lu is an emerging radionuclide due to its chemical and nuclear characteristics (E: 497 keV, E: 113 and 208 keV, half-life T1/2: 6.7 days). It has a penetration range in tissue between 0.5 mm to 2 mm, allowing for the irradiation of tumor tissues. Being a simultaneous beta and gamma emitter, its application can be monitored using SPECT.
We report a clinical case of treatment with 177Lutetium-PSMA-617 in a patient with MHRPC, who had completed all available conventional therapeutic options. The treated patient met the determined inclusion/exclusion criteria and clinical and paraclinical controls were performed to assess the response to treatment, adverse effects, and treatment safety. Our patient received 5 doses of lutetium and had an incredible improvement.
Baum et al. [7] studied the toxicity and efficacy of treatment with 177Lu-PSMA in 56 patients with mCRPC. The authors administered a median dose of 5.8 GBq of this radiopharmaceutical per cycle (1 to 5 cycles). A significant absorbed dose was demonstrated both in bone and lymph node metastases (median = 3.3 mGy/MBq) with minimal toxicity, primarily at the level of the salivary glands (2 patients), which can be expressed as mild to moderate and transient xerostomia. It is noteworthy that no cases of significant hematological toxicity were reported. The authors recorded a slight post-treatment reduction in red blood cell and white blood cell counts, with no cases of thrombocytopenia. Additionally, pain severity decreased in one third of the patients. In this study, PSA levels fell by more than 50% in 62% of the patients. Furthermore, the CT showed a partial remission in 20% of the patients, while the PET/CT with 68Ga-PSMA demonstrated a partial response in 56% of the patients [6].
Similar results were reported by Kratochwil et al. [8] in which a retrospective evaluation was conducted on a series of 30 patients with CPMHR who underwent therapy with 1-3 cycles of 177Lu-PSMA, with a maximum administered activity of 6 GBq per cycle. All had lesions detected by PET/CT with 68Ga-PSMA. A response evaluated by a decrease in PSA was obtained in 70% of the patients, with decreases greater than 50% in 43% of the patients. Hematological toxicity was also mild-moderate and predictable. Fatigue, nausea, and xerostomia were described in less than 10% of cases. The mean absorbed tumor dose varied between 6 and 22 Gy/GBq for the first cycle. Dosimetric studies demonstrated a dose of 0.75 Gy/GBq for kidneys, 0.03 Gy/GBq for bone marrow, and 1.4 Gy/GBq for salivary glands [7].
Therefore, all studies agree that there is a low profile of toxicity, with critical organs being bone marrow, salivary glands, and kidneys. Fatigue, nausea, and xerostomia are seen in less than 10% of cases, and no significant cases of hematological, renal, or hepatic toxicity were reported [8].
In our study, the patient had a biochemical response for PSA, with an initial value of 6.0 ng/mL and a decrease to 0.1 ng/mL after 5 doses. The PET/CT images with 68Ga-PSMA-11 showed partial remission.
Conclusion
This clinical case of the use of 177Lutetium-PSMA-617 shows it to be a new therapeutic option that improves quality of life and has a good safety profile in patients with metastatic castration-resistant prostate cancer who have exhausted conventional therapeutic options and have low or minimal toxicity levels.
We conclude that therapy with 177Lutetium-PSMA-617 is a safe and well-tolerated therapeutic option for patients with MCRPC, improving the quality of life of the patients.
References
- Yap TA, Zivi A, Omlin A, de Bono JS. The changing therapeutic landscape of castration-resistant prostate cancer. Nat Rev Clin Oncol, 2011; 8(10): 597-610. doi: 10.1038/nrclinonc.2011.117.
- Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. International journal of cancer, 2015; 136: E359-386.
- Emmett L, Willowson K, Violet J, Shin J, Blanksby A, Lee J. Lutetium 177 PSMA radionuclide therapy for men with prostate cancer: a review of the current literature and discussion of practical aspects of therapy J Med Radiat Sci, 2017; 64: 52–60. doi: 10.1002/jmrs.22
- Kratochwil C, Afshar-Oromieh A, Kopka K, Haberkorn U, Giesel FL. Current Status of Prostate-Specific Membrane Antigen Targeting in Nuclear Medicine: Clinical Translation of Chelator Containing Prostate-Specific Membrane Antigen Ligands Into Diagnostics and Therapy for Prostate Cancer. Semin Nucl Med, 2016; 46(5): 405-418. doi: 10.1053/j.semnuclmed.2016.04.004.
- Weineisen M, Schottelius M, Simecek J, Baum RP, Yildiz A, Beykan S, et al. 68Ga- and 177Lu-Labeled PSMA I&T: Optimization of a PSMA-Targeted Theranostic Concept and First Proof-of-Concept Human Studies. J Nucl Med, 2015; 56(8): 1169-76. doi: 10.2967/jnumed.115.158550.
- Sartor O, de Bono J, Chi KN, Fizazi K, Herrmann K, Rahbar K, et al. VISION Investigators. Lutetium-177-PSMA-617 for Metastatic Castration-Resistant Prostate Cancer. N Engl J Med, 2021; 385(12): 1091-1103. doi: 10.1056/NEJMoa2107322.
- Baum RP, Kulkarni HR, Schuchardt C, Singh A, Wirtz M, Wiessalla S, et al. 177Lu-Labeled Prostate-Specific Membrane Antigen Radioligand Therapy of Metastatic Castration-Resistant Prostate Cancer: Safety and Efficacy. J Nucl Med, 2016; 57(7): 1006-1013. doi: 10.2967/jnumed.115.168443.
- Kratochwil C, Giesel FL, Stefanova M, Benešová M, Bronzel M, Afshar-Oromieh A, et al. PSMA-Targeted Radionuclide Therapy of Metastatic Castration-Resistant Prostate Cancer with 177Lu-Labeled PSMA-617. J Nucl Med, 2016; 57(8): 1170-1176. doi: 10.2967/jnumed.115.171397.

