Candida Albicans Endocarditis: A Case Report Highlighting the Emerging Demand for Updated Therapeutic Management
Pelagia Kefala Karli1, Vasilios Dimou2, Chariton Sapouridis2, Sofia Terzi2, Theodora Eleni Plakomyti2, Angeliki Mouzarou2,*
1Department of Internal Medicine, General Hospital Paphos, State Health Services Organization, Paphos, Cyprus
2Department of Cardiology, General Hospital Paphos, State Health Services Organization, Paphos, Cyprus
Received Date: 24/05/2025; Published Date: 02/07/2025
*Corresponding author: Angeliki Mouzarou, Department of Cardiology, Paphos General Hospital, State Health Services Organization, Paphos 8026, Cyprus
Abstract
Although fungal endocarditis is not encountered in clinical practice frequently and affects mainly patients with predisposing conditions, it carries a significant risk of mortality. The clinical features of most of the cases are obscured and patients often present to the emergency department with fever of unknown origin. We report herein such a case of a patient with underlying prosthetic mitral valve. The initial blood cultures grew Candida albicans and a large vegetation was observed on the bioprosthetic valve. Although surgical intervention was suggested, the patient considered unfit for surgical removal of the prosthesis and opted to be treated with antifungals. The presented case illuminates the fact that although current guidelines advocate for a combination approach, the feasibility of surgical treatment is frequently limited, as many patients present in a critically ill state that precludes operative management. This discrepancy between guideline recommendations and clinical reality highlights the need for updated management guidance.
Keywords: Fungal endocarditis; Candida; Surgery; Guidelines; Antifungals
Introduction
Fungal endocarditis (FE) is a rare cause of infective endocarditis accounting for only 2-4% of all cases; yet the last decades its incidence shows an upward trend [1]. The main pathogen species implicated are Candida spp., with Candida albicans being responsible for almost half of the reported cases. Aspergilus species account for the 25% of the remaining cases [1].
FE is an entity that predominately affects immunocompromised individuals, intravenous drug users and patients with prosthetic heart valves. Also, those with intravascular devices, a history of heart surgery or indwelling foreign materials such as central venous catheters as well as individuals receiving prolonged parental nutrition are at increased risk [2].
The diagnosis of FE is particularly challenging due to the nonspecific nature of its clinical presentation. Most patients present with prolonged fever of unknown origin, fatigue, sweating and altered mental status lacking the classic signs of bacterial endocarditis [1,2]. A new cardiac murmur or a quality-change in a previously well-known murmur may raise suspicion for FE, particularly when accompanied by signs of peripheral embolization [2].
Moreover, the diagnosis is complicated by the lack of highly sensitive laboratory techniques [3]. In clinical practice nearly half of the cases demonstrate negative blood cultures and thus, despite the vegetations seen in echocardiography it remains a challenge to meet the Duke’s diagnostic criteria [3]. Thus, still nowadays the diagnosis relies heavily on clinical judgment and imaging findings.
The treatment of FE should be aggressive due to its fatal nature. Current guidelines recommend a multi-factorial approach, combining antifungal agents with surgical intervention on the infected valve when indicated [3, 4]. Although the statistics show more favorable outcomes in patients who undergo surgical management, many of them in actual clinical settings are elderly, have multiple comorbidities, and present in critical clinical condition, rendering them unsuitable candidates for surgery.
Here we describe a case of prosthetic valve FE in an apparently immunocompetent patient and its clinical course. We aim to offer an understanding of this potentially fatal condition and highlight the gap in current therapy guidelines which are not followed in everyday clinical practice due to poor surgical candidacy.
Case Report
A 75-year-old man, initially admitted to Internal Medicine for fever of unknown origin and fatigue, was transferred to our Cardiology Department. The patient had been recently hospitalized due to moderate pericardial effusion and pleural effusion 3-months after undergoing coronary artery bypass grafting, mitral valve replacement, pulmonary vein isolation and left atrial appendage closure. He had a history of atrial fibrillation, hypertension, dyslipidemia, obstructive sleep apnea, and chronic kidney disease.
Upon admission, the patient was afebrile, hemodynamically stable, and physical examination revealed no abnormalities such as onychomycosis, peripheral emboli, or new heart murmur.
Trends of inflammatory markers during hospitalization are displayed in Figure 1.
Given the increased risk of infective endocarditis, a transthoracic echocardiography was performed, revealing a preserved left ventricular ejection fraction of 50% and a mobile mass attached to the mitral valve (Figure 2). Transesophageal echocardiography (TOE) confirmed a 1.4x1.4cm vegetation causing significant stenosis (mean pressure gradient 10mmHg) and mild valvular regurgitation (Figure 3).
Furthermore, 3-sets of blood cultures were obtained, all of which grew Candida albicans, with no bacterial or additional fungal growth detected. Based on the above findings, a diagnosis of FE was established. However, given the high intraoperative risk, the patient was deemed inoperable by the cardiac surgeons. Consequently, antimicrobial therapy was initiated with gentamycin 210mg IV daily, rifampin 600mg IV twice daily, vancomycin IV 1gr twice daily and caspofungin IV 150mg daily, guided by the blood cultures’ susceptibility testing results [5].
Additionally, coronary computed tomography angiography demonstrated total occlusion of the marginal branch venous graft, bilateral pleural effusions, and large hypodense formations within the bioprosthetic mitral valve (Figure 4).
Five-days after initiation of the therapeutic regimen, the patient developed symptoms suggestive of arterial embolization of the right lower extremity. Further evaluation confirmed ischemia (Rutheford IIa) due to thrombosis of the right common femoral artery (Figure 5). However, the patient declined surgery given the significant perioperative risk.
Follow-up TOE demonstrated a marked reduction in the dimensions of the vegetation (Figure 6), as well as a decrease in the mean transvalvular pressure gradient to 5 mmHg. Additionally, 7-days after admission blood cultures became negative. Despite these findings, the clinical status of the patient continued to deteriorate. Given the absence of conclusive evidence regarding the efficacy and optimal duration of conservative management, the patient was transferred to a cardiothoracic center for possible surgical intervention.
After 6-days the patient was re-admitted to our clinic in critical condition and altered mental status. Given his overall poor clinical status, conservative management was recommended. Although brain computed tomography revealed no acute pathology, the patient succumbed to complications of Candida endocarditis 1-day after readmission.
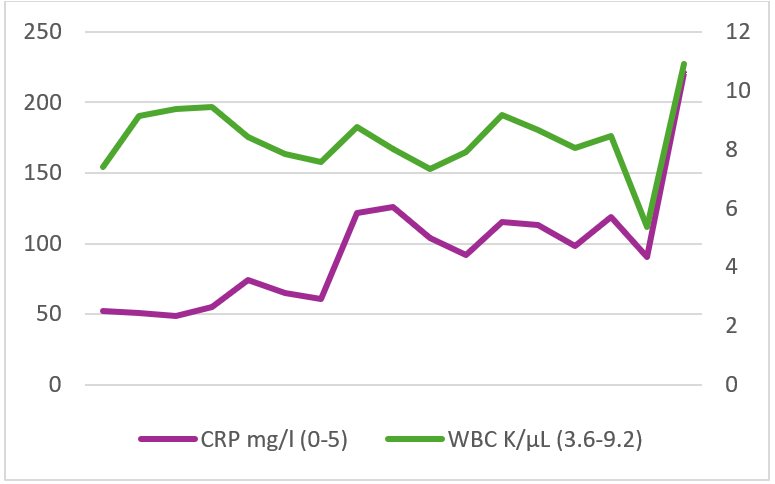
Figure 1: Time course of white blood cell count and serum C-reactive protein level during patient hospitalization. WBC, White blood cell; CRP, C-reactive protein.
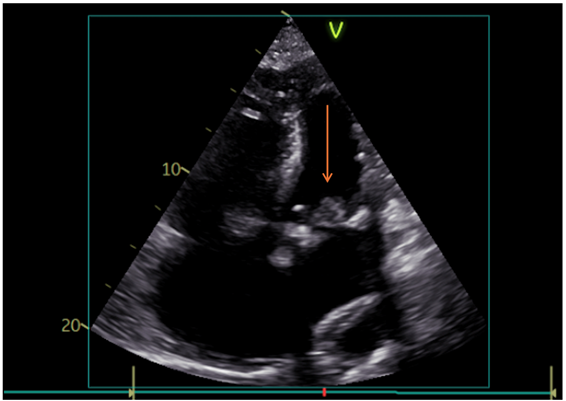
Figure 2: Vegetation on the bioprosthetic mitral valve as seen on transthoracic echocardiography on admission (arrow).
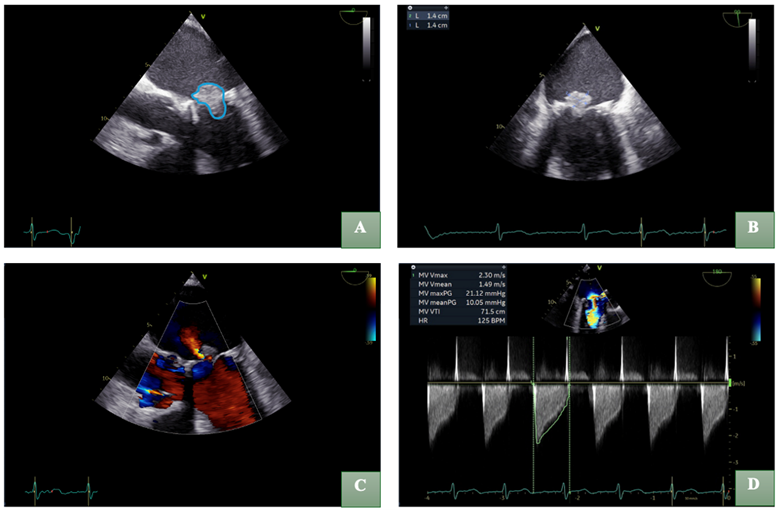
Figure 3: Transesophageal echocardiography (TOE) confirmed a vegetation, delineated by a blue outline (A) with dimensions measuring 1.4x1.4cm on magnified imaging (B) resulting in significant mitral stenosis and mild valvular regurgitation (C). Mitral valve maximal velocity (MV Vmax) 2.30 m/s; Mitral valve mean velocity (MV Vmean) 1.49 m/s; Mitral valve maximal pressure (MV max PG) 21.12 mmHg; Mitral valve mean pressure gradient (MV mean PG) 10.05 mmHg; Mitral valve velocity time integral (MV VTI) 71.5cm; Heart rate (HR) 125 beats per minute (D).
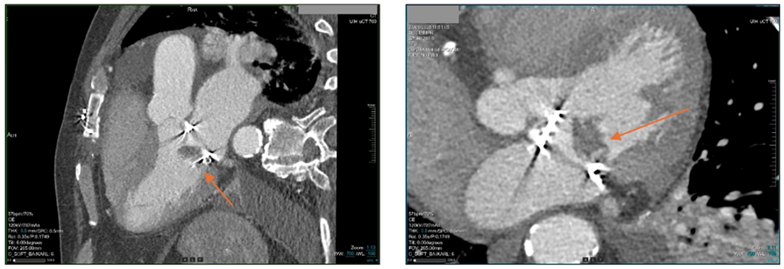
Figure 4: Coronary computed tomography angiography demonstrating the existence of large hypodense formations in the bioprosthetic mitral valve (arrow).
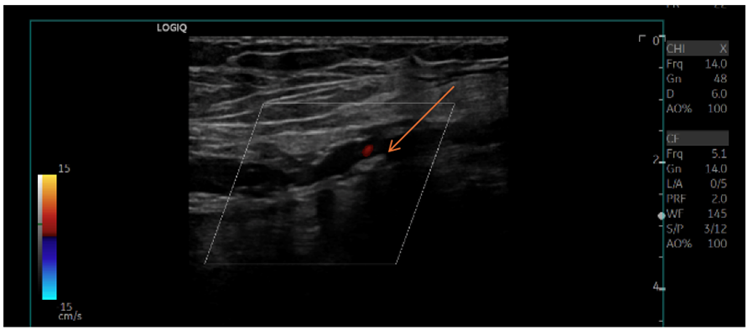
Figure 5: Doppler ultrasound of right lower extremity revealing the presence of thrombus in the right common femoral artery (arrow).
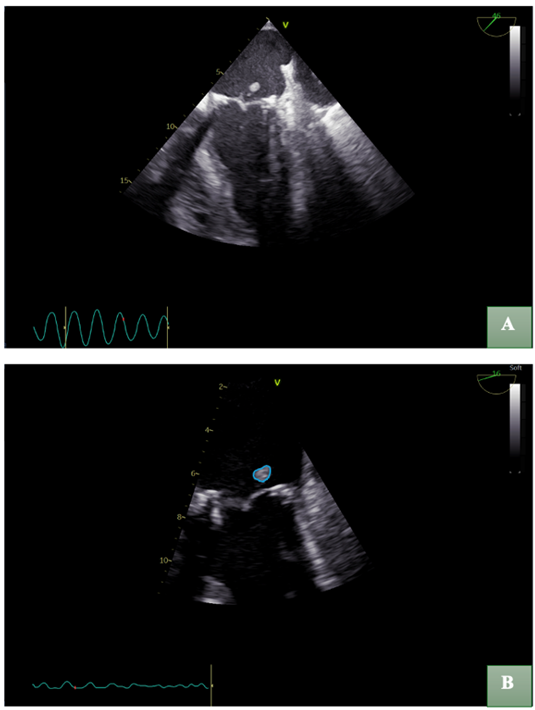
Figure 6: Follow-up transesophageal echocardiography (TOE) demonstrated a substantial reduction in vegetation size. (A) Distant view; (B) Zoomed-in view, illustrating the residual vegetation in a blue outline.
Discussion
Prosthetic valve endocarditis (PVE) is responsible for approximately 10-30 % of all forms of infective endocarditis and it is considered the most significant cause of morbidity and mortality [5]. The risk of acquiring infective endocarditis for patients is higher after the first year of their valve replacement and accounts for nearly 40% of all cases of PVE [6]. Candida species cause almost half of FE cases with Candida albicans being responsible for the vast majority [1].
Early recognition of fungal endocarditis requires a high index of suspicion by clinicians to initiate appropriate therapy and improve patient outcomes. The high mortality despite prompt treatment, the lack of sensitivity of the modified Dukes criteria, and the reduced accuracy of the recommended diagnostic methods make the diagnosis challenging. The diagnosis is based mainly on echocardiography, blood cultures, and histopathology. In cases of FE TOE is more sensitive and usually demonstrates larger vegetations when compared to cases of bacterial endocarditis [1]. In the case reported a large 1.4x1.4cm mass was identified on mitral prosthesis by TOE resulting in arterial embolization in the lower extremities; a common complication of FE [1].
Although in our case blood cultures revealed the causative agent, they are marked by low sensitivity (50-75%) and thus, in the last years newer techniques are gaining ground such as biomarkers (e.g., beta D-glucan and glucomannan) and polymerase chain reaction (PCR) [1]. Other imaging techniques that can be utilized are cardiac computed tomography (CCT), FDG-PET/CT and indium-111 leukocyte scintigraphy [1]. CCT can be used in diagnosis, identify the complications and anatomical variations making it helpful for surgical planning [1]. In our case CT scan underscored the large vegetation in the bioprosthetic mitral valve.
Nowadays, there is no universally accepted consensus on the optimal antifungal regimen, timing of surgical intervention, or criteria for selecting appropriate surgical candidates. However, the combination of antimicrobial regimen and surgery are considered to be the cornerstone of therapy; especially in patients with prosthetic heart valves where the presence of the fungal biofilm and the poor blood supply increase medical resistance [7].
Treatment usually includes liposomal amphotericin B with or without flucytosine. However, latest studies note the role of echinocandins either alone or in combination with the above-mentioned agents; especially in patients intolerant to amphotericin B [1]. Several studies showed increased survival in patients having received both antifungal agents and surgical intervention [8, 9] whereas increased mortality was noted with antifungal therapy alone [3,9].
However, successful treatment of FE only with antimicrobial therapy has been reported [10,11]. A recent study stated that medication-only treatment should be the initial approach; especially in patients with small-vegetations on imaging and few complications [12]. European Society of Cardiology guidelines suggest surgery should be selected on an elective or urgent basis depending on patients’ clinical condition [5]. On the other hand, other studies recommend that the early surgical intervention results in better prognosis [5,13].
In our case, although the persistent fungaemia and the size of vegetation made the patient candidate for surgery, antifungal therapy was adopted as the initial management approach because the patient was evaluated as frail for surgical intervention due to high intraoperative risk. It is noted that surgery is being performed approximately in 25-53% of all endocarditis cases [14]. A recent study showed that older age and the presence of heart failure at presentation are independently related with lower odds of surgical success and that although in one third of the cases surgery was indicated according to the guidelines the surgeons did not follow them because of the estimated decreased severity of infective endocarditis [15].
Conclusion
Candida albicans is the main culprit of FE marked by increased mortality rates. In clinical settings not only diagnosis but also treatment of FE remains a challenge. The limited implementation of advanced diagnostic techniques, the scarcity of evidence from randomized controlled trials, and the absence of uniform and detailed guidelines contribute to diagnostic delays, therapeutic uncertainty, and persistently high morbidity and mortality. A combination of systemic antifungal therapy and valve replacement surgery, when feasible, has been associated with improved clinical outcomes and survival. However, the aggressive nature of FE often results in rapid patient deterioration, highlighting the urgent need to integrate novel diagnostic techniques into routine practice and to revise current surgical candidacy criteria to better align with clinical realities and improve prognosis.
Author contributions: The authors contributed to the conception and design of this case report, and drafting of the manuscript. All authors reviewed and approved the final version of the manuscript.
Conflicts of Interest: None to declare.
Grand Information: The authors received no specific funding for this work.
References
- Thompson GR, 3rd, Jenks JD, Baddley JW, Lewis JS, 2nd, Egger M, Schwartz IS, et al. Fungal Endocarditis: Pathophysiology, Epidemiology, Clinical Presentation, Diagnosis, and Management. Clin Microbiol Rev, 2023; 36(3): e0001923.
- Yuan SM. Fungal Endocarditis. Braz J Cardiovasc Surg, 2016; 31(3): 252-255.
- Jamil Y, Akinleye A, Mirzaei M, Lempel M, Farhat K, Pan S. Candida endocarditis: Update on management considerations. World J Cardiol, 2023; 15(10): 469-478.
- Habib G, Lancellotti P, Antunes MJ, Bongiorni MG, Casalta JP, Del Zotti F, et al. ESC Guidelines for the management of infective endocarditis: The Task Force for the Management of Infective Endocarditis of the European Society of Cardiology (ESC). Endorsed by: European Association for Cardio-Thoracic Surgery (EACTS), the European Association of Nuclear Medicine (EANM). Eur Heart J, 2015; 36(44): 3075-3128.
- Delgado V, Ajmone Marsan N, de Waha S, Bonaros N, Brida M, Burri H, et al. ESC Guidelines for the management of endocarditis: Developed by the task force on the management of endocarditis of the European Society of Cardiology (ESC) Endorsed by the European Association for Cardio-Thoracic Surgery (EACTS) and the European Association of Nuclear Medicine (EANM). European Heart Journal, 2023; 44(39): 3948-4042.
- Wang A, Athan E, Pappas PA, Fowler VG, Jr., Olaison L, Paré C, et al. Contemporary clinical profile and outcome of prosthetic valve endocarditis. Jama, 2007; 297(12): 1354-1361.
- Boland JM, Chung HH, Robberts FJ, Wilson WR, Steckelberg JM, Baddour LM, et al. Fungal prosthetic valve endocarditis: Mayo Clinic experience with a clinicopathological analysis. Mycoses, 2011; 54(4): 354-360.
- Giuliano S, Guastalegname M, Russo A, Falcone M, Ravasio V, Rizzi M, et al. Candida endocarditis: systematic literature review from 1997 to 2014 and analysis of 29 cases from the Italian Study of Endocarditis. Expert Rev Anti Infect Ther, 2017; 15(9): 807-818.
- Meena DS, Kumar D, Agarwal M, Bohra GK, Choudhary R, Samantaray S, et al. Clinical features, diagnosis and treatment outcome of fungal endocarditis: A systematic review of reported cases. Mycoses, 2022; 65(3): 294-302.
- Noguchi M, Takai H, Eishi K, Atogami S. Prosthetic valve endocarditis due to Candida albicans treated successfully with medical treatment alone. Jpn J Thorac Cardiovasc Surg, 2004; 52(6): 318-321.
- Rajendram R, Alp NJ, Mitchell AR, Bowler IC, Forfar JC. Candida prosthetic valve endocarditis cured by caspofungin therapy without valve replacement. Clin Infect Dis, 2005; 40(9): e72-74.
- Arnold CJ, Johnson M, Bayer AS, Bradley S, Giannitsioti E, Miró JM, et al. Candida infective endocarditis: an observational cohort study with a focus on therapy. Antimicrob Agents Chemother, 2015; 59(4): 2365-2373.
- Pierrotti LC, Baddour LM. Fungal endocarditis, 1995-2000. Chest, 2002; 122(1): 302-310.
- Liesman RM, Pritt BS, Maleszewski JJ, Patel R. Laboratory Diagnosis of Infective Endocarditis. J Clin Microbiol, 2017; 55(9): 2599-2608.
- Rivoisy C, Vena A, Schaeffer L, Charlier C, Fontanet A, Delahaye F, et al. Prosthetic Valve Candida spp. Endocarditis: New Insights Into Long-term Prognosis-The ESCAPE Study. Clin Infect Dis, 2018; 66(6): 825-832.

