A P-Anca Positive Patient With Recently Treated Vasculitis Presenting with Shortness of Breath and Fever: A Diagnostic Dilemma
Ji Son1*, Vishal Vashistha MD2, Kelsey McHugh1, David Gugliotti2, Swapna Kollikonda1
1Department of OBGYN, Cleveland Clinic Women’s Health Institute, USA
2Cleveland Clinic Medicine Institute, USA
3Cleveland Clinic Robert J. Tomsich Pathology and Laboratory Medicine Institute, USA
Received Date: 04/11/2020; Published Date: 07/12/2020
*Corresponding author: Ji Son MD, MS, Department of OBGYN, Cleveland Clinic Women’s Health Institute, 9500 Euclid Ave, Desk A81, Cleveland OH 44195, USA. Email: sonj@ccf.org, Phone: 216-296-1698
Case Report
A 65 year old male with recently diagnosed P-ANCA (MPO+) vasculitis presented to his primary care physician with mild dyspnea for the preceding 3-4 weeks, productive cough, and low-grade fever. At initial presentation six months prior, he complained of rash, leg swelling, sinus pressure, hearing loss, and tingling sesations in his extremities. Due to concern for an antibody-associated vasculitis, both kidney and skin biopsies were obtained. Renal biopsy showed a necrotizing glomerulonephritis with glomerular fibrinoid necrosis, cellular glomerular crescents, and scattered mesangial IgA and C3 deposition. Skin pathology was interpreted as leukocytoclastic vasculitis, with direct immunofluorescence demonstrating perivascular complement C3 deposition without immunoreactivity for IgG, IgA, and IgM. Concordantly, ANCA titers showed positive P-ANCA and negative C-ANCA, and the patient was discharged with diagnosis of P-ANCA vasculitis with overlapping IgA nephropathy. Patient was started on cyclophophamide, pulse dosed prednisone followed by taper from 60 mg to 30mg at presentation, and monthly inhaled pentamidine for Pneumocystic Jiroveci Pneumonia (PJP) prophylaxis. Because an array of etiologies could be responsible for his current symptomatology, his primary care physician orderd a chest x-ray, which showed non-specific reticular opacities (Figure 1). These radiologic findings in combination with his clinical presentation led to direct admission for further evaluation.
Once admitted to the general medicine service, the patient’s O2 saturation was noted at 91%, and he had 3+ pitting lower extremity edema despite a stable dose of torsemide. Arterial blood gas revealed a PaO2 of 59 mm Hg. A chest CT without contrast showed ground glass attenuation with interlobular septal thickening (Figure 2).
Differential diagnosis included infection in the setting of immunosuppression, vasculitis flare presenting with pulmonary changes, cyclophosphamide-induced pneumonitis, and cyclophosphamide-induced cardiotoxicity. Infectious workup including blood and urine cultures were negative. Cardiotoxicity was ruled out by lack of jugular venous distention on exam, normal NT Pro-BNP, and unremarkable echocardiogram.
A bronchoscopic evaluation was then performed. Bronchoalveolar lavage ruled out unusual infectious etiologies and diffuse alveolar hemorrhage, while biopsy ultimately showed organizing pneumonia (Figure 3). Based on the chronology of events, the diagnosis of cyclophosphamide-induced pneumonitis was favored. The patient was discharged with oxygen on a slow taper of 60 mg prednisone daily with plans to alter his immunosuppression regimen. Over the next 3 months, his symptoms improved significantly. He was ultimately transitioned to azathioprine and rituximab for management of his underlying vasculitis. By 6 months following his initial presentation for pulmonary symptoms, the patient was using only 10 mg of prednisone daily.
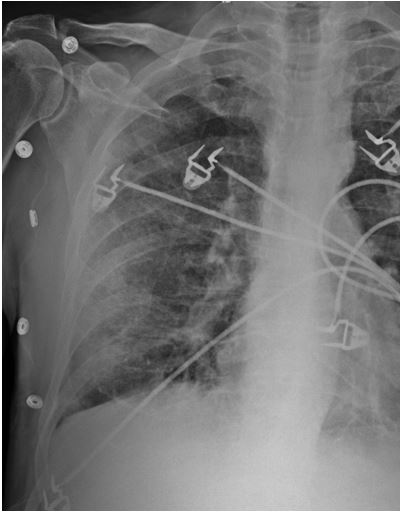
Figure 1: Lytic lesions in the scull in CT scan.
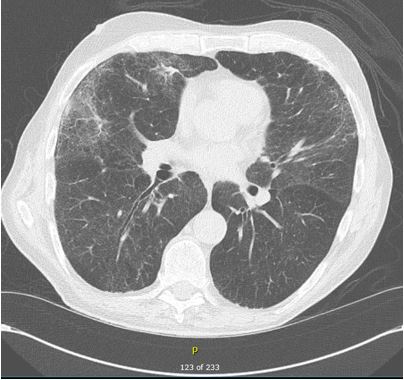
Figure 2: Lytic lesions in the scull in CT scan.
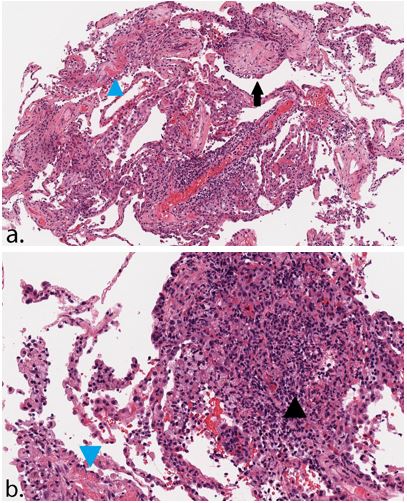
Figure 3a&b. Lung biopsy showing alveolated lung parenchyma with alveolar plugs of fibromyxoid tissue (arrow) and foci of organizing fibrin (blue arrowhead), acute inflammation, and rare aggregates of epithelioid histiocytes (black arrowhead) consistent with organizing and acute fibrinous pneumonia, H&E 100x (a), 200x (b).
Cyclophosphamide-Induced Pneumonitis
Surveillance for drug toxicity remains a critical component of managing patients with vasculitides. While the combination of prednisone and cyclophosphosphamide leads to control of diease in 80-90% of patients with Granulomatosis with Polyangiitis (GPA), this regimen is associated with dose-dependent treatment-related morbidity in over 50% of patients [1]. Such toxicity has led to increased use of rituximab, which may be cost-prohibitive in resource limited settings. Our patient differed slightly from the typical P-ANCA positive population because of the presence of IgA nephropathy rather than pauci-immune complex deposition.
Pulmonary manifestations of cyclophosphamide toxicity are rare with frequency less than 1% [2]. Interestingly, Malik et al. found that four of the six patients with cyclophosphamide-induced pulmonary toxicity in their study had GPA; this may suggest an underlying susceptibility in this population. Clinical features of pulmonary toxicity follow two patterns: an early onset acute pneumonitis and a chronic progressive pneumonitis [3]. Early onset pneumonitis develops within 1-6 months after exposure and is likely to improve if the drug is discontinued with early initiation of corticosteroid [4]. CT findings include ground-glass pattern with upper-lobe predominance.
Histopathologic findings are relatively non-specific and may show diffuse alveolar damage, cryptogenic organizing pneumonia, and alveolar hemorrhage. Late onset pneumonitis occurs in the setting of prolonged cyclophosphamide use and leads to progressive pulmonary fibrosis that is not responsive to steroid treatment. The mechanism of pulmonary injury in either case involves the metabolism of cyclophosphamide into alkylating metabolites and acrolein, which may alter the cell membrane state [5]. Other side effects associated with long-term use of cyclophosphamide include hair loss, leukopenia, hemorrhagic cystitis, infertility, and secondary malignancies.
Conclusion
The toxicities from medications used to treat vasculitides can lead to similar presentations to the actual underlying condition. A thorough chronologic history, understanding of the adverse effects from treatments, and appropriate diagnostic work-up are necessary to identify the etiology of symptoms.
Conflict of Interest
None
References
- Reinhold-Keller E.,Beuge, N.,Latza, U.,deGroot,K.,Rudert, H.,Nolle, B.,Heller, M.,Gross, W. An interdisciplinary approach to the care of patients with wegener's granulomatosis. Arthritis Rheumatol. 2000;43(5):1021-1032.
- Twohig, K., Matthay, R. Pulmonary effects of cytotoxic agents other than bleomycin. Clinc Chest Med. 1990; 11(1):31-54.
- Malik, S., Myers, J., DeRemee, R., Specks, U. Lung toxicity associated with cyclophosphamide use. Two distinct patterns. Am J of Resp Crit Care Med. 1996; 154(6):1851-1856.
- Spector, J., Zimbler, H., Ross, J. Early-onset cyclophosphamide-induced interstitial pneumonitis. JAMA. 1979; 242(26):2852.
- Patel, J. Metabolism and pulmonary toxicity of cyclophosphamide. Pharmacol. Ther. 1990; 47(1): 137-146.

