Some Audiologic Characteristics in Individuals with Sotos Syndrome
Samuel RA1, Lieblong S2, Nannette N3 and Schaefer GB4
1Department of Audiology and Speech Pathology and Otolaryngology – Head& Neck Surgery, University of Arkansas for Medical Sciences, USA
2Department of Audiology, Ear, Nose, & Throat Group, Memphis, Tennessee, USA
3 Department of Audiology, Nova Southeastern University, USA
4Department of Genetics and Pediatrics, University of Arkansas for Medical Sciences, USA
Received Date: 12/08/2020; Published Date: 25/08/2020
*Corresponding author: Samuel R Atcherson, Department of Audiology and Speech Pathology, University of Arkansas for Medical Sciences, Little Rock, Arkansas, USA. Tel: +1 501-526-4235; E-mail: sratcherson@uams.edu
Abstract
We conducted an audiological assessment of 27 children and adults with Sotos syndrome in order to provide information about this population’s hearing and auditory processing abilities. Otoscopy, tympanometry, audiologic screening, distortion-product otoacoustic emissions (DPOAE) screening, speech-in-noise testing using the QuickSIN (SNR loss), and Dichotic Digits Test made up the test battery. Except for a small percentage of cases, otoscopy, tympanometry, and pure tone screening results were unremarkable and not unlike the general population. However, adults had large ear canal volumes similar to those reported in older adults in the general population. DPOAE screening was attempted with greater than expected referral rates, most likely due to cooperation issues and noise levels. SNR loss was generally higher (poorer) than published norms, and Dichotic Digit test results revealed a slight right ear advantage and an overall depression of scores in both ears compared to normative data. Results of the audiologic screenings indicated that the majority of individuals with Sotos syndrome do not appear to have hearing sensitivity issues co-occurring with the syndrome. However, QuickSIN and Dichotic Digit test results indicate that those with Sotos syndrome may have speech-in-noise and callosal-specific auditory transfer issues that together negatively impact complex listening.
Keywords: Sotos Syndrome; Hearing; Auditory Processing
Introduction
Sotos syndrome is an overgrowth syndrome that is characterized by excessive physical growth, characteristic facial features, developmental delays, and an advanced bone age [1]. The etiology of the syndrome in the majority of cases has recently been attributed to mutations in the NSD1 (nuclear SET- domain 1) gene on chromosome 5 [2]. Those diagnosed with Sotos syndrome often have mild to severe intellectual disabilities, delayed motor skills, and impaired social and cognitive development [3]. Speech and language delays and/or disorders have also been reported in those diagnosed with Sotos syndrome [4]. Advanced bone age and the growth and size is another characteristic among those with Sotos syndrome [5]. There is no standard course of treatment for Sotos syndrome with the current recommendations focusing on treating the symptoms that are present. Classic Sotos syndrome occurs in one in every 10,000-15,000 births, making it a rare syndrome [4]. Due to the varying symptoms and presenting features, in the past it has been common for those with Sotos syndrome to be misdiagnosed with other syndromes [4]. Example syndromes that may be similar to Sotos include Weaver-Smith, Beckwith-Wiedemann, Simpson dysmorphia, and Fragile X. Confirmation of any of these syndromes can be obtained through genetic testing.
Currently, there is a paucity of information about auditory function and auditory processing abilities in individuals with Sotos syndrome. In one otolaryngologic study [6], a retrospective medical records review yielded seventeen patients between the ages of 2 and 19 years diagnosed with Sotos syndrome, five of whom had a confirmed NSD1 mutation. Of those confirmed with NSD1, four were under routine care of an otolaryngologist. Two of five presented with high frequency sensorineural hearing loss. Another common abnormality reported for those with Sotos syndrome involves changes in the midline structures of the brain [7,8]. Among other abnormalities, those diagnosed with Sotos syndrome may have a variety of abnormalities of the corpus callosum including hypogenesis of the structure. With the “thin” corpus callosum, we hypothesized that individuals with Sotos syndrome may have auditory processing issues, which contribute to their characteristic social and cognitive deficiencies.
Clinical Approach
The annual conference of the Sotos Syndrome Support Association (http://sotossyndrome.org/) made it possible to obtain a convenience sample of children and adults with Sotos syndrome from all over the world in a supportive and safe environment. The following intake data was collected: date of birth (for chronological age calculation); gender; history of hearing device use; history of hyperbilirubinemia, seizures, heart defects, and confirmation of genetic testing (i.e., NSD1). Audiologic testing included otoscopy, tympanometry, pure tone air conduction screening (0.5, 1, 2, and 4 kHz at 20 dB HL), and pure tone air conduction threshold testing for those who did not pass the hearing screening (0.25, 0.5, 1, 2, 4, and 8 kHz). For automated distortion-product otoacoustic emission (DPOAE) screening, a response of 3 out of 4 DPOAE test frequencies constituted a pass in that ear [9]. Auditory processing testing/screening consisted of the speech-in-noise testing using the QuickSIN (Etymotic Research, Elk Grove Village, IL), and Dichotic Digits Test (Auditec, St. Louis, MO). All testing was conducted in a two-room suite a conference hotel with adjoining rooms. Room noise measurements were conducted and were below maximum permissible noise levels (ANSI S3.1-1999). The test procedures were approved by the University of Arkansas for Medical Sciences Institutional Review Board (#203107).
Inclusion and Exclusion Criteria
Inclusion criteria were: (a) confirmed diagnosis of classic Sotos syndrome using the “strict criteria” (see e.g., [3,4,7]); (b) if DNA testing had been performed, a confirmatory diagnosis via a known NSD1 mutation was present; (c) age 5 years and older; and (d) verbal consent/assent and/or have parental consent to participate. Exclusion criteria were: (a) cases without a “strict” diagnosis, and (b) cases younger than 5 years of age.
Case Report
A total of 27 cases ranged in age from 5 years 6 months to 36 years 1 month. The oldest child recruited was 14 years 9 months and the youngest adult was 18 years 7 months. There were 10 male and 17 female cases. None reported previous use of hearing devices. Nineteen of 27 participants (70.4%) had known history of hyperbilirubinemia as infants, 12 of 27 (44.4%) had known history of seizures, 12 of 27 (44.4%) had known history of heart defects, and 15 of 27 participants (55.6%) had confirmed NSD1 mutation. Table 1 provides an overview of medical history and other characteristics.
Table 1: Medical history and other characteristics.
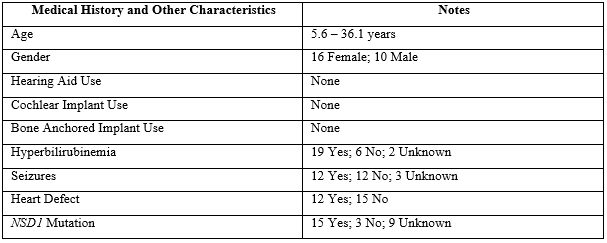
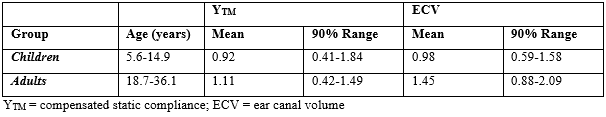
Of the 54 ears, otoscopy was unremarkable with the exception of 4 partially-occluded ear canals and 1 completely occluded ear canal. Tympanometry revealed a Type-A configuration for 49 ears, a Type-B configuration for 3 ears, and a Type-C configuration for 2 ears (N=54). Due to reports of advanced bone age and the growth and size in Sotos syndrome (e.g., [5]), static admittance (YTM), tympanometric peak pressure (TPP), and ear canal volume (ECV) were examined only for Type-A configurations separately for children (n=21) and adults (n=5). Table 2 shows YTM and ECV data (right and left ears combined) for the children and adults compared to published normative data [10].
Table 2: Tympanometric results by age for Sotos syndrome cases.
For the pure tone screening, 23 cases passed in both ears and 4 referred. Pure tone audiometry was completed on 2 cases with parental request, and 1 of these 2 had previously confirmed hearing loss. Separating pure tone screening by ear, 26 passed and 1 referred for the right ear; 23 passed and 4 referred for the left ear. For DPOAE screening results, 10 passed in both ears and 7 referred or could not be tested.
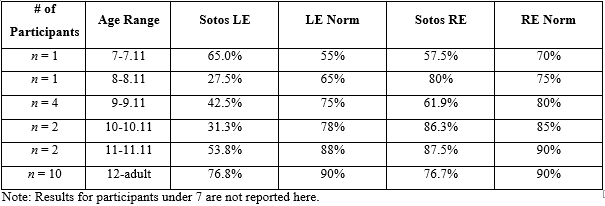
The two remaining speech tests, QuickSIN and Dichotic Digits test, were administered to as many participants who could complete the tasks cooperatively. Twenty-two of 27 participants were able to complete the QuickSIN and 21 of 27 participants completed Dichotic Digit testing. The QuickSIN and Dichotic Digit scores are shown in Tables 3 and 4, respectively, by age group with a side-by-side comparison to published normative data [11,12].
Table 3: Average score results by age for QuickSIN SNR loss compared to published norms.
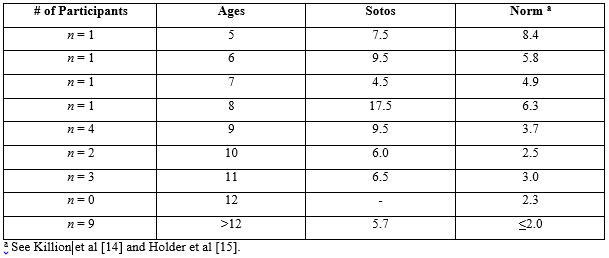
Table 4: Average score results by age on Dichotic Digits test compared to published norms.
Discussion
A loss of hearing sensitivity does not seem to be characteristic of children and young adults diagnosed with Sotos syndrome, and the prevalence of hearing loss in this population is comparable to what is seen in the general population. Although one might expect that the advanced bone aging characteristic of Sotos syndrome might be apparent in associated cartilaginous changes in the ear canal resulting in disproportionately higher ear canal volumes, this did not prove to the case. Both YTM and ECV values in the children with Sotos syndrome tended to cluster at the upper limits of normal. Similar to the children, both YTM and ECV values in the adults with Sotos syndrome also tended to cluster at the upper limits of normal. Interestingly, these 5 adult participants, ages 18.7 to 36.1 years, had ECVs much more consistent with older adults in the general population (e.g., [5]), which may relate to the observation of advanced bone age in individuals with Sotos syndrome. Inconsistent with pure tone and tympanometry screening, the DPOAE screening resulted in nearly two-thirds (17 out of 27 participants) referral rate (or could not test) by participant regardless of ear, and almost half the referral rate (or could not test) when viewed by ear alone. For those ears tested and referred on the DPOAE screening, inability to cooperate and/or participant noise levels were the culprit. This is an important consideration for future consideration, since pure tone screening and tympanometry are likely to be more useful with routine screening among this population.
Twenty-two of the 27 participants completed the QuickSIN. Four participants were unable to be conditioned to the task and one participant was not tested due to the reported hearing loss. Individual results from this study suggest that the majority of the participants would have trouble listening in noisy conditions. Twenty-one of 27 participants completed the Dichotic Digit Test. Five participants were unable to be conditioned to the task and one participant was excluded from testing due to the hearing loss. Dichotic Digit Test results were grouped by age and averaged for each group for a comparison to normative data. QuickSIN scores across all ages point to poorer than normal SNR loss in participants with Sotos syndrome [14,15]. For Dichotic Digits, the poorer left than right ear scores follows the pattern for normative data, and there is an observable overall depression of right and left ear scores that persists into adulthood.
Conclusion
Overall, this case series suggests that those who are diagnosed with Sotos syndrome do not have a co-occurring hearing loss demonstrate hearing sensitivity consistent with the general population. However, those diagnosed with Sotos syndrome are likely to have some auditory issues related to cerebral dysgenesis. Specifically, individuals with Sotos syndrome may have some difficulties with complex listening situations that persists into adulthood. Information about their auditory characteristics provides professionals with baseline information for working with individuals diagnosed with this condition and a foundation for further exploration regarding their auditory processing capabilities. Due to the varying clinical manifestations that can occur with Sotos syndrome, additional information about possible audiologic and auditory processing characteristics may aid in appropriate service provision for those diagnosed with Sotos syndrome.
Aknowledgements
The authors are grateful to the families and organizers of the Sotos Syndrome Support Association conference. In addition, the authors thank Patti Martin, Ph.D. of Arkansas Children’s Hospital for loaning some equipment. The authors thank graduate clinician volunteers from the Genetics of Hearing Loss course who helped with data collection: Sarah Benton, Mary Liz Crigler, Rose Dockery, Carrie Eaton, Elissa Honea, Andrea Knapp, Stacey Kolb, Tessa Menchaca, and Abby Smith.
References:
- Cloe TR. Hughes HE. Sotos syndrome: A study of the diagnostic criteria and natural history. J Med. Genet.1994;31:20-32.
- Kurotaki N, Imaizumi K, Harada N. et al. Haploinsufficiency of NSD1 causes Sotos syndrome. Nat Genet. 2002;30:365-366.
- Ball LJ, Sullivan MD, Dulany S, et al. Speech‐language characteristics of children with Sotos syndrome. Am J Med Genet A. 2005;36:363-367.
- Anderson R, Buehler B, Schaefer GB. Sotos syndrome: A Handbook for Families, 2nd Edition. Omaha, NE: Munroe-Meyer Institute, University of Nebraska Medical Center. 2000.
- Rao VH, Beuhler BA, Schaefer GB. Accelerated linear growth and advanced bone age in Sotos syndrome is not associated with abnormalities of collagen metabolism. Clin Biochem. 1998;31:241-249.
- Gaudreau P, Zizak V, Gallagher TQ. The otolaryngologic manifestations of Sotos syndrome. Int J Pediatr Otorhinolaryngol. 2013;77:1861-1863.
- Schaefer GB, Bodensteiner JB, Buehler BA, et al. The neuroimaging findings in Sotos syndrome. Am J Med Genet. 1997;68:462-465.
- Hinkley LB, Marco EJ, Findlay AM, et al. The role of corpus callosum development in functional connectivity and cognitive processing. PloS ONE. 2012;7:39804.
- Christensen L. A Universal Pass/Refer Criterion for DPOAEs: Is it Possible? Hear Rev. 2000;7(2):22,24,29,30.
- Margolis RH, Heller JW. Screening tympanometry: Criteria for medical referral. Audiology. 1987;26:197–208.
- Musiek FE. Assessment of central auditory dysfunction: the dichotic digit test revisited. Ear Hear. 1983;4:79-83.
- Bellis TJ. Assessment and Management of Central Auditory Processing Disorders in the Educational Setting: From Science to Practice (2nd ed.). Clifton Park, NY: Thompson Delmar Learning. 2003.
- Wiley TL, Cruickshanks KJ, Nondahl DM, Tweed TS, Klein R, & Klein, BEK. Tympanometric measures in older adults. J Am Acad Audiol. 1996;7:260-268.
- Killion MC, Niquette PA, Gudmundsen GI, et al. Development of a quick speech-in-noise test for measuring signal-to-noise ratio loss in normal-hearing and hearing-impaired listeners. J Acoust Soc Am. 2004;116:2395-2405.
- Holder JT, Sheffield SW, Gifford RH. Speech understanding in children with normal hearing: sound field normative data for BabyBio, BKB-SIN, and QuickSIN. Otol Neurotol. 2016;37:e50-e55.

