Bicuspid Aortic with Severe Aortic Insufficiency and Polycystic Hepatorenal Disease: An Exceptional Combination
Bouamoud A*, Bouazaze M, Zahidi Alaoui H, Malki M and Pr Asfalou I
Mohamed V Military Instruction Hospital, Rabat, Morocco
Avicenna University Hospital Rabat, Morocco
Received Date: 08/05/2025; Published Date: 16/06/2025
*Corresponding author: Bouamoud A, Mohamed V Military Instruction Hospital, Rabat ; Avicenna University Hospital Rabat, Morocco
Abstract
Autosomal dominant polycystic kidney disease is the most common inherited kidney disease. Polycystic kidney disease is part of the group of conditions known as "ciliopathies", whose involvement of several organs results from dysfunction of the primary cilia. Cardiovascular damage caused by autosomal dominant polycystic kidney disease is numerous and sometimes appears early in the patient's life.
In this report, we describe the association of polycystic hepatorenal disease and bicuspid aortic valve with severe leakage. To date, no other cases have been reported.
Through our observation we will discuss the epidemiological, clinical and pathophysiological aspect of polycystic hepatorenal cystic disease, as well as the difficulty involved in heart valve surgery in our case, and we will focus on the hemorrhagic risk of anticoagulation on hepatorenal cysts as well as on the type of valve to choose.
Introduction
Autosomal Dominant Polycystic Kidney Disease (ADPKD) is the most common inherited kidney disease. Polycystic kidney disease is part of the group of conditions known as "ciliopathies", whose involvement of several organs results from a dysfunction of the primary cilia The cardiovascular damage of autosomal dominant polycystic kidney disease is numerous and sometimes appears early in the patient's life.
In this report, we describe the association of polycystic hepatorenal disease and bicuspid aortic valve with severe leakage. To date, no other cases have been reported [1,2].
Case Report
This is a 50-year-old patient, with a cardiovascular risk factor of high blood pressure on dual therapy valsartan and amlodipine, known to be a carrier of polycystic kidney disease complicated by high blood pressure and renal failure at the hemodialysis stage for 8 years and hemorrhage of kidney cysts revealed by hematuria.
Cardiac evaluation as part of the preoperative assessment of a nephrectomy was requested.
Clinically, the patient had stage II/III dyspnoea that was not well improved by dialysis sessions. The clinical examination found a very distended abdomen with collateral venous circulation, and hepatomegaly with a hepatic arrow at 14 cm. A systolic blood pressure of 120 mmHg (after two hours of taking his anti-hypertensive treatment) and a diastolic blood pressure of 50 mmHg. On cardiac auscultation, an aspirative diastolic murmur is found at the aortic focus and at the left edge of the sternum.
The ECG shows a sinus rhythm with left ventricular hypertrophy of the systolodiastolic type. The 48-hour ECG holter did not show atrial fibrillation.
The ETT objectified a bicuspid aortic valve type 0, with severe aortic leakage: vena contracta at 8 mm, an ORS at 50 mm2 and a regurgitating volume (RV) at 80 ml; a dilated left ventricle with a telediastolic diameter at 64 mm and an LVEF at 60%; a left atrium dilated at 25 cm². The ascending aorta is undilated measured at 30 mm (Figures 3 and 4).
Faced with the observation of hepatomegaly and abdominal distention on clinical examination, an abdominal CT scan was requested. Thus, we were able to objectify polyrenal hepatocellular cystic disease (Figures 1 and 2).
After multidisciplinary discussions between cardiologists, cardiac surgeons, nephrologists and gastrologists it was decided to do an aortic valve replacement with a biological valve, however the patient refused any surgery.
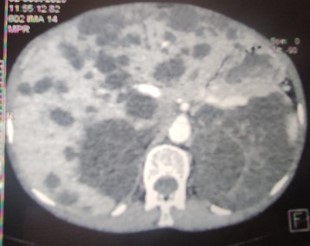
Figure 1: Axial CT scan image of the abdomen revealing a polycystic liver.
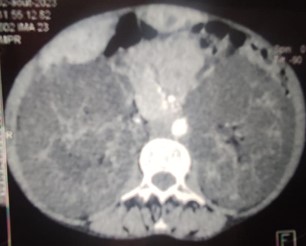
Figure 2: Axial CT scan image of the abdomen showing two large polycystic kidneys.
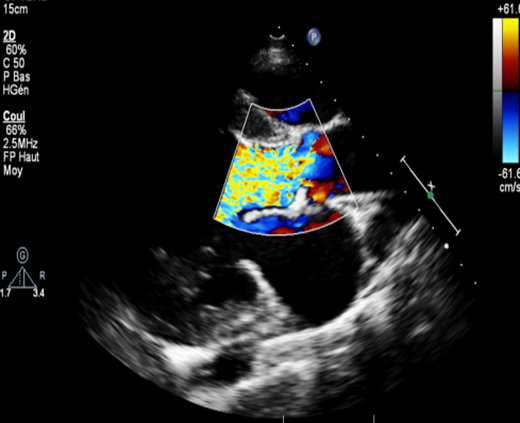
Figure 3: Parasternal long-axis view with color Doppler showing severe aortic regurgitation.
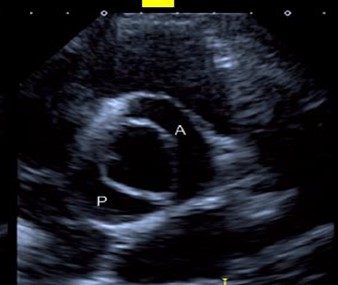
Figure 4: Parasternal short-axis view showing a type 0 bicuspid aortic valve, open during systole.
Discussion
ADPKD has an estimated prevalence of between 1/800 and 1/2500 [1].
Three genes are involved in this disease: PKD1, PKD2 and a recently identified gene, GANAB, which represents only a few cases at the moment and is often associated with liver damage. 1]
The PKD1 and PKD2 genes encode proteins named polycystin-1 and polycystin-2, respectively. These transmembrane proteins are expressed almost ubiquitously in the primary cilia.
Liver cysts are the most common extrarenal manifestation in patients with mutations in the PKD1, PKD2 and GANAB genes. However, this must be differentiated from isolated polycystic liver disease, without renal or systemic involvement and which is linked to mutations in the PRKCSH or SEC63 genes [1]. The symptoms of liver cysts are related to the mass effect with compression of the liver parenchyma and adjacent structures, which can lead to chronic pain, dyspnea, early satiety, gastroesophageal reflux disease or lower back pain. The liver volume can become, in some cases, very large and threaten the diet of these patients. Malnutrition ensues and preteriorates the survival of these patients, who are often already weakened by kidney failure [1].
Our patient reported dyspnea not improved by dialysis, which suggests that it is probably dyspnea related to the mass effect of the cysts.
Acute complications of liver cysts are infection and hemorrhage, and are marked by abrupt abdominal pain in both cases [1,4].
Numerous studies have demonstrated an association between mutations linked to the PKD1 and PKD2 genes and alteration of the artery walls. Polycystins 1 and 2 are also present in smooth muscle cells, as well as on the surface of endothelial cells in all major vessels. Vascular disorders associated with polycystic kidney disease include intracranial aneurysms, dolichoectasis, intrathoracic and cervicocephalic aortic dissections, and coronary artery aneurysms [1]
A prevalence of 15% mitral prolapse, 30% mitral regurgitation, 10 to 20% aortic insufficiency and 5% tricuspid prolapse was measured in patients with polycystic disease, prevalences markedly increased compared to other family members not affected by polycystic disease. Polycystic disease is part of a group of conditions known as "ciliopathies," whose involvement of multiple organs results from dysfunction of the primary cilia, as well as aortic bicupidia. To date, no other cases of bicuspid aortic valve with aortic leakage associated with ADPD have been reported. [2] [3]
According to our analysis of the cases in the literature, we did not find any similar case discussing the replacement of the aortic valve, in particular the type of valve: biological or mechanical; given the risk of haemorrhagic complications of cysts under anticoagulation; We opted for a biological valve.
Conclusion
Polycystic hepatorenal disease has a poorer prognosis than isolated polycystic kidney disease. The association of bicuspid aortic and polycystic hepatorenal cystic disease would be possible if we consider that these two pathologies are ciliopathy, however no case has been described in the literature, making it still difficult to make a decision and to make a therapeutic orientation for our patient [3].
References
- Bonny O. Swiss Medical Journal: Extrarenal involvement of polycystic kidney disease: clinical signs, screening and management. Swiss Medical Journal, 2017; 13(551): pp. 450-456.
- Karp N, Grosse-Wortmann L, Bowdin S. Severe aortic stenosis, bicuspid aortic valve and atrial septal defect in a child with Joubert Syndrome and Related Disorders (JSRD) – A case report and review of congenital heart defects reported in the human ciliopathies. European Journal of Medical Genetics, 2012; 55(11): p. 605-610.
- Hamzaoui M, et al. Cardiovascular Impairment Associated with Autosomal Dominant Polycystic Kidney Disease. Nephrology & Therapeutics, 2021; 17(1): pp. 18-29.
- Gabriel GC, Young CB, Lo CW. Role of cilia in the pathogenesis of congenital heart disease. Seminars in Cell & Developmental Biology, 2021; 110: p. 2-10.

