The value of Duplex Ultrasound Surveillance Protocol in Early Detection and Salvage of Failing Infrainguinal Bypass Vein Grafts
Mina Abdelmalak1,*, James Chapman2, Fatika Waseem3, Chris Lowe4 and Ragai Makar5
1Manchester Vascular Centre, Manchester NHS Foundation Trust, UK
2Liverpool Orthopaedics and Trauma Service, University Hospitals of Liverpool NHS Foundation Trust, School of Medicine, Faculty of Health and Life Sciences, University of Liverpool, UK
3Leeds Medical University, UK
4Vascular surgery department, Oldham NHS Foundation Trust, UK
5Vascular surgery department, Countess of Chester NHS Foundation Trust, UK
Received Date: 27/01/2026; Published Date: 06/04/2026
*Corresponding author: Mina Abdelmalak, Manchester Vascular Centre, Manchester Royal Infirmary, Oxford Road, Manchester, M13 9WL, UK
Abstract
Introduction: A Duplex Ultrasound (DUS) surveillance protocol performed at 6 weeks, 3, 6, 12, and 24 months during the first two years after infrainguinal bypass surgery with autogenous vein grafts has been shown to significantly improve the early detection and treatment of failing bypass vein grafts. This approach has led to an increase in the two-year patency rate of bypass vein grafts and a preventing of the complications associated with the recurrence of Critical Limb Ischemia (CLI).
Objective: Infrainguinal vein bypass grafting remains a cornerstone in the management of lower-limb peripheral arterial disease; however, graft stenosis and thrombosis frequently threaten long-term patency and limb salvage. Approximately 25–30% of vein grafts develop significant stenoses within the first postoperative year. Duplex Ultrasound Scanning (DUS) is considered the most accurate non-invasive tool for early detection of such lesions. Nevertheless, its routine use in structured programmes remains controversial due to cost-effectiveness and additional workload on vascular laboratories. This study aimed to evaluate the clinical impact and value of DUS surveillance in detecting and managing failing vein grafts.
Methods: A retrospective analysis was performed on all patients who underwent infrainguinal bypass surgery with autogenous vein grafts between 2016 and 2021. DUS surveillance data were reviewed to identify graft abnormalities and subsequent clinical or interventional actions, assessing whether early detection through DUS contributed to improved outcomes.
Results: Among 240 procedures, 201 (84%) used autogenous vein grafts versus 39 (16%) prosthetic grafts. The anatomical distribution comprised 150 femoropopliteal (62.5%), 70 femorodistal (29.2%), and 8 popliteo-tibial (3.3%) bypasses. DUS surveillance at 6, 12, and 24 months yielded 509 scans, with abnormalities detected in 176 (34.6%), most commonly juxta-anastomotic or graft body stenoses. Thirty-two patients were recalled for urgent graft salvage despite being asymptomatic. Interventions included balloon angioplasty (n=26), thrombolysis (n=3), and hybrid procedures (n=3); 4 grafts required repeat interventions. Grafts failure rates were higher within the first postoperative year (71.9%), consistent with the pattern of intimal hyperplasia. Of 46 patients requiring further procedures or amputations, graft-preserving interventions (37, 80.3%) significantly exceeded amputations (9, 19.6%; p < 0.001).
Conclusion: DUS surveillance effectively identifies asymptomatic failing vein grafts, facilitating early intervention and improved graft patency. A structured surveillance protocol at 6 weeks, 3, 6, 12, and 24 months is recommended, provided adequate vascular laboratory resources are available.
Keywords: Infrainguinal bypass; Vein graft; Duplex Ultrasound surveillance
Introduction
Infrainguinal vein bypass graft surgeries are commonly performed for patients with lower-limb peripheral arterial disease. Nonetheless, vein grafts may form lesions or stenoses, which decrease blood flow and lead to thrombosis. These stenoses can be detected in 25% to 30% of vein bypass grafts during the first year [1-4].
Duplex ultrasound scanning is currently the best method for detecting stenotic lesions threatening graft patency during follow-up [5]. Correcting such lesions may improve graft patency and limb salvage rates [6,7]. The literature regarding the value and benefits of Duplex Ultrasound (DUS) surveillance protocols remains controversial. Ongoing debate centres on whether such protocols provide meaningful clinical benefit by facilitating early detection and intervention for failing vein grafts, improving long-term graft patency, or whether they impose an undue burden on vascular services with questionable cost-effectiveness.
Cost-effectiveness is a crucial consideration within the current healthcare system [8,9]. Duplex surveillance programmes are costly to establish and maintain, given the initial expenditure for the equipment, hiring a trained vascular technologist, and funding for the additional interventions performed [9]. It is estimated that if Duplex surveillance of all vein grafts could prevent 5% of patients from requiring an amputation, the savings would be substantial enough to warrant the costs of setting up a surveillance programme [10].
Although amputation is the most clinically relevant measure of graft failure, graft occlusion does not necessarily lead to amputation [12]. Unfortunately, the few reports published [11,13-15] tend to argue in favour of duplex surveillance based solely on patency without consideration for limb salvage.
Methods
This is a single-centre retrospective analysis of a clinical cohort of patients treated with infrainguinal bypass surgery from 2016 to the first quarter of 2021. The study evaluates the value of DUS graft surveillance in detecting early stenotic lesions in the bypass graft, either in the graft body or juxta-anastomotic, and offers a consequent intervention based on the scan findings.
The study population consists of all patients who underwent an infra-inguinal bypass operation using an autogenous vein during this period. Data on demographic details, clinical presentation, revascularisation interventions, abnormal findings on DUS scans, and graft salvage interventions and their outcomes were recorded. Additionally, a concomitant quality improvement project was started to identify patient compliance with the surveillance protocol and whether any factors lead to missed scan invitations or between the main operating hub and the spoke sites.
The current surveillance protocol includes scans every 6 weeks, followed by intervals at 3, 6, 12, 18, and 24 months unless unforeseen findings require a different follow-up pattern. Patients with synthetic grafts used during bypass surgery are excluded, as are all patients deemed unsuitable for surveillance scans for various reasons, such as death or occluded grafts. Surveillance scans were performed by UK-accredited vascular sonographers within the local vascular laboratory.
Data on the clinical course and outcomes were extracted to create a custom database for analysis. The main goal was to identify stenotic lesions that could threaten the patency of the graft. We aimed to determine if identifying these lesions early led to effective interventions to prevent graft thrombosis and possible limb loss. Patients were divided into two groups: those with Doppler Ultrasound (DUS) reports showing positive findings of mild, moderate, or severe stenotic lesions in the graft body or juxta-anastomotic and those with normal scan results. We then examined the characteristics of each group.
Statistical analysis was performed with Stats Direct statistical software (Stats Direct Ltd, Merseyside, UK). Scale variables were expressed as median ± range and categorical parameters as absolute count and percentage. The Shapiro-Wilk test was applied to determine data normality. Where data were categorical, they were compared using the Fisher exact two-sided test and where continuous, a two-sample t-test was used for normal data and a Mann-Whitney U test for non-normal data. Statistical significance was accepted at the P <0.05 level.
The study was regarded as an audit by the Hospital Trust’s Research and Development team and registered with the audit department (audit number 24483; August 2021).
Results
During the study period, 240 infrainguinal bypass surgeries were performed. This included 150 femoropopliteal bypasses, 70 femorotibial bypasses, and eight popliteotibial bypasses. Additionally, four patients underwent femoro-femoral bypasses, three had SFA to tibial bypasses, and four received pop-pedal bypasses. One patient had a mid-SFA to below-knee popliteal bypass (Table 1). Of the surgeries, 201 used autogenous vein grafts, while 39 used synthetic grafts, which were excluded from analysis.
A total of 509 DUS scans were performed. Positive findings included juxta-anastomotic stenosis (either proximal or distal) and/or graft body stenosis of varying degrees, with 176 out of 509 scans (34.57%) showing these abnormalities. Additionally, 39 grafts (19.4% of the total vein grafts) were identified as “failing grafts” due to severe stenosis of 70% or more of the graft lumen or peak systolic velocity >300cm/sec, which threatened their patency, prompting the need for urgent interventions (Tables 2 and 3).
Due to the graft surveillance program, 39 out of 201 patients (19.4%) have been offered follow-up procedures to salvage bypass vein grafts diagnosed with impending failure caused by severe stenotic lesions (Table 3). Of these, 37 grafts were treated with balloon angioplasty, 1 with thrombolysis, and 1 with a hybrid procedure required to repair a graft rupture during angioplasty (Figure 1). Additionally, four grafts were treated twice at different times. The interventions achieved technical success on the day and kept bypass grafts running as proved on the next follow-up scans in 32 out of the 39 grafts (82.05%).
The bar chart illustrates the distribution of different graft salvage interventions performed. Angioplasty was the most frequently used intervention (N = 37), whereas thrombolysis and hybrid procedures (angioplasty combined with open repair) were less commonly performed, each accounting for only one case (N = 1).
The re-intervention rate was higher in the first year of surveillance, consistent with the pathophysiology of intimal hyperplasia, which has its peak incidence within the first year of bypass surgery. Twelve versus six (66.6%) interventions were performed in the first and second years of DUS graft surveillance.
This study has limitations, notably the absence of a control group and no clinical surveillance protocol to monitor circulation improvement after bypass vein graft operations using vascular lab tests and clinical findings, such as Ankle-Brachial Pressure Index (ABPI), Toe Pressure (TP), Transcutaneous Pressure of Oxygen (TCPO2), and the healing of ulcers or resolution of rest pain. However, among the 46 patients who required intervention or amputation and participated in a surveillance programme, significantly more received graft-saving interventions than those who underwent amputation. This suggests that surveillance may provide benefits by enabling the early identification of graft issues, indicating that these differences are significant (Table 4).
The findings indicate a moderately positive relationship between positive results on the DUS surveillance scans and subsequent interventions at various time points: at 6 weeks (Φ = 0.334, p < .001), at 3 months (Φ = 0.362, p < .001), and at 6 months (Φ = 0.336, p < .001) (Figure 2).
At the 2-year mark, there is a strong correlation between positive findings in DUS surveillance scans and the performance of interventions (Φ = 0.498, p<.001) (Figure 2). This indicates that when a patient shows positive findings, we typically take action to perform interventions aimed at salvaging the graft. While no control group exists for comparison, this suggests that our practice effectively responds to surveillance findings. This implies that regular surveillance might be a crucial follow-up method to prevent graft failure and the need for repeat procedures or limb loss.
The graph illustrates the correlation between positive DUS surveillance findings and interventions at different times. A moderate correlation was observed at 6 weeks (Φ = 0.334, p < .001), 3 months (Φ = 0.362, p < .001), and 6 months (Φ = 0.336, p < .001). At 2 years, the correlation was stronger (Φ = 0.498, p < .001), suggesting that surveillance findings frequently lead to intervention and may help prevent graft failure.
Table 1: Anatomical Configurations of Infrainguinal Bypass Surgeries and Their Distribution (N = 240).
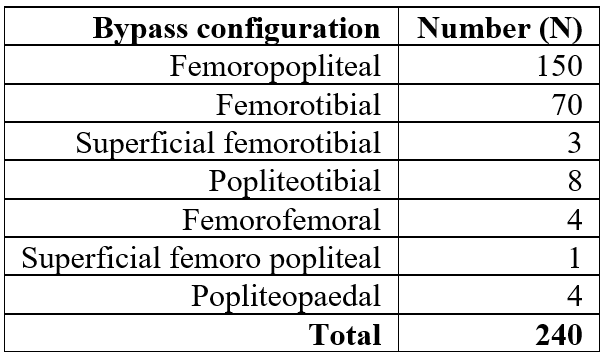
Table 2: Findings of DUS Surveillance Scans.

Note: DUS = Duplex Ultrasound Surveillance. Significant findings refer to abnormalities detected in the anastomosis or graft body, which may require further clinical intervention.
Table 3: Findings of DUS Surveillance Scans per Graft.

Note: DUS = Duplex Ultrasound Surveillance. Significant findings refer to abnormalities detected in the graft that necessitate clinical intervention.
Table 4: Impact of Surveillance on Graft-Saving Interventions and Amputation Outcomes.
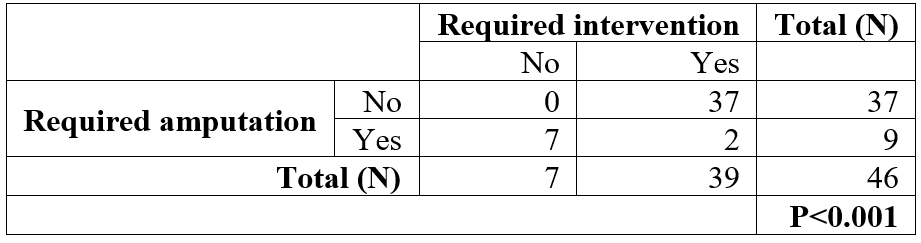
Note: P < 0.001, indicating a statistically significant difference in outcomes. Surveillance appears to facilitate early intervention, potentially reducing the need for amputation.
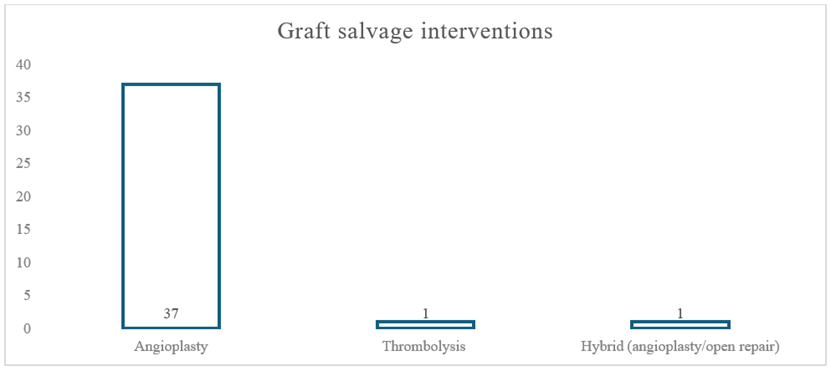
Figure 1: Graft Salvage Interventions.
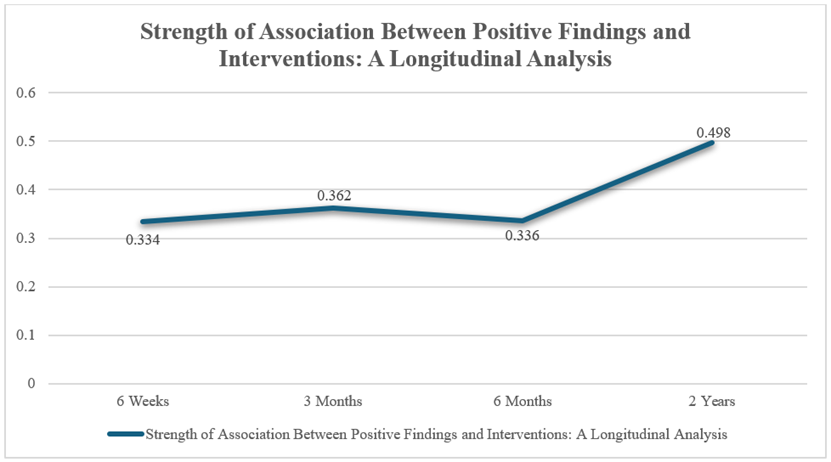
Figure 2: Strength of Association Between Positive DUS Surveillance Findings and Subsequent.
Discussion
The literature examines DUS surveillance for infrainguinal bypass vein grafts. It highlights conflicting viewpoints about its effectiveness in detecting early graft failure and its impact on the healthcare system, including the costs and workload for vascular laboratories and specialists.
Golledge et al [5] assessed infrainguinal vein graft outcomes based on studies that provided occlusion rates, comparing 2,680 duplex surveillance patients with 3,969 non-surveillance patients. The levels of distal anastomosis and the presence of critical ischaemia were found to be similar in both groups. However, only six of the 17 studies reported amputation rates; only two were Randomised Controlled Trials (RCTs), and both were small.
In 2005, Davies et al [16] published the Principal Results of the Vein Graft Surveillance Randomised Trial (VGST) [16], which was a multicentre, prospective, randomised, controlled trial involving a total of 594 patients with a patent vein graft at 30 days after surgery. These patients were randomised to either a clinical or duplex follow-up programme at six weeks and subsequently at 3, 6, 9, 12, and 18 months postoperatively. The clinical and duplex surveillance groups exhibited similar amputation rates (7% for each group) and vascular mortality rates (3% versus 4%) over 18 months. However, more patients in the clinical group experienced vein graft stenosis at 18 months (19% versus 12%, P=0.04). Nevertheless, the primary patency, primary assisted patency, and secondary patency rates were comparable in the clinical group (69%, 76%, and 80%) and the duplex group (67%, 76%, and 79%). There were no significant differences in health-related quality of life; however, the average health service costs incurred by the duplex surveillance programme were higher, at £495 (95% CI £183 to £807) per patient [16]. Based on these findings, the trial concluded that intensive surveillance with duplex scanning did not provide any additional benefit in limb salvage rates for patients undergoing vein bypass graft operations, albeit at an increased cost.
The actual DUS surveillance cost at our hospital may be lower than reported in previous studies [16]. Outpatient slots for surveillance scans incur an opportunity cost of over £100, potentially displacing other vascular patients in need. Currently, post-bypass patients are usually reviewed once at six weeks after surgery and then discharged unless they exhibit symptoms. Without regular DUS surveillance, many may not return until they experience graft failure, which risks irreversible damage and loss of the bypass. Notably, treating an occluded graft is often more expensive and complex than regular DUS surveillance, both financially and in terms of patient health outcomes.
In 2017, Abu Dabrh et al [17] published a systematic review and meta-analysis of duplex ultrasound surveillance for infrainguinal vein bypass grafts, including 15 studies. The meta-analysis indicated that DUS surveillance was not associated with a significant change in primary, secondary, or assisted primary patency or mortality compared to ankle-brachial index and clinical examination. Furthermore, DUS surveillance was linked to a non-statistically significant reduction in the amputation rate (odds ratio, 0.70 [95% confidence interval, 0.23-2.13]). The quality of evidence was deemed low due to imprecision (small number of events and wide confidence intervals) and a high risk of bias in the primary literature. Consequently, the meta-analysis concluded that a recommendation for routine DUS surveillance of infrainguinal vein grafts remains reliant on low-quality evidence.
[17] DUS provides an opportunity for early intervention. Due to its non-invasive nature and low cost, vascular surgeons may utilise DUS to individualise the follow-up of lower extremity vein grafts [17].
On the other hand, the Society for Vascular Surgery (SVS) practice guidelines on follow-up after arterial vascular surgery procedures, published in 2018, state that “Based on the high prevalence of abnormalities detected by DUS, as well as the relatively low associated costs and risks, we recommend clinical examination, ABI, and DUS for infrainguinal vein graft surveillance. This should include an early postoperative baseline evaluation and follow-ups at 3, 6, and 12 months and at least annually thereafter. More frequent surveillance may be considered when uncorrected abnormalities are identified on DUS or when alternative vein conduits (other than great saphenous vein) are used," with a strength of recommendation of 1 (Strong) and a quality of evidence of B (Moderate) [18].
DUS surveillance effectively detects stenoses and aids in graft occlusion prevention, though it carries a risk of overtreatment. Mills et al [4] found that critical stenoses, which involve over 70% narrowing of the graft diameter, have about an 80% chance of blockage, while many intermediate lesions remain stable with careful monitoring. This presents a dilemma: weighing early treatment benefits against exposing patients to unnecessary risk. Treating stable stenoses can lead to avoidable risks and increased healthcare costs, often without clear benefits for patients. Anatomical findings do not always reflect how serious the problem is, leading to unnecessary interventions. To improve outcomes, it is helpful to include functional tests during DUS, like measuring Peak Systolic Velocity (PSV) ratios and comparing these with clinical signs, such as Ankle-Brachial Index (ABI) changes. A focused approach, treating only high-risk or symptomatic patients, may help reduce overtreatment while keeping surveillance effective.
In our experience, Duplex Ultrasound (DUS) surveillance was effective in identifying failing grafts with asymptomatic but hemodynamically significant stenotic lesions that were likely to result in imminent graft failure. Early detection through routine surveillance allowed for timely intervention, potentially preventing graft thrombosis and mitigating the associated complications of Peripheral Vascular Disease (PVD). Without the implementation of the current DUS protocol—including defined surveillance intervals and structured reporting—up to 32 grafts in our cohort may have progressed to occlusion.
Study limitations
The main limitations of this study are that it is a single-centre experience with no control group for comparison, specifically regarding clinical follow-up, as the DUS surveillance programme with its multiple scan regimen has already been implemented in the unit in collaboration with the vascular laboratory. Furthermore, during data retrieval, several patients were found to be non-compliant with the surveillance programme for various reasons, including the COVID-19 period, missed scan appointments, missing invitations for scans, or other factors.
Our data shows that several patients had DUS scans indicating significant stenosis, but timely action was not taken for various reasons. As a result, 4 grafts ended up occluding. These findings highlight the importance of regular and proactive DUS monitoring to identify and manage stenosis before it leads to graft failure. This evidence further supports the need for DUS surveillance, even without a formal control group.
Quality Improvement Project (QIP)
Following the identification of several patients who did not fully enrol in the DUS surveillance programme for various reasons between the main hub and the spoke sites, a quality improvement programme was conducted to audit the compliance and attendance of infrainguinal bypass vein graft patients in the first two years following the operation. Furthermore, a dedicated spreadsheet was created to include bypass operations, automatically generating the dates for surveillance scans and highlighting them when they are due. This has been overseen by a surveillance coordinator, who acts on any missing scans to minimise non-compliance with the programme.
Conclusion
DUS graft surveillance has proven highly valuable in detecting and salvaging asymptomatic failing bypass vein grafts early. Whenever vascular laboratory staffing and facilities are available, DUS surveillance is recommended at 6 weeks, 3, 6, 12, and 24 months after bypass operation. This allows for timely salvage interventions and decreases the incidence and complications of recurrent critical limb ischaemia symptoms following occluded bypass grafts.
Presented at the following meetings:
Charing Cross Vascular Symposium 2021
ESVS annual meeting 2024
Funding & Conflicts of Interest: No declaration
References
- Sayers RD, Raptis S, Berce M, Miller JH. Long-term results of femorotibial bypass with vein or polytetrafluoroethylene. Br J Surg,1998; 85: 934-938.
- Moody P, DeCossart LM, Douglas HM, Harris PL. Asymptomatic strictures in femoropopliteal vein grafts. Eur J Vasc Surg,1989; 3: 737-740.
- Szilagyi DE, Elliott JP, Hageman JH, Smith RF, Dall’Olmo CA. The biologic fate of autogenous vein implants as arterial substitutes: clinical, angiographic and histopathologic observations in femoropopliteal operations for atherosclerosis. Ann Surg,1973; 178: 232-246.
- Mills JL. Mechanisms of vein graft failure: the location, distribution, and characteristics of lesions that predispose to graft failure. Semin Vasc Surg,1993; 6: 78-91.
- Golledge J, Beattie DK, Greenhalgh RM, Davies AH. Have the results of infrainguinal bypass improved with the widespread utilisation of postoperative surveillance? Eur J Vasc Endovasc Surg,1996; 11: 388-392.
- Davies AH, Magee TR, Tennant SGW, Baird RN, Horrocks M. Criteria for identification of the “at risk” infrainguinal bypass graft. Eur J Vasc Surg,1994; 8: 315-319.
- Moody P, Gould DA, Harris PL. Vein graft surveillance improves patency in femoropopliteal bypass. Eur J Vasc Surg, 1990; 4: 117-121.
- Gibbs RGJ, Beattie DK, Greenhalgh RM, Davies AH. Vein graft surveillance: current trends. Br J Surg,1997; 84: 63.
- Grigg M, Nicolaides AN, Wolfe JH. Can postoperative surveillance of femoro-distal vein grafts be justified? In: Greenhalgh RM, Jamieson CW, Nicolaides AN, editors. Limb Salvage and Amputation in Vascular Disease. Philadelphia (PA): WB Saunders, 1988; p. 259-270.
- Davies AH, Hawdon AJ, Sydes MR, Thompson SG. Is duplex surveillance of value after leg vein bypass grafting? Principal results of the Vein Graft Surveillance Randomised Trial (VGST). Circulation, 2005; 112(13): 1985-1991.
- Berkowitz HD, Greenstein SM. Improved patency in reversed femoral-infrapopliteal autogenous vein grafts by early detection and treatment of the failing graft. J Vasc Surg,1987; 5: 755-761.
- Nehler MR, Moneta GL, Yeager RA, Edwards JM, Taylor LMJ, Porter JM. Surgical treatment of threatened reversed infrainguinal vein grafts. J Vasc Surg,1994; 20: 558-563.
- Nielsen TG. Natural history of infrainguinal vein bypass stenoses: early lesions increase the risk of thrombosis. Eur J Vasc Endovasc Surg,1996; 12: 60-64.
- Davies AH, Hawdon AJ, Sydes MR, Thompson SG. Is duplex surveillance of value after leg vein bypass grafting? Principal results of the Vein Graft Surveillance Randomised Trial (VGST). Circulation,2005; 112: 1985-1991.
- Mohammed K, Farah W, Haydour Q, Zierler RE, Wang Z, Prokop LJ, et al. Systematic review and meta-analysis of duplex ultrasound surveillance for infrainguinal vein bypass grafts. J Vasc Surg,2017; 66: 1885-1891.
- Zierler RE, Jordan WD, Lal BK, Mussa F, Leers S, Fulton J, et al. The Society for Vascular Surgery practice guidelines on follow-up after vascular surgery arterial procedures. J Vasc Surg,2018; 68: 256-284. doi: 10.1016/j.jvs.2018.04.018.

