Influence of Chlorhexidine-Modified Dentin Primers on Glass-Fiber Post Bond Strength After Aging
Fernanda Calvo Costa1,*, Simone Yumi Sudo Canineo1, Tarcisio José Arruda Paes Junior2, Rubens Nisie Tango2 and Paula Carolina Komori de Carvalho2
1Master's Program in Science and Technology applied to Dentistry, São Paulo State University - UNESP, Sao Jose dos Campos 12245-000, SP, Brazil
2Department of Dental Materials and Prosthodontics, Institute of Science and Technology, Sao Paulo State University—UNESP, Sao Jose dos Campos 12245-000, SP, Brazil
Received Date: 22/01/2026; Published Date: 20/03/2026
*Corresponding author: Fernanda Calvo Costa, Master's Program in Science and Technology applied to Dentistry, São Paulo State University - UNESP, Sao Jose dos Campos 12245-000, SP, Brazil
Abstract
This study evaluated the effect of chlorhexidine digluconate (CHX)-modified dentin primers on the bond strength of glass fiber posts, both immediately and after thermo-mechanical aging. Sixty bovine incisors were endodontically treated and randomly assigned to three groups: control (etch-and-rinse adhesive), 0.12% CHX-modified primer in alcoholic solution, and 0.12% CHX-modified primer in aqueous solution. Glass fiber posts were cemented with dual-cure resin cement. Half of the specimens were subjected to thermo-mechanical aging (1,200,000 cycles; 5 °C/55 °C; 88 N load). All samples were sectioned into 2 mm slices at cervical, middle, and apical thirds and tested for push-out bond strength. Fracture modes were examined under a stereomicroscope. Data were analyzed with three-way ANOVA and Tukey’s test (α = 0.05). Bond strength was significantly influenced by root depth (P < 0.001) and by primer × aging interaction (P = 0.018). Cervical slices showed higher values than middle and apical thirds. In the immediate condition, CHX in alcoholic solution promoted higher bond strength compared to the control, but after aging no significant differences were observed among groups (P > 0.05).
The incorporation of 0.12% CHX into etch-and-rinse primers did not compromise the bond strength of glass fiber posts to radicular dentin. However, root depth strongly influenced adhesion, with lower values in the apical region.
Keywords: Chlorhexidine; Dental Bonding; Dentin; Dentin-Bonding Agents; Post and core technique
Introduction
Metal posts have been used as the gold standard to restore endodontically treated teeth with large structural loss. However, they are unesthetic and can lead to catastrophic fracture of the structure due to their high elastic modulus [1-2].
Moreover, esthetic considerations have become increasingly important when selecting intra-radicular posts. Metal posts, due to their opacity, can cause a shadowing effect at the gingival margin, particularly in the anterior region or in patients with a thin gingival phenotype. This effect is exacerbated by modern metal-free restorative materials, which are more translucent and may fail to mask the underlying dark structure. Glass fiber posts, being more translucent and dentin-colored, offer superior esthetics and reduce the likelihood of visual compromise in the final restoration.
Glass fiber posts provide enhanced esthetics, resulting from their high translucency and compatibility with resin cement shade [2,3]. Furthermore, a more homogeneous stress distribution is achieved with these posts [4-7], minimizing the risk of vertical tooth fractures [1,8]. However, debonding is the most common failure, resulting in complete dislodging from the root canal [9-11]. Recent finite element analyses have also shown that, in challenging clinical scenarios such as noncircular canals and the absence of a ferrule, different post and core materials significantly influence stress distribution and the risk of debonding [12].
Bonding to root dentin represents a significant challenge due to its distinct morphology and the technical difficulties associated with resin bonding in this region, including limited light penetration for efficient curing [13]. Additionally, the presence of residual gutta-percha further complicates the process by reducing the surface area available for bonding [5]. These factors highlight the importance of optimizing bonding techniques and materials to enhance adhesion in this critical area.
The degradation of the adhesive layer can be catalyzed by endogenous enzymes, metalloproteinases (MMP) [3,14-15], present in saliva and in the extracellular matrix of human cells, which are involved in metabolic activity, remodeling and degradation of several types of collagens. Besides MMP, the cathepsins, which are cysteine proteases and also function as hydrolases, are enzymes that hydrolyze peptide or ester bonds in the presence of water [16,17]. They are expressed during the synthesis of the extracellular matrix and become encapsulated in dentin tissue after the mineralization process is finished, remaining inactive [18-19], but can be activated if, for some reason, the dentin is demineralized [14,20], for example, by the use of adhesive systems applied after total acid etching ("total etch") or even with self-etching adhesives [21]. Chlorhexidine Digluconate (CHX), a synthetic protease inhibitor, effectively prevents the degradation of collagen matrices by chelating metal ions, thereby stopping the activation of the degrading enzymatic function [3,22-23].
Recent studies have investigated encapsulated forms of chlorhexidine incorporated into adhesive systems, aiming for sustained release and prolonged enzymatic inhibition, thereby enhancing the longevity of dentin bonding [24].
As few studies have investigated the benefits of CHX on the bond strength (BS) to radicular dentin [3], this study aimed to evaluate the influence of a CHX-modified etch-and-rinse adhesive system on glass fiber post bond strength to root dentin upon thermo-mechanical artificial aging.
Therefore, the null hypothesis tested was that the incorporation of 0.12% chlorhexidine digluconate, either in aqueous or alcoholic solution, into dentin primers has no effect on the bond strength of glass fiber posts to radicular dentin, irrespective of root depth or thermo-mechanical aging.
Methods
Sixty fresh bovine incisors were stored in buffered 0.1% thymol until cleaning. The tooth crowns were removed using a double-faced diamond disc (KG Sorensen, Cotia-SP, Brazil), under water cooling, and the root length was standardized to 15 mm. The root canals were prepared with Gates-Glidden No. 3, with a working length of 12 mm.
The dentin was rinsed with 20 mL of saline solution, acid etched (37 % phosphoric acid, Dentsply, Petrópolis - RJ, Brazil) for 15 seconds, rinsed with water for 15 seconds and dried with absorbent paper cones. In the control group, two consecutive layers of primer (Adper Scotchbond Multi-Purpose Adhesive System, 3M ESPE, St. Paul-MN, USA) were applied with a disposable micro-brush (KG Sorensen) and were air-dried for 5 seconds. Two layers of adhesive (Adper Scotchbond Multi-Purpose Adhesive System) were applied using a disposable micro-brush (KG Sorensen, Cotia) for 1 minute and air-dried for 5 seconds, followed by light-curing for 20 s (1400 mW/cm2, Valo, Ultradent, USA).
Modified primers were prepared by adding 5 μl of either 50% CHX aqueous solution (Byofórmula, São José dos Campos-SP, Brazil) or 50% CHX alcoholic solution (Byofórmula, Brazil) to 995 μl of Adper Scotchbond Multi-Purpose Adhesive System primer (3M ESPE) to obtain 0.12 % CHX – modified primers. These primers were applied as in the control group.
AllCem CORE dual resin cement (FGM, Joinville-SC, Brazil) was introduced into the canal using self-mixing tips and an intracanal dispenser. The glass fiber post (White Post DC # 3, FGM, Joinville-SC, Brazil) was silanated, inserted into the canal and an axial load 750 g was applied during light-curing (120 s, Valo). After storage (37°C in water in a dark container) during 24 hours after polymerization, half of the specimens were prepared for push-out bond strength (BS) test. Slices from different root levels: cervical; mid-section and apical were obtained in a cutting machine (Isomet, Buehler, Lake Bluff, USA). Each slice was mounted in a push-out testing device in a universal testing machine (EMIC DL1000, EMIC, São José dos Pinhais, PR, Brazil) and were tested with 1 mm diameter metal tip at a crosshead speed of 1 mm/min until fracture.
Silicone matrices shaped as upper canines (8 mm in height) were used to buildup resin composite crowns (Filtek Z350, 3M ESPE, Sumaré - SP, Brazil) on the remainder specimens. The roots were coated with wax (N° 7), up to 2 mm of the cervical margin. The teeth were embedded in self-cured acrylic resin in PVC molds. After polymerization, the wax was removed, and a polyether impression material (Impregum Soft, 3M ESPE) was applied to simulate the periodontal ligament.
The specimens underwent thermomechanical cycling using the Thermomechanical Wear Equipment (Erios Equipamentos, São Paulo-SP, Brazil) under the following conditions: a load of 88 N at a frequency of 3.8 Hz for 1,200,000 cycles, with temperature variations between 5 °C and 55 °C, with 60 seconds of immersion and 30 seconds of dwell time. The load was applied using a metallic spherical tip with a diameter of 4 mm, positioned on the lingual surface 3 mm from the incisal edge. Following the aging process, the specimens were sectioned and subsequently tested under the same conditions as the control group.
The fractured surfaces were analyzed in a Stereomicroscope (20 x, Discovery V20-Zeiss, Munich, Germany) and were classified as adhesive, cohesive and mixed fracture [25-26].
The results of BS (δ) were transformed into MPa using the following formula:
δ = C [load for sample rupture (kgf)] / A [interface area (mm2)]
The calculation of the adhesive area (A) was performed according to the following formula [27]:
A = π.g (generatrix of the trunk). [R1 (radius of the smallest base) +R2 (radius of the largest base)]
For the calculation of g, the following formula was used:
g2 = h2 (section height) + [R2-R1]2
The R1 and R2 values were obtained by measuring the internal diameters of the smallest and largest bases, respectively. These diameters and the height of the samples were measured with a digital caliper (Starret, Itu - SP, Brazil).
Bond strength data (MPa) were submitted to 3-way ANOVA and to Tukey test (Software Minitab), both with α = 0.05.
Results
To improve clarity and visual interpretation, the main findings on bond strength, categorized by root depth and treatment group, are presented in graphical representations (Figures 1, 2). Detailed statistical results are provided in the supporting information.
A three-way analysis of variance (ANOVA) revealed significant effects of root depth (P < 0.001) and a significant interaction between the primer and aging (P = 0.018), as presented in Table 1. As shown in Figure 1, bond strength was highest in the cervical third, followed by the middle third, with the lowest values observed in the apical third. The letters (A, B, C) above the bars indicate statistical differences between groups.
Table 1: Results of the 3-way (ANOVA) for BS values (MPa).
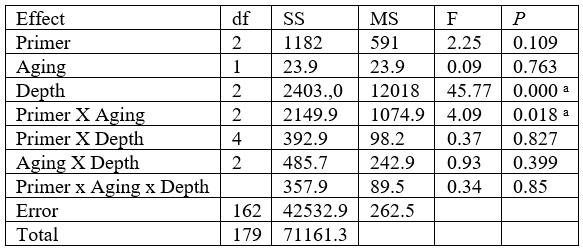
The bond strength values in this table refer to the combined results of both immediate and thermomechanically aged samples. The analysis accounts for root depth across all conditions.
BS, bond strength; df, degree of freedom; SS, sum of squares; MS, mean square.
aStatistical significance (p < 0.05).
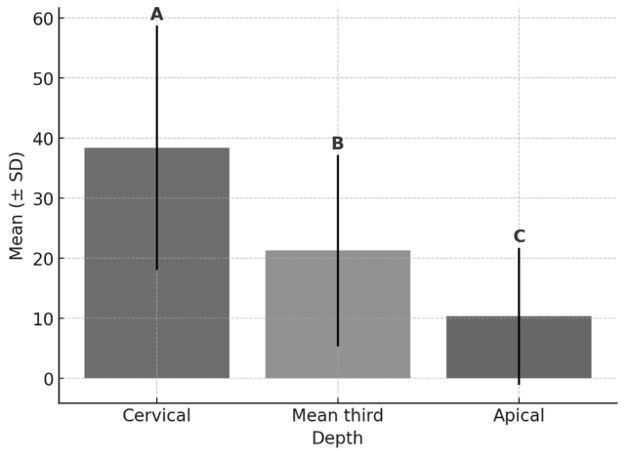
Figure 1: Results of Tukey test for comparison of BS mean values (MPa) among different depths. Bars with different letters indicate a statistically significant difference (p < 0.05).
The Figure 2 summarizes the bond strength values according to the primer x aging interaction. In the im-mediate evaluation group, the CHX-modified primer in an alcoholic solution demonstrated a significantly higher bond strength compared to the control group. However, after thermo-mechanical aging, no significant differences were detected among the primers. The letters (A, AB, B) above the bars also indicate statistical differences between groups.
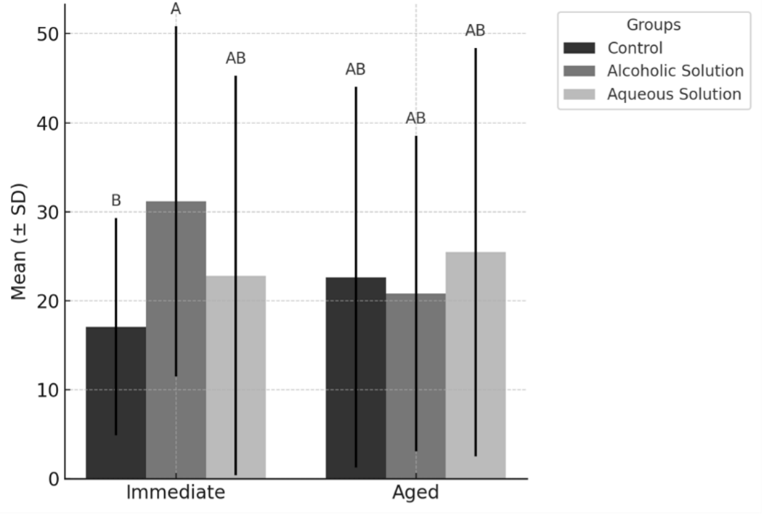
Figure 2: Results of Tukey test results for comparison of BS mean values (MPa) for primer x aging interaction. Bars with different letters indicate a statistically significant difference (p < 0.05).
Discussion
The thermomechanical aging protocol used in this study (1.2 million cycles, 88 N, 3.8 Hz) simulates approximately 5 years of clinical function based on average masticatory frequency and force reported in the literature [28]. Although rigorous, this protocol was selected to accelerate aging and reveal potential degradation not visible under short-term conditions.
Indeed, thermomechanical aging may affect both the collagen and the adhesive resin. Previous studies have shown that hydrophilic adhesive components can undergo hydrolytic degradation, leading to reduced bond strength [29]. However, since only adhesive failures were analyzed, it is likely that the interface remained the primary weak point, supporting the focus on MMP-related mechanisms.
Only adhesive failures were selected to isolate interfacial bonding effectiveness, minimizing variability introduced by cohesive or mixed fractures. This approach follows the protocol of previous studies [30] to ensure consistency and interpretability of bond strength values. Nonetheless, this limitation is acknowledged, and further studies could stratify all fracture types for a more comprehensive analysis.
According to the results of this study, the incorporation of CHX in the primer did not modify the bond strength, thus corroborating previous studies [21,22,31,33], except in the immediate group treated with CHX alcoholic solution (Figure 2). Studies have shown that ethanol as a solvent of adhesive monomers enhances adhesive penetration into collagen fibers, promoting better encapsulation [32,34-36], which may contribute to a more durable hybrid layer [37]. Furthermore, CHX in ethanol-based adhesives is relatively more resistant to displacement compared to aqueous solution [38] and does not impair bonding to dentin, resulting in a homogeneous hybrid layer [39].
Regarding the thermal-mechanical cycled groups, it can be speculated that similar bond strength values were obtained among groups due to the combined effects of resin material degradation, which gradually absorbs water and becomes more permeable, and the degradation of the exposed demineralized dentin substrate, which is also prone to hydrolysis [10]. This is in line with recent biomechanical findings indicating that post debonding remains one of the most frequent failures, particularly in cases with compromised root anatomy, as demonstrated in finite element studies on CAD–CAM posts and cores [12]. The layer of unprotected collagen fibrils, not encapsulated by mineral or monomeric reinforcement, represents the initial targets of proteolytic enzyme action (MMPs and cathepsins), which cause collagen fibril degradation and, subsequently, failure of the dentin/resin adhesive inter-face [15].
The results presented in Figure 1 corroborate previous studies showing that the most apical section exhibited the lowest bond strength values [9,20]. This region contains a high number of dentinal tubules and its proximity to the pulp chamber increases the density and diameter of the tubules, with higher humidity and lower intertubular dentin content [37,40].
Furthermore, the encapsulation of CHX in adhesive systems has been shown to enable controlled release, extending its proteolytic inhibition and offering a promising approach for improving the stability of the adhesive interface [24].
The main limitations of this study include the exclusive use of an etch-and-rinse adhesive and the high number of thermal-mechanical cycles applied. A stepwise aging protocol could be employed in future studies to detect the specific point at which bonding failure occurs. Additionally, alternative dentin treatments aimed at improving long-term bonding performance with glass fiber posts should be explored.
Conclusion
Based on the results of this in vitro study, it can be concluded that the incorporation of chlorhexidine (0.12% CHX) into dentin primers does not compromise the bond strength or long-term stability of glass fiber posts to radicular dentin following thermal-mechanical aging. Furthermore, bond strength varied according to root canal depth, with the cervical third exhibiting the highest values and the apical third the lowest, highlighting the influence of root anatomy on adhesive performance.
Conflict of interest: No potential conflict of interest relevant to this article was reported.
Funding: None
Acknowledgements: Not applied
References
- Corazza PH, di Domênico MB, Facenda JC, Merlo ÉG, Borba M, Özcan M. Fiberglass versus cast metal posts: a practical review based on mechanical properties. Braz Dent Sci, 2022; 25. doi: 10.4322/bds.2022.e3442.
- Maciel CM, Baroudi K, Oliveira KCF, dos Santos C, Lima CS, Vitti RP. Clinical longevity of metallic versus fiberglass intraradicular posts: a systematic review. Braz Dent Sci, 2024; 27. doi: 10.4322/bds.2024.e4114.
- Zhou J, Yang X, Chen L, Liu X, Ma L, Tan J. Pre-treatment of radicular dentin by self-etch primer containing chlorhexidine can improve fiber post bond durability. Dent Mater J, 2013; 32: 248–255. doi: 10.4012/dmj.2012-134.
- Munitic MS, Bago I, Glockner K, Kqiku L, Gabric D, Anic I. Effect of different laser treatments on the bond strength of intracanal fiber posts cemented with self-adhesive resin cement. J Prosthodontics, 2018; 28: 1-7. doi: 10.1111/jopr.12738.
- Cecchin D, Farina AP, Souza MA, Da Cunha Pereira C. Effect of root-canal sealer on the bond strength of fiberglass post to root dentin. Acta Odontol Scand, 2011; 69: 95–100. doi: 10.1111/j.1365-2591.2010.01831.x.
- Lindblad RM, Lassila LVJ, Salo V, Vallittu PK, Tjäderhane L. Effect of chlorhexidine on initial adhesion of fiber-reinforced post to root canal. J Dent, 2010; 38: 796–801. doi: 10.1016/j.jdent.2010.06.011.
- Lalama M, Rocha MG, O'Neil E, Zoidis P. Polyetheretherketone (PEEK) post and cores restorations: A 3D accuracy analysis between Heat-Pressed and CAD-CAM fabrication methods. J Prosthodontics, 2021; 31: 537-542. doi: 10.1111/jopr.13452.
- da Rosa RA, Barreto MS, Moraes Rdo A, Broch J, Bier CAS, Só MVR, et al. Influence of endodontic sealer composition and time of fiber post cementation on sealer adhesiveness to bovine root dentin. Braz Dent J, 2013; 24: 241–246. doi: 10.1590/0103-6440201302154.
- Ferrari M, Mannocci F, Vichi A, Cagidiaco MC, Mjör IA. Bonding to root canal: Structural characteristics of the substrate. Am J Dent, 2000; 13: 255–260.
- Carrilho MRO, Carvalho RM, Tay FR, Pashley DH. Effects of storage media on mechanical properties of adhesive systems. Am J Dent, 2004; 17: 104–108.
- Spencer P, Wang Y, Katz JL. Identification of collagen encapsulation at the dentin/adhesive interface. J Adhes Dent, 2004; 6: 91–95.
- Phattanaphirom C, Poovarodom P, Angwaravong O, Angwarawong T. Effect of different CAD-CAM post and core materials in endodontically treated teeth without ferrule and with non-circular root canals: 3D finite element analysis. J Prosthodont Res, 2025; 69(3): 394-400. https://doi.org/10.2186/jpr.JPR_D_24_00170.
- Tay FR, Loushine RJ, Lambrechts P, Weller RN, Pashley DH. Geometric factors affecting dentin bonding in root canals: A theoretical modeling approach. J Endod, 2005; 31: 584–589. doi: 10.1097/01.don.0000168891.23486.de.
- Bedran-Russo AKB, Pereira PNR, Duarte WR, Okuyama K, Yamauchi M. Removal of dentin matrix proteoglycans by trypsin digestion and its effect on dentin bonding. J Biomed Mater Res - Part B Appl Biomater, 2008; 85: 261–266. doi: 10.1002/jbm.b.30944.
- Pashley DH, Tay FR, Yiu C, Hashimoto M, Breschi L, Carvalho RM, et al. Collagen degradation by host-derived enzymes during aging. J Dent Res, 2004; 83: 216–221. doi: 10.1177/154405910408300306.
- Frassetto A, Breschi L, Turco G, Marchesi G, Di Lenarda R, Tay FR, et al. Mechanisms of degradation of the hybrid layer in adhesive dentistry and therapeutic agents to improve bond durability - A literature review. Dent Mater, 2016; 32: e41–53. doi: 10.1016/j.dental.2015.11.007.
- Hannas AR, Pereira JC, Granjeiro JM, Tjäderhane L. The role of matrix metalloproteinases in the oral environment. Acta Odontol Scand, 2007; 65: 1–13. doi: 10.1080/00016350600963640.
- Boskey AL. The role of extracellular matrix components in dentin mineralization. Crit Rev Oral Biol Med, 1991; 2: 369–387. doi: 10.1177/10454411910020030501.
- Mazzoni A, Pashley DH, Nishitani Y, Breschi L, Mannello F, Tjäderhane L, et al. Reactivation of inactivated endogenous proteolytic activities in phosphoric acid-etched dentine by etch-and-rinse adhesives. Biomater, 2006; 27: 4470–4476. doi: 10.1016/j.biomaterials.2006.01.040.
- Fukae M, Kaneko I, Tanabe T, Shimizu M. Metalloproteinases in the mineralized compartments of porcine dentine as detected by substrate-gel electrophoresis. Arch Oral Biol, 1991; 36: 567–573. doi: 10.1016/0003-9969(91)90106-5.
- Zhou J, Tan J, Chen L, Li D, Tan Y. The incorporation of chlorhexidine in a two-step self-etching adhesive preserves dentin bond in vitro. J Dent, 2009; 37: 807–812. doi: 10.1016/j.jdent.2009.06.011.
- Breschi L, Mazzoni A, Nato F, Carrilho M, Tjäderhane L, Jr AR, et al. Chlorhexidine stabilizes the adhesive interface a two-year in vitro study. Dent Mater, 2013; 26: 1–12. doi: 10.1016/j.dental.2009.11.153.
- Osorio R, Yamauti M, Osorio E, Román JS, Toledano M. Zinc-doped dentin adhesive for collagen protection at the hybrid layer. Eur J Oral Sci, 2011; 119: 401–410. doi: 10.1111/j.1600-0722.2011.00853.x.
- Kalagi S, Feitosa SA, Münchow EA, Martins VM, Karczewski AE, Cook NB, et al. Chlorhexidine-modified nanotubes and their effects on the polymerization and bonding performance of a dental adhesive. Dent Mater, 2020; 36: 687-697. doi: 10.1016/j.dental.2020.03.007.
- Sengün A, Ünlü N, Özer F, Öztürk B. Bond strength of five current adhesives to caries-affected dentin. J Oral Rehabil, 2002; 29: 777–781. doi: 10.1046/j.1365-2842.2002.00871.x.
- Komori PC, Pashley DH, Tjäderhane L, Breschi L, Mazzoni A, de Goes MF, et al. Effect of 2% chlorhexidine digluconate on the bond strength to normal versus caries-affected dentin. Oper Dent, 2009; 34: 157-165. doi: 10.2341/08-55.
- Bitter K, Meyer-Lueckel H, Priehn K, Kanjuparambil JP, Neumann K, Kielbassa AM. Effects of luting agent and thermocycling on bond strengths to root canal dentine. Int Endod J, 2006; 39: 809–818. doi: 10.1111/j.1365-2591.2006.01155.x.
- Gale MS, Darvell BW. Thermal cycling procedures for laboratory testing of dental restorations. J Dent, 1999; 27: 89-99. doi: 10.1016/s0300-5712(98)00037-2.
- Breschi L, Mazzoni A, Ruggeri A, Cadenaro M, Di Lenarda R, De Stefano Dorigo E. Dental adhesion review: aging and stability of the bonded interface. Dent Mater, 2008; 24: 90-101. doi: 10.1016/j.dental.2007.02.009.
- Sengün A, Unlü N, Ozer F, OztUrk B. Bond strength of five current adhesives to caries-affected dentin. J Oral Rehabil, 2002; 29: 777-781. doi: 10.1046/j.1365-2842.2002.00871.x.
- Stanislawczuk R, Amaral RC, Zander-Grande C, Gagler D, Reis A, Loguercio AD. Chlorhexidine-containing Acid Conditioner Preserves the Longevity of Resin-dentin Bonds. Oper Dent, 2009; 34: 481–490. doi: 10.2341/08-016-L.
- Cecchin D, Almeida JFA, Gomes BPFA, Zaia AA, Ferraz CCR. Deproteinization technique stabilizes the adhesion of the fiberglass post relined with resin composite to root canal. J Biomed Mater Res - Part B Appl Biomater, 2012; 100: 577-583. doi: 10.1002/jbm.b.31946.
- Cheng L, Zhang K, Weir MD, Liu H, Zhou X, Xu HHK. Effects of antibacterial primers with quaternary ammonium and nano-silver on Streptococcus mutans impregnated in human dentin blocks. Dent Mater, 2013; 29: 462–472. doi: 10.1016/j.dental.2013.01.011.
- Carrilho MR, Tay FR, Donnelly AM, Agee KA, Ricardo M, Hosaka K, et al. Membrane permeability properties of dental adhesive films. J Biomed Mater Res - Part B Appl Biomater, 2009; 88: 706–721. doi: 10.1002/jbm.b.30968.
- Ito S, Saito T, Tay FR, Carvalho RM, Yoshiyama M, Pashley DH. Water content and apparent stiffness of non-caries versus caries-affected human dentin. J Biomed Mater Res - Part B Appl Biomater, 2005; 72: 109–116. doi: 10.1002/jbm.b.30130.
- Malacarne J, Carvalho RM, de Goes MF, Svizero N, Pashley DH, Tay FR, et al. Water sorption/solubility of dental adhesive resins. Dent Mater, 2006; 22: 973–980. doi: 10.1016/j.dental.2005.11.020.
- Garberoglio R, Brännström M, Scanning electron microscopic investigation of human dentinal tubules. Arch Oral Biol, 1976; 21: 355–362. doi: 10.1016/s0003-9969(76)80003-9.
- Gendron R, Grenier D, Sorsa T, Mayrand D. Inhibition of the activities of matrix metalloproteinases 2, 8, and 9 by chlorhexidine. Clin Diagn Lab Immunol, 1999; 6: 437–439. doi: 10.1128/CDLI.6.3.437-439.1999.
- Carrilho MR, Carvalho RM, Sousa EN, Nicolau J, Breschi L, Mazzoni A, et al. Substantivity of chlorhexidine to human dentin. Dent Mater, 2010; 26: 779–785. doi: 10.1016/j.dental.2010.04.002.
- Perdigão J. Dentin Bonding-Variables related to the clinical situation and the substrate treatment. Dent Mater, 2010; 26: 24–37. doi: 10.1016/j.dental.2009.11.149.

