Association Between Cannabis Use and Altered Sperm Parameters in Infertile Men
Kbirou A1,2,*, Msaddar O1, Adli O2, Daghdagh Y1,2, Moataz A1,2, Dakir M1, Debbagh A1 And Abouaieb R1,2
1Andrology Department, Ibn Rochd University Hospital, Casablanca, Morocco
2Sexual Health Laboratory, Faculty of Medicine and Pharmacy, Hassan II University, Casablanca, Morocco
Received Date: 04/12/2025; Published Date: 16/01/2026
*Corresponding author: Kbirou Adil, Andrology Department, Ibn Rochd University Hospital, Casablanca, Morocco; Sexual Health Laboratory, Faculty of Medicine and Pharmacy, Hassan II University, Casablanca, Morocco
Abstract
Introduction: Cannabis use is an increasing behavior and now represents a significant public health issue. The objective of this study is to assess the frequency of cannabis use among infertile men and to investigate its association with clinical and biological alterations in male reproductive function.
Materials and Methods: This was a descriptive and analytical cross-sectional study conducted between 2018 and 2020, including 150 men consulting for couple infertility. The data analyzed included medical and urogenital history, substance abuse habits (including cannabis use), clinical examination, scrotal Doppler ultrasound, and semen analysis.
Results: The mean age of the study population was 37.5 years (range 25–50 years). Infertility was primary in 88% of cases and had been present for an average of 7.5 years. Cannabis use was reported in 10% of patients. A history of urogenital infection was present in 9.33% of men, and a high-risk occupation was noted in 10.30%. Clinical examination and scrotal Doppler ultrasound revealed a varicocele in 42% of cases. Semen analysis was abnormal in 71% of patients: oligospermia (34%), asthenospermia (25%), and azoospermia (11.3%). Sperm abnormalities were more frequent in cannabis users, particularly in terms of concentration and motility.
Conclusion: Cannabis use, present in one out of ten infertile men in our series, appears to be associated with alterations in sperm parameters. These results underscore the need to integrate the assessment of substance use, particularly cannabis use, into the investigation of male infertility.
Keywords: Cannabis; Reproduction; Male; Sperm
Introduction
Infertility represents a global public health challenge, affecting approximately 80 million people worldwide and impacting nearly one in ten couples, whether it is primary or secondary infertility. In the past, the focus was primarily on the woman when investigating infertility in a couple, but it is now established that the male partner contributes significantly to many cases. This recognition has led to substantial development in andrological investigations, allowing for a better understanding of the male causes of infertility.
Meanwhile, the use of Cannabis sativa is growing worldwide and primarily involves men of reproductive age. Several studies—based on clinical, biological, or experimental data—suggest that cannabis could impair male fertility. Indeed, deleterious effects have been observed on sperm parameters (decreased sperm count and concentration, alterations in morphology, motility, and viability) as well as a possible disruption of the reproductive hormonal axis [1].
More specifically: meta-analyses and systematic reviews indicate that cannabis use is associated with decreased sperm concentration, morphological abnormalities, reduced sperm motility and viability, and inhibition of capacitation and fertilization capacity [2].
In animal models, exposure to the main psychoactive compound in cannabis, Delta-9-tetrahydrocannabinol (THC), has been associated with testicular atrophy, decreased spermatogenesis, and dysfunctions in sperm motility and morphology.
In addition, some data in humans report hormonal disturbances in regular cannabis users [3].
However, the results are not unanimous. Some clinical studies have not found a significant impact of cannabis on sperm concentration or count. This discrepancy could be explained by the variability of the populations studied, the patterns of use (frequency, quantity, duration), or the methodologies (self-reporting, measured parameters, statistical adjustments).
In the Moroccan context—where male infertility is too often overlooked and cannabis use remains difficult to assess due to stigma—there is a real need for original data. Our study aims to determine, within a population of men consulting for infertility in a urology department, the prevalence of cannabis use and to analyze its association with clinical, paraclinical, and sperm characteristics.
Thus, this work aims to make a local contribution to the world literature and to draw the attention of health professionals to the potential role of toxic habits — particularly cannabis — in male infertility, with a view to more complete and adapted care for the couples concerned.
Materials and Methods
This is a descriptive and analytical cross-sectional study carried out in the urology department of the Ibn Rochd University Hospital in Casablanca between January 2018 and December 2020.
The study involved 150 men seeking treatment for couple infertility, defined as the inability to conceive after at least 12 months of regular unprotected intercourse. Patients aged 18 to 55 years were included, while those with a serious systemic disease, treatment that could impair fertility, or recent testicular surgery were excluded.
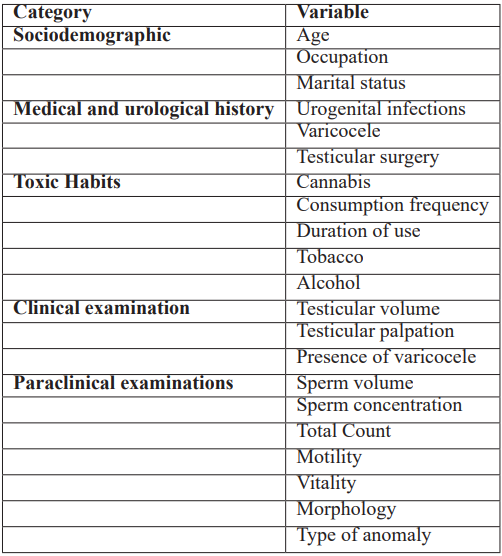
For each patient, a standardized questionnaire was used to collect sociodemographic data, medical and urological history, as well as toxic habits, in particular cannabis use (Table 1).
This was considered positive if the patient reported at least weekly use for three months.
The clinical examination included assessment of the external genitalia, testicular palpation, search for varicocele and measurement of testicular volume.
A scrotal Doppler ultrasound was performed in all patients to confirm clinical abnormalities and to assess the testicular parenchyma.
Two spermograms were performed at least three weeks apart in accordance with WHO recommendations.
The parameters analyzed were sperm volume, concentration and count, motility, vitality and morphology according to strict Kruger criteria.
The study was conducted in accordance with the confidentiality of information and the ethical principles of the Declaration of Helsinki. Informed consent was obtained from each participant.
Table 1: Variables Studied.
Results
Our study included 150 patients consulting for couple infertility. The mean age was 37.5 years (range: 25–50 years), with a majority of patients (43.33%, n = 42) in the 35–40 age group. Almost all patients were monogamous (99.33%, n = 149) and only one patient was polygamous (0.66%), with a mean marriage duration of 6.5 years (range: 2–11 years).
Infertility was primary in 88% of cases (n = 132) and secondary in 12% (n = 18), with an average duration of 6 years (range: 2–10 years).
Regarding risky habits and behaviors, 41.23% of patients were smokers, 13% were chronic cannabis users, and 7% had a chronic alcohol consumption pattern. Occupational exposure to risk was found in 12% of patients, primarily in the agricultural sector (8%).
Among the medical and urological history, 9.33% of patients had experienced a urogenital infection (orchitis or STI), 2.66% had a history of cryptorchidism, 2.66% of testicular trauma, 2% of hydrocele and 0.66% of unilateral orchiectomy following testicular torsion.
Clinical examination revealed a varicocele in 42% of patients, followed by testicular atrophy in 20% of cases and epididymal abnormalities (nodules or cysts) in 6.24% of patients. The combination of gynecomastia, small testes, hypopilescence, and micropenis was present in 1.96% of patients (Figure 1).
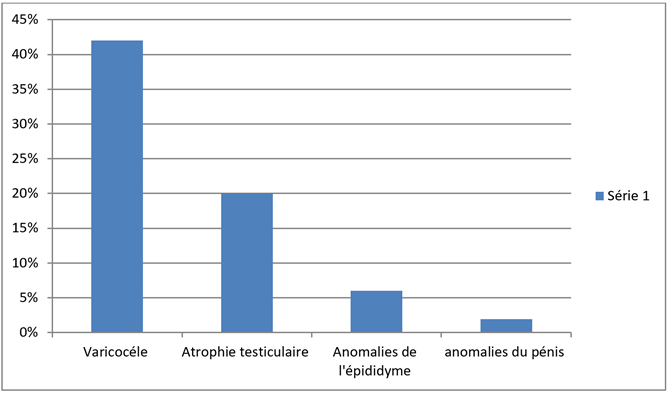
Figure 1: Clinical abnormalities.
The spermogram, interpreted according to WHO standards, was abnormal in 74% of cases. Quantitative abnormalities included oligospermia (33%) and azoospermia (13%), while qualitative abnormalities included asthenospermia (19%), teratospermia (6%) and necrospermia (3%) (Figure 2).
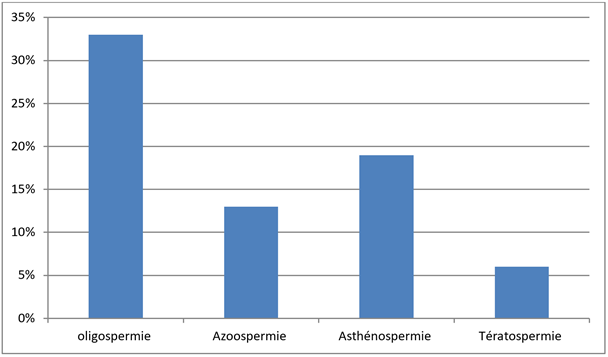
Figure 2: Sperm abnormalities.
Hormone levels (morning testosterone and plasma FSH) in patients with severe azoospermia or oligospermia were low in 1.96% of cases, and normal in the others.
Genetic testing was performed on three patients, revealing Klinefelter syndrome (XXY) in one patient.
Finally, when specifically analyzing the impact of cannabis, we observed that sperm abnormalities were more frequent among chronic users (13% of the sample), with a higher prevalence of oligospermia and asthenospermia compared to non-users.
Discussion
Specific analysis of cannabis users shows that they had a higher frequency of sperm abnormalities, particularly oligospermia and asthenospermia.
These observations are consistent with data from the literature, which indicate that the main psychoactive component of cannabis, Δ9-tetrahydrocannabinol (THC), can alter spermatogenesis and sperm function by acting on the hypothalamic-pituitary axis and on the CatSper calcium channels present in the sperm, thereby disrupting motility, capacitation and the acrosomal reaction [4-5].
Experimental studies in animals and in vitro have also demonstrated that THC can induce testicular atrophy, reduce sperm production and affect sperm morphology and viability [6].
However, some human studies have not found a significant effect on all hormonal or sperm parameters, highlighting the heterogeneity of results depending on the dose, duration of consumption and the population studied [7].
Our results also highlight the high prevalence of other classic risk factors, such as varicocele and occupational exposure, confirming that male infertility is often multifactorial.
The absence or rarity of hormonal abnormalities in our series suggests that male infertility is primarily related to local abnormalities of the testicle or genital tract rather than to systemic hormonal disorders.
Several limitations must be highlighted. The collection of cannabis consumption data relies on self-reporting, which can lead to underestimation, particularly in a social context where the subject remains taboo.
The number of consumers remains relatively small, which limits the statistical power to establish robust associations.
Furthermore, the study is cross-sectional and therefore cannot establish a causal relationship between cannabis use and infertility. Finally, we did not directly measure THC levels or those of its metabolites, which could have refined the dose-response analysis.
These results have important clinical implications. It appears essential to systematically include the assessment of lifestyle habits, including cannabis use, in the evaluation of infertile men.
Patients should be informed of the potential negative impact of cannabis on sperm quality and encouraged to modify these behaviors before any attempt at procreation.
For research, prospective multicentre studies with longitudinal follow-up, objective biological measurement of THC and analysis of advanced sperm markers (DNA fragmentation, chromatin integrity, fertilization capacity) are needed to better understand the link between cannabis use and male fertility [8].
References
- Payne KS, Mazur DJ, Hotaling JM, Pastuszak AW. Cannabis and Male Fertility: A Systematic Review. J Urol, 2019; 202(4): 674-681. doi: 10.1097/JU.0000000000000248.
- Hamed MA, Ekundina VO, Akhigbe RE. Psychoactive drugs and male fertility: impacts and mechanisms. Reprod Biol Endocrinol, 2023; 21: 69. https://doi.org/10.1186/s12958-023-01098-2
- Teixeira TA, Iori I, Andrade G, Saldiva PHN, Drevet JR, Costa EMF, et al. Marijuana Is Associated With a Hormonal Imbalance Among Several Habits Related to Male Infertility: A Retrospective Study. Front Reprod Health, 2022; 4: 820451. doi: 10.3389/frph.2022.820451.
- Gundersen TD, Jørgensen N, Andersson AM, et al. Association between use of marijuana and male reproductive hormones and semen quality: A systematic review. Hum Reprod, 2019; 34(9): 17501762.
- Rossato M, Monga M, Rago V, et al. Δ9Tetrahydrocannabinol odd human sperm function via CatSper channels. Hum Reprod, 2019; 40(5): 796807.
- Kumar S, Bansal R, Singh A. Effects of cannabinoids on male reproductive health: Evidence from experimental studies. J Clin Med, 2020; 8(5): 732.
- Mendonca FL, Braga DP, Hallak J, et al. Cannabis use and male reproductive health: Clinical and epidemiological evidence. Andrology, 2021; 9(2): 456467.
- Abd El-Ghany H, El-Sherbiny W, Mostafa T, et al. Effects of chronic cannabis exposure on sperm DNA integrity and fertility outcomes. J Clin Med, 2022; 11(12): 3850.

