Superior Efficacy of Direct-Acting Antivirals Over Interferon in HCV-Associated Mixed Cryoglobulinemia: Clinical and Virological Insights
Moffa Livia, Valeri Martina, Francesca Chiappini, Raffaele Ferri, Alessandro Di Gasbarro, Jacopo Vecchiet, Falasca Katia* and Ucciferri Claudio
Clinic of Infectious Diseases – Department of Medicine and Science of Aging, “G. d’Annunzio” University Chieti-Pescara, Italy
Received Date: 28/11/2025; Published Date: 12/01/2026
*Corresponding author: Prof. Katia Falasca, Clinic of Infectious Diseases, Dept. of Medicine and Science of Aging, “G. d’Annunzio” University, School of Medicine, Via dei Vestini, 66013 Chieti, Italy
Abstract
Background: Mixed Cryoglobulinemia (MC) is the most frequent extrahepatic manifestation of hepatitis C virus (HCV) infection. While Direct-Acting Antivirals (DAAs) have significantly improved sustained virological response (SVR) rates, their impact on MC remission—especially in patients with advanced or symptomatic disease—remains incompletely defined. This study aimed to compare the efficacy of IFN-based versus DAAS-based therapies in achieving virological and clinical remission of MC, and to identify potential negative prognostic factors.
Methods: We retrospectively analyzed 35 patients with chronic HCV infection and active MC treated with either IFN-based (n=15) or DAAs-based (n=20) regimens. Clinical response was defined as the disappearance of serum cryoglobulins and resolution of MC-related signs and symptoms. Baseline clinical and laboratory parameters were compared between responders and non-responders.
Results: Overall, 34.3% (12/35) of patients achieved clinical remission. The DAAS group showed higher response rates (45% vs. 20%) and significantly better SVR (94.7% vs. 60%) than the IFN group, despite a higher burden of comorbidities. Proteinuria and arthralgia at baseline were associated with poor outcomes. In particular, 100% of patients with proteinuria (10/10) failed to achieve remission, suggesting a strong negative predictive value. Prior IFN-based therapy and corticosteroid use were also associated with reduced response rates, though not statistically significant.
Conclusion: DAAs demonstrated superior virological and clinical efficacy compared to IFN-based regimens in patients with HCV-related MC. The presence of baseline proteinuria emerged as the most significant predictor of treatment failure, underscoring the importance of early intervention before renal involvement. Our findings support the preferential use of DAAs in this patient population and suggest that proteinuria should be carefully evaluated as a negative prognostic marker in treatment planning.
Keywords: Hepatitis; Mixed cryoglobulinemia; Proteinuria; Vasculitis; Sustained virological response
Introduction
Globally, up to 170 million individuals are chronically infected with Hepatitis C Virus (HCV), with an estimated 1.5 million new cases each year, making HCV a significant public health concern. The virus exhibits a pronounced hepatotropism and progresses to chronic infection in approximately 70% of cases. The typical disease course begins with steatosis, advances to fibrosis and cirrhosis, and may eventually lead to hepatocellular carcinoma [1-3].
In addition to liver-related complications, HCV is associated with extrahepatic manifestations in approximately 10% of infected individuals. Among these, mixed cryoglobulinemic vasculitis (MCV) is the most prevalent. Circulating cryoglobulins are found in 20–50% of HCV-positive patients, and up to 15% develop symptomatic cryoglobulinemic vasculitis [4].
Cryoglobulinemic vasculitis is an immune complex–mediated systemic vasculitis that primarily affects small- and medium-sized vessels [5]. It often involves multiple organ systems, most commonly the skin, followed by kidneys, central nervous system, joints, and vasculature. The hallmark clinical triad includes purpura (typically on the lower limbs), asthenia, and arthralgia. Severe cases may include glomerulonephritis, peripheral neuropathy, ulcers, and progression to lymphoproliferative disorders, particularly low-grade Non-Hodgkin Lymphoma (NHL). The etiological role of HCV in lymphoproliferative diseases is well-supported by epidemiological data and the clinical response to antiviral therapy in HCV-related NHL [6-11].
Early diagnosis is essential to prevent irreversible organ damage and optimize treatment. Since disease remission correlates closely with HCV clearance, the primary therapeutic goal is achieving a Sustained Virologic Response (SVR). Direct-Acting Antivirals (DAAs) have replaced interferon (IFN) and ribavirin as the standard of care due to higher efficacy and improved safety profiles [12-14]. While IFN-based therapies previously achieved SVR in only about 50% of HCV-related MCV cases, DAAs have demonstrated SVR rates close to 100% [13,15,16].
Despite this, the comparative advantage of DAAs—given their non-immunomodulatory mechanism—over IFN-based therapies in managing cryoglobulinemia remains unclear. Few studies have directly evaluated their efficacy in this context [17].
Therefore, the aim of our study was to compare the effectiveness of IFN and DAAS regimens in patients with HCV-associated cryoglobulinemia and to identify predictive factors for poor therapeutic response.
Patients and Methods
We conducted a retrospective, single-center study including 35 patients diagnosed with active HCV-related cryoglobulinemia, recruited from 2003 to 2019 at the Infectious Disease Department of SS. Annunziata Hospital in Chieti, Italy.
Inclusion Criteria
- Age ≥18 years, Informed consent, positive serum HCV RNA 6 mounts and presence of cryoglobulins in serum (symptomatic or asymptomatic)
Exclusion Criteria
- Non-active cryoglobulinemia, Co-infection with HIV or HBV, Prior treatment with rituximab or plasmapheresis
Endpoints
- Primary endpoint: Biochemical response (cryoglobulins negative) 12 weeks post-treatment.
- Secondary endpoints: Identification of clinical predictors of treatment response
Data Collection
We collected clinical, virological, and laboratory data at baseline, mid-therapy, end-of-therapy, and 12 weeks post-therapy.
Demographic data (Age, sex, nationality), Clinical data( BMI, comorbidities, HCV risk factors and medications for comorbidities), Virologic data( HCV RNA quantified by real-time PCR (Roche TaqMan CoBas), Genotyping via fluorescent automatic sequencing (Inno-LiPA), Cryocrit (cryoglobulin concentration) assessed after 7 days at 4°C), Liver and renal function test( AST, ALT, bilirubin, γ-GT, creatinine, eGFR (CKD-EPI), Complete blood count, Urinalysis (proteinuria, hematuria), Treatment regimens( DAAs group: Ombitasvir/paritaprevir/ritonavir ± dasabuvir, glecaprevir/pibrentasvir, sofosbuvir-based combinations versus IFN group: Pegylated IFN-α2a or α2b, with or without ribavirin/corticosteroids), Vasculitis evaluation: (Renal damage: proteinuria, eGFR, Cutaneous and joint involvement ( purpura, ulcers, arthralgia), Neurological symptoms as pain (VAS scale), neuropathy, Onset of hypertension or diabetes)
Statistical Analysis
Categorical variables were compared using the Chi-square test. Quantitative variables are expressed as medians with ranges. Statistical significance was defined as p < 0.05. Analyses were performed using SPSS Advanced Statistics™ v13 (Chicago, IL, USA).
Results
Of the 35 patients, 15 received IFN-based therapy (IFN cohort) and 20 received DAAs (DAAs cohort).
All were Caucasian; 29% male (n=10), 71% female (n=25). Mean age: 69.1 years (IFN) vs. 65.3 years (DAAs).
Table 1 shows the characteristics of the patients 6 months before starting HCV therapy. We identified different risk factors for HCV infection and comorbidities in Figure 1 and Figure 2. Among patients that took IFN, 73% of them took maximum 1 drug and 27% 2 or more drugs. In the DAAS cohort, 35% of patients took maximum 1 drug, instead 65% of them took 2 or more. These data were significant (p=0,014). In the DAAs group, 10% received ribavirin, and 5% corticosteroids, versus the IFN group, where 87% received ribavirin and 27% corticosteroid (Table 2).
Efficacy Outcomes
Regarding HCV treatment, SVR12 rates were obtained in 60% of the IFN group and 94.7% of the DAAs group.
Our study showed that 35% of the subject responded to treatment with 0% criocrit at 12 weeks. In detail, success rate in the DAAs cohort was 45% and 25% in the IFN cohort. Unfortunately, these data were shown not to be significant (p> 0,05). Undetectable blood cryoglobulins at the end of the therapy, showed that 50% were men and 28% were woman.
Among relapser patients, no one respond to our therapy, instead, among naïve patients 33,3% had no detectable cryoglobulins (0% cryocrit).
Regarding non-HCV related drug therapy, we noted that 100% of patients taking corticosteroids did not achieve response on cryoglobulins (p= 0,081). In addition, we looked closely to the correlation between clinical manifestations of vasculitis and response to treatment. We observed that patients that had more signs or symptoms of cryoglobulinemia responded less to therapy. Although these data were not statistically significant. Among patients that didn’t respond to therapy, 91% reported arthralgias (p≤0,04) and 100% reported neuropathy (non-significant). Among non-responder patients, 43% showed proteinuria. In the group of patients that responded no one had proteinuria (p≤0,03). Additionally, 69% of patients in the DAAs cohort who did not have proteinuria before HCV treatment achieved negative cryoglobulins at follow-up. (p≤0,02).
Table 1: Characteristics of the patients.
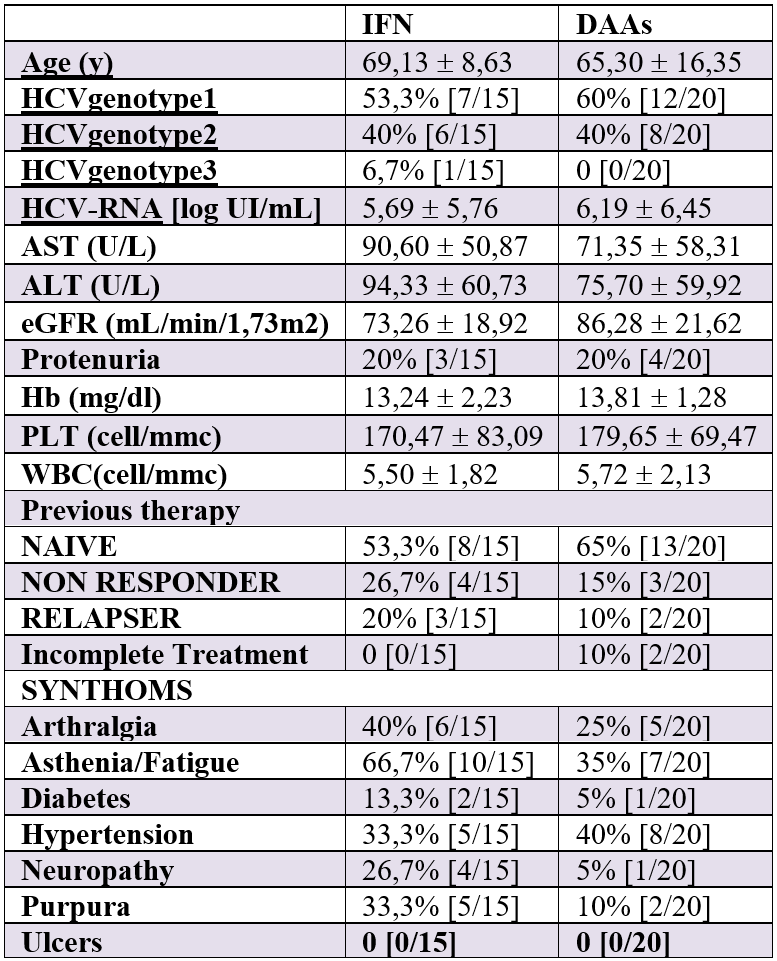
Table 2: Clinical predictors of cryoglobulin response to HCV treatment.

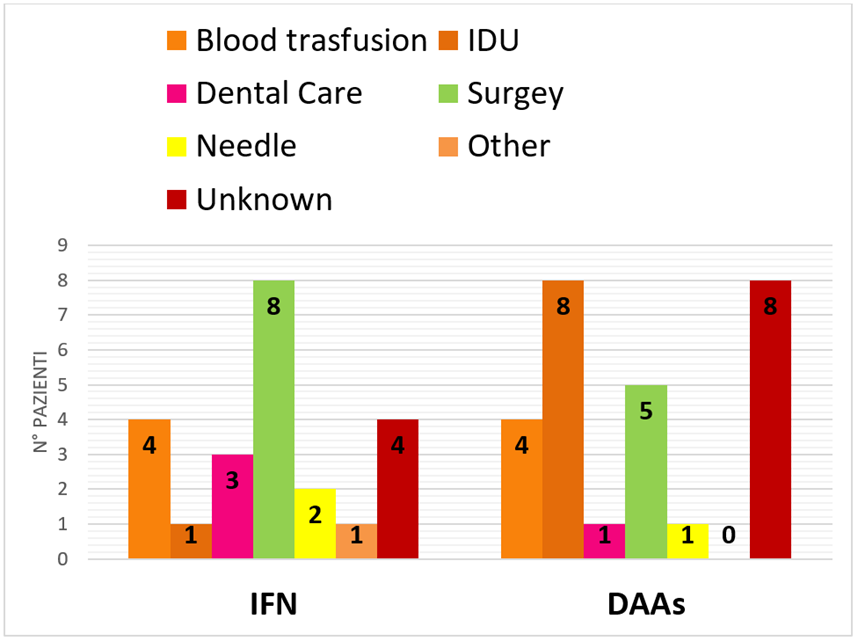
Figure 1: HCV Risk factor.
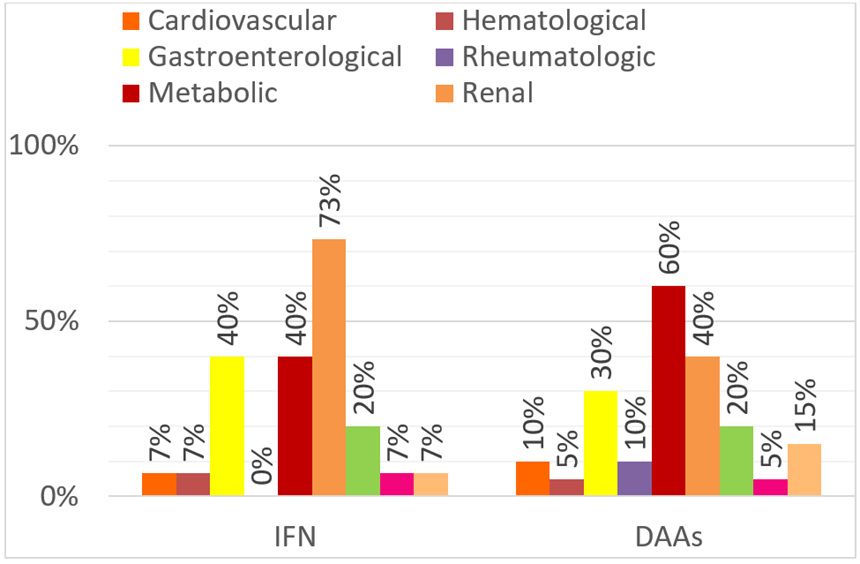
Figure 2: Comorbidities.
Discussion
Mixed Cryoglobulinemia (MC) remains the most frequent extrahepatic manifestation of Hepatitis C Virus (HCV) infection. While Direct-Acting Antivirals (DAAs) achieve Sustained Virological Response (SVR) in nearly all treated patients, cryoglobulin clearance is not guaranteed, particularly in those with advanced or symptomatic vasculitis(16). Our study confirmed a higher SVR rate with DAAs compared to interferon (IFN)-based therapy (94.7% vs. 60%), alongside a greater proportion of cryoglobulin clearance, despite a higher comorbidity burden in the DAAS cohort. These findings highlight the superior efficacy and tolerability of DAAs, consistent with previous studies demonstrating improved clinical outcomes [15,17,18].
Proteinuria and arthralgia emerged as negative prognostic markers in our cohort, with proteinuria showing a stronger predictive value. Patients with baseline renal involvement were less likely to achieve complete cryoglobulinemia remission, aligning with the VASCUVALDIC-3 study and other literature [10,19]. MC-related vasculitis is characterized by immune complex deposition in blood vessels and tissues such as the skin, joints, kidneys, and peripheral nerves. The classic clinical triad—arthralgia, asthenia, and purpura—remains the most common presentation [20-22]. Given the direct etiopathogenic role of HCV in MC, antiviral therapy remains the cornerstone of treatment. In contrast, DAAs not only improve SVR rates, but also appear to improve the remission of MC symptoms. Several authors have reported complete remission of vasculitis in approximately one-third of patients who achieved SVR12 [23], other studies have also shown that treatment with DAAs significantly improved the management of HCV-related cryoglobulins reported complete remission of vasculitis in 36% of patients who achieved SVR24, with SVR rates ranging from 74% to 100%, other study showed DAAs have significantly improved the management [24,25]. Few studies have directly compared DAAs and IFN, but the VASCUVALDIC-3 trial reported remission of vasculitis symptoms in 95.2% of patients treated with DAAs [10].
In our study of 35 patients with chronic HCV infection and active MC, 34.3% (12/35) achieved complete remission—defined as undetectable serum cryoglobulins and absence of vasculitis symptoms. Notably, 75% of responders were in the DAAS cohort and 25% in the IFN cohort. Response rates were 45% (9/20) for DAAs and 20% (3/15) for IFN.
The female-to-male ratio in our sample was 2.5:1, slightly lower than the 3:1 ratio reported in other analysis [26] While no studies were found directly comparing treatment response by sex, our data showed that 50% of males responded to therapy, compared to 28% of females—a difference that did not reach statistical significance.
Regarding treatment history, our findings suggested better outcomes in treatment-naïve patients support: none of the relapsed patients achieved remission, whereas 33.3% of naïve patients responded.
Interestingly, comorbidity burden differed between cohorts: 27% of IFN-treated patients versus 65% of DAAS-treated patients had received two or more medications before initiating antiviral therapy. Despite this, the DAAS cohort showed higher SVR and cryocrit reduction, reinforcing the superior efficacy and tolerability of DAAs [11].
Concerning adjunct therapies, 100% of patients treated with corticosteroids failed to respond to antiviral therapy (p = 0.081). This is consistent with findings from Mazzaro et al. who reported poor virological responses among corticosteroid users [27].
Overall, greater disease severity, particularly as evidenced by peripheral neuropathy or systemic vasculitis, was associated with poorer outcomes. The VASCUVALDIC-3 study reported a threefold reduction in therapy success among patients with severe vasculitis or neuropathy [7,18].
We identified two significant baseline predictors of poor treatment response: arthralgia and proteinuria. Among patients with arthralgia, 91% did not achieve remission. However, the most striking finding was the association between proteinuria and treatment failure: 100% of patients with proteinuria (10/10) failed to respond, irrespective of treatment type. This was also true for patients in the DAAS cohort, suggesting that proteinuria is a robust negative prognostic factor—stronger even than arthralgia.
Conclusion
Our study reinforces evidence that DAAs outperform IFN-based regimens in both virological and clinical outcomes for HCV-related MC. Despite a higher comorbidity burden, DAAS-treated patients achieved superior SVR rates and cryoglobulin clearance. Baseline proteinuria and arthralgia were associated with poor response, with proteinuria emerging as a particularly strong negative predictor. Prior IFN therapy and corticosteroid use were also linked to reduced efficacy, although not always reaching statistical significance. These findings emphasize the need for early diagnosis and prompt initiation of DAAS therapy, particularly before the onset of renal involvement, to improve patient outcomes in HCV-related mixed cryoglobulinemia.
Acknowledgement: All authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding.
Declaration: The study was conducted in accordance with the Declaration of Helsinki, all patients provided written informed consent to participate in the study
Data Availability Statement: The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Conflicts of Interest: The authors declare no conflict of interest.
References
- Webster DP, Klenerman P, Dusheiko GM. Hepatitis C. Lancet, 2015; 385(9973): 1124-1135.
- Falasca K, Ucciferri C, Dalessandro M, Zingariello P, Mancino P, Petrarca C, et al. Cytokine patterns correlate with liver damage in patients with chronic hepatitis B and C. Ann Clin Lab Sci, 2006; 36(2):144-150.
- Li J, Zhou J, Li P, Wang Y, Ridderhof N, Al-Tawfiq JA, et al. The global prevalence and impact of steatotic liver disease and viral infections: A systematic review and meta-analysis. Hepatol Commun, 2025; 9(5).
- Schamberg NJ, Lake-Bakaar GV. Hepatitis C Virus-related Mixed Cryoglobulinemia: Pathogenesis, Clinica Manifestations, and New Therapies. Gastroenterol Hepatol (N Y), 2007; 3(9): 695-703.
- Cacoub P, Vieira M, Saadoun D. Cryoglobulinemia - One Name for Two Diseases. N Engl J Med, 2024; 391(15): 1426-1439.
- Ferri C. Mixed cryoglobulinemia. Orphanet J Rare Dis, 2008; 3: 25.
- Cacoub P, Commarmond C, Sadoun D, Desbois AC. Hepatitis C Virus Infection and Rheumatic Diseases: The Impact of Direct-Acting Antiviral Agents. Rheum Dis Clin North Am, 2017; 43(1): 123-132.
- Cacoub P, Comarmond C, Desbois AC, Saadoun D. Rheumatologic Manifestations of Hepatitis C Virus Infection. Clin Liver Dis, 2017; 21(3): 455-464.
- Falasca K, Mancino P, Ucciferri C, Dalessandro M, Zingariello P, Lattanzio FM, et al. Inflammatory cytokines and S-100b protein in patients with hepatitis C infection and cryoglobulinemias. Clin Invest Med, 2007; 30(5): E167-176.
- Cacoub P, Si Ahmed SN, Ferfar Y, Pol S, Thabut D, Hezode C, et al. Long-term Efficacy of Interferon-Free Antiviral Treatment Regimens in Patients With Hepatitis C Virus-Associated Cryoglobulinemia Vasculitis. Clin Gastroenterol Hepatol, 2019; 17(3): 518-526.
- Ferri C, Antonelli A, Mascia MT, Sebastiani M, Fallahi P, Ferrari D, et al. HCV-related autoimmune and neoplastic disorders: the HCV syndrome. Dig Liver Dis, 2007; 39 Suppl 1: S13-21.
- Falasca K, Ucciferri C, Mancino P, Gorgoretti V, Pizzigallo E, Vecchiet J. Use of epoetin beta during combination therapy of infection with hepatitis c virus with ribavirin improves a sustained viral response. J Med Virol, 2010; 82(1): 49-56.
- Ucciferri C, Occhionero A, Vecchiet J, Falasca K. Cardiac Toxicity Associated with HCV Direct Antiviral Agents. Mediterr J Hematol Infect Dis, 2018; 10(1): e2018069.
- Ucciferri C, Vecchiet J, Pizzigallo E, Falasca K. Optimization of comorbidity therapy to achieve sustained viral response in HCV patients. Hepatogastroenterology, 2012; 59(117): 1589-1590.
- Sise ME, Bloom AK, Wisocky J, Lin MV, Gustafson JL, Lundquist AL, et al. Treatment of hepatitis C virus-associated mixed cryoglobulinemia with direct-acting antiviral agents. Hepatology, 2016; 63(2): 408-417.
- Nava FA, Mangia A, Riglietta M, Somaini L, Foschi FG, Claar E, et al. Analysis of Patients' Characteristics and Treatment Profile of People Who Use Drugs (PWUDs) with and without a Co-Diagnosis of Viral Hepatitis C: A Real-World Retrospective Italian Analysis. Ther Clin Risk Manag, 2023; 19: 645-656.
- Kondili LA, Monti M, Quaranta MG, Gragnani L, Panetta V, Brancaccio G, et al. A prospective study of direct-acting antiviral effectiveness and relapse risk in HCV cryoglobulinemic vasculitis by the Italian PITER cohort. Hepatology, 2022; 76(1): 220-232.
- Cacoub P, Vautier M, Desbois AC, Lafuma A, Saadoun D. Effectiveness and cost of hepatitis C virus cryoglobulinaemia vasculitis treatment: From interferon-based to direct-acting antivirals era. Liver Int, 2017; 37(12): 1805-1813.
- Comarmond C, Garrido M, Pol S, Desbois AC, Costopoulos M, Le Garff-Tavernier M, et al. Direct-Acting Antiviral Therapy Restores Immune Tolerance to Patients With Hepatitis C Virus-Induced Cryoglobulinemia Vasculitis. Gastroenterology, 2017; 152(8): 2052-62 e2.
- Dammacco F, Sansonno D. Therapy for hepatitis C virus-related cryoglobulinemic vasculitis. N Engl J Med, 2013; 369(11): 1035-1045.
- Pluta A, Gutkowski K. Hepatitis C virus associated with mixed cryoglobulinemia, pathogenesis, clinical manifestations, diagnosis and treatment options. Pol Arch Med Wewn, 2003; 109(3): 305-311.
- De Vita S. Treatment of mixed cryoglobulinemia: a rheumatology perspective. Clin Exp Rheumatol, 2011; 29(1 Suppl 64): S99-103.
- Bunchorntavakul C, Reddy KR. Hepatic Manifestations of Lymphoproliferative Disorders. Clin Liver Dis, 2019; 23(2): 293-308.
- Lauletta G, Russi S, Pavone F, Vacca A, Dammacco F. Direct-acting antiviral agents in the therapy of hepatitis C virus-related mixed cryoglobulinaemia: a single-centre experience. Arthritis Res Ther, 2017; 19(1): 74.
- Tonutti A, Polverini D, De Nicola S, Ceribelli A, Soleri M, De Santis M, et al. The evolving scenario of HCV-related mixed cryoglobulinemia and B-cell lymphoma in the era of direct-acting antivirals. Expert Rev Anti Infect Ther, 2025; 23(1): 19-30.
- Terrier B, Cacoub P. Cryoglobulinemia vasculitis: an update. Curr Opin Rheumatol, 2013; 25(1): 10-18.
- Mazzaro C, Panarello G, Carniello S, Faelli A, Mazzi G, Crovatto M, et al. Interferon versus steroids in patients with hepatitis C virus-associated cryoglobulinaemic glomerulonephritis. Dig Liver Dis, 2000; 32(8): 708-715.

