Intraluminal Migration of an Abdominal Drain into the Gastric Cavity After Gastric Bypass: A Case Report
Benjamín Jesús Moya-Leal*, Luis Adrian Alvarez-Lozada, Luis Guillermo Menchaca-Ramos, Claudia Mayela Cardosa-Gonzalez, Mario Leal-Maldonado, Martha Patricia Valdez-Caballero, María Fernanda Balderas-Sandoval, Martín Sergio Rodríguez-Garza1, Estefanía Gómez-Charnichart and Angélica Sofía Lopez-Rojas
ISSSTE Hospital Regional de Monterrey, Monterrey, Nuevo Leon, Mexico
Received Date: 23/11/2025; Published Date: 29/12/2025
*Corresponding author: Benjamín Jesús Moya-Leal, MD, ISSSTE Hospital Regional de Monterrey, Monterrey, Nuevo Leon, Adolfo López Mateos 122, Col. Burócratas Federales, 64380 Monterrey, N.L, Mexico
Abstract
Abdominal drains have long been used as a tool for the early detection of complications such as anastomotic leaks or hemorrhage. They also facilitate the evacuation of bile, gastric secretions, or inflammatory fluid, thereby preventing intra-abdominal collections and abscess formation. However, migration of a drain into the gastric lumen is an exceedingly rare event. Anastomotic leak is one of the most feared complications following Roux-en-Y gastric bypass, with an incidence ranging from 1% to 5%.
We present the case of a 27-year-old male with a body mass index of 56 kg/m² and a history of Nissen fundoplication in childhood, who underwent laparoscopic Roux-en-Y gastric bypass. On postoperative day 10, he developed abdominal pain, tachycardia, and sero-bilious drainage, consistent with an anastomotic leak that required exploratory laparotomy and repair of the dehiscence. Despite intervention, he persisted with high-output drainage and was referred to our unit, arriving with severe malnutrition, anemia, and leukocytosis. Initial imaging did not demonstrate active leakage. Subsequent endoscopy revealed intragastric migration of a Blake drain (~10 cm) and a 6-mm fistulous orifice. The drain was repositioned under direct visualization, a nasogastric tube was placed, and conservative management for postoperative gastrointestinal fistula was initiated. The patient’s condition progressively improved, with closure of the leak confirmed, and he was discharged in good condition with adequate oral tolerance.
Although drain placement in gastric bypass surgery has specific indications, intragastric migration is rare. Awareness of diagnostic and therapeutic tools such as endoscopy enables timely management. Close postoperative surveillance is essential, particularly in patients with atypical evolution, as minimally invasive strategies may achieve successful resolution.
Keywords: Roux-en-Y Gastric Bypass; Anastomotic Leak; Blake Drain; Drain Migration; Bariatric Surgery; Endoscopy; Postoperative Complications
Introduction
Within bariatric surgery, Roux-en-Y gastric bypass is one of the most commonly performed procedures for the management of morbid obesity, due to its high efficacy in weight reduction and improvement of metabolic comorbidities. However, this technique is not without complications, the most significant of which include bleeding, stenosis, and anastomotic leak. The latter has a reported incidence ranging from 1% to 5% and carries a potential risk of mortality, often necessitating urgent surgical reintervention [1,2].
In the postoperative setting, the use of abdominal drains, such as the Blake drain, is a common practice for monitoring and early detection of complications. However, prolonged retention or unsupervised positioning may be associated with adverse events, including infection, local pain, and, in exceptional cases, intraluminal migration. Erosion and penetration of the drain into the gastric lumen through the suture line constitute an extremely rare phenomenon, with only a few cases documented in the literature [3].
This report describes an unusual case of intragastric migration of a Blake drain, diagnosed by endoscopy one month after gastrojejunal anastomosis reconstruction, performed due to a leak secondary to gastric bypass. The current case report follows the CARE checklist for documenting clinical cases.
Case Report
We report the case of a 27-year-old male with morbid obesity (BMI 56 kg/m²), Asperger syndrome, and a sedentary lifestyle, with a history of Nissen fundoplication, who underwent conventional laparoscopic Roux-en-Y gastric bypass. On postoperative day 10, he developed abdominal pain, tachycardia, and serobilious output through the drain, suggesting an anastomotic leak. He was taken for an exploratory laparotomy, during which a gastrojejunal anastomotic dehiscence was identified and repaired. Due to a protracted postoperative course, he was referred to our unit with persistent signs of leakage.
Upon arrival, laboratory studies revealed severe malnutrition according to CONUT, anemia, and leukocytosis. Parenteral nutrition was initiated with Kaviben solution (1700 kcal in 2700 mL), starting at 100 mL over 24 hours, and the patient remained NPO. The patient received a total of four units of packed red blood cells for anemia. A subsequent esophagogastroduodenal series did not reveal any leak (Figure 1). Despite these measures, the patient continued to have serobilious drain output, nausea, and persistent vomiting. Follow-up CT imaging confirmed prior findings, without evidence of active leakage.
An endoscopic evaluation was performed, revealing a patent gastrojejunal anastomosis without leak (Figure 2). A Blake drain was found to have migrated approximately 10 cm into the gastric lumen (Figure 3). Under direct endoscopic visualization, the drain was withdrawn 12 cm until it was completely outside the stomach, revealing a 6-mm fistulous orifice. The drain was then repositioned and connected to a collection system, and a nasogastric tube was placed as part of conservative management while awaiting fistula closure.
A follow-up esophagogastroduodenal series performed five days later demonstrated appropriate contrast passage without evidence of leakage or delayed gastric emptying (Figure 4). Enteral diet was reintroduced seven days after drain manipulation and repositioning. Management of the low-output fistula included continued parenteral nutrition and administration of octreotide. The Blake drain was removed after output decreased significantly. After discharge, the patient remained immobile at home, consistent with his prior lifestyle, which, according to the family’s report by phone, may have contributed to what appeared to be a pulmonary embolism; however, no clinical evidence is available to support this. The patient subsequently died at home one week later.

Figure 1: Initial gastroesophageal series: The gastric pouch showed postsurgical changes from bariatric surgery, without evidence of extrinsic compression. There was adequate passage of contrast into the intestinal tract, with delayed emptying, no leakage, and normal transit of the contrast material.
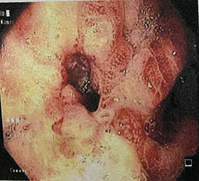
Figure 2: Endoscopic image showing a patent anastomosis with no evidence of leakage.
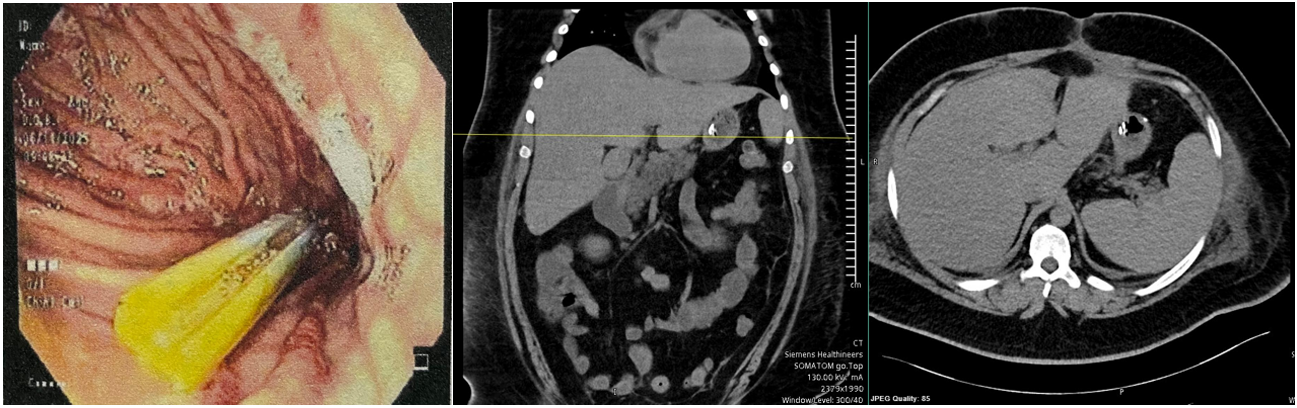
Figure 3: A. Abdominal CT showing a migrated drain within the gastric cavity, without evidence of collections or leaks. B. Endoscopic image showing a drain migrated into the gastric lumen.
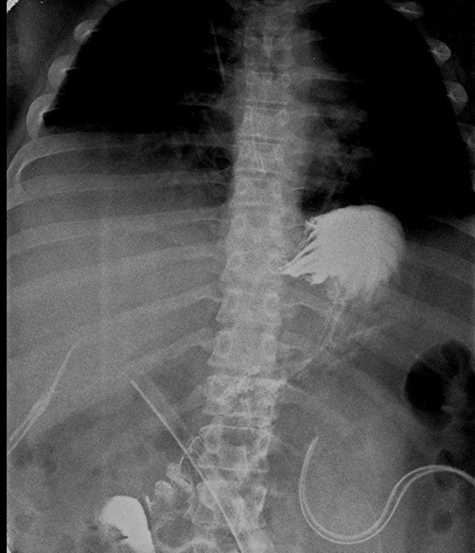
Figure 4: Esophagogastroduodenal series after tube repositioning: there was adequate passage of contrast material.
Discussion
Migration of devices, such as drains or catheters, from their intended position following gastrointestinal surgery is relatively common, occurring in over 28% of placed drains [4]. However, migration into the gastrointestinal lumen is an uncommon phenomenon. In the context of bariatric surgery, there are few reports describing intraluminal drain migration [3,5]. Other case reports have described this same phenomenon, although the populations often involve patients undergoing gastrectomy for oncologic indications, which may introduce additional comorbidities that increase the risk of complications [6-9]. Anastomotic leaks, on the other hand, remain one of the most feared complications in this type of surgery, and timely diagnosis and early intervention are crucial determinants of patient outcomes.
Some of the mechanisms proposed as triggers for these events include constant pressure of the drain against the suture line, combined with inflammation and tissue fragility in the context of a prior leak or impaired healing [7]. Prolonged contact with friable and vulnerable tissues may lead to progressive erosion and penetration into the gastric lumen. In this case, the Blake drain, repositioned after reintervention for the leak, likely remained in close contact with the anastomotic site, facilitating migration into the gastric cavity. Detection was achieved due to clinical suspicion and the performance of endoscopy one month after surgery.
Interestingly, several previously published case reports/series of intraluminal drain migration involved patients who also had anastomotic leakage [6-8]. In the report by Lai et al. (2010), the migrated drain was visualized through a mucosal defect over the anastomotic site. In Wilmot et al.’s case series (2007), two of four patients had anastomotic leakage; additionally, among 57 patients with leakage in their broader cohort, 4 (7%) had intraluminal drain migration [7]. In the study by Janež et al. (2025), no anastomotic dehiscence was observed, though a postoperative collection near the anastomosis was present, and the drain entry point was ultimately located 2 cm distal to the anastomosis [6]. Across these reports, authors noted pro-inflammatory states that could impair healing. In our patient, anastomotic leakage was also present; however, obesity–malnutrition with severe CONUT was also considered a key factor contributing to the intraluminal migration.
The literature supports the use of endoscopy as both a diagnostic and therapeutic tool in such cases, allowing safe drain removal and assessment of anastomotic integrity [6,10-12]. Spontaneous closure of small fistulous tracts is possible with conservative management, including nasogastric tube placement, parenteral nutrition, and fasting [13].
This case emphasizes the importance of close postoperative monitoring in patients with atypical evolution after reintervention for leaks and highlights the need for caution regarding prolonged use and placement of drains in bariatric surgery.
Conclusion
While abdominal drains are valuable for postoperative monitoring, proper placement and fixation under direct visualization are essential to prevent rare but serious complications, such as intragastric migration. This case underscores the importance of endoscopy not only for accurate diagnosis but also for minimally invasive management. Close postoperative surveillance is critical, particularly in patients with atypical clinical evolution, to ensure timely detection and resolution of complications.
Authorship Criteria:
- Concept and design of study or acquisition of data or analysis and interpretation of data: BJ M-L; LA A-L; MP V-C; MF B-S; MS R-G; AS L-R.
- Drafting the article or revising it critically for important intellectual content: BJ M-L; LA A-L; LG M-R; CM C-G; M L-M; E G-C.
- Final approval of the version to be published: LG M-R; CM C-G.
- Clinical data collection and case documentation: BJ M-L; LA A-L.
- Review and organization of imaging studies and diagnostic findings: MP V-C; MF B-S.
- Comprehensive literature review and evidence synthesis: LG M-R; MS R-G; AS L-R.
- Drafting of the initial manuscript, including case description and discussion: BJ M-L; LA A-L; M L-M.
- Critical analysis of the case in the context of current medical evidence: BJ M-L; LG M-R; CM C-G.
- Manuscript editing, refinement, and academic formatting according to journal guidelines: BJ M-L; LA A-L; LG M-R; CM C-G; E G-C.
- Preparation of figures, tables, and supportive materials for publication: M L-M; E G-C.
Guarantor: BJ M-L and LA A-L.
Conflicts of Interest: The authors declare no conflicts of interest.
Funding: This research received no external funding.
Acknowledgments: The authors declare that there are no acknowledgments for this manuscript.
References
- American Society for Metabolic and Bariatric Surgery (ASMBS). Clinical Issues Committee Guidelines, 2022.
- Chang SH, Freeman NLB, Lee JA, Stoll CRT, Calhoun AJ, Eagon JC, et al. Early major complications after bariatric surgery in the USA, 2003-2014: a systematic review and meta-analysis. Obes Rev, 2018; 19(4): 529-537. doi: 10.1111/obr.12647.
- Tola GG, Tesso BA, Moges TG, Abebe DM. An extraluminal migration of a gastrointestinal foreign body to the anterior abdominal wall: A rare case report. Int J Surg Case Rep, 2024; 120: 109794. doi: 10.1016/j.ijscr.2024.109794.
- Gilbert A, Ortega-Deballon P, Di Giacomo G, Cheynel N, Rat P, Facy O. Intraperitoneal drains move. J Visc Surg, 2018; 155(2): 105-110. doi: 10.1016/j.jviscsurg.2017.10.001.
- Narendra N, Sandar N, Jhagroo G, Anand S. Intraluminal migration of a Jackson-Pratt drain: an unusual complication of gastric bypass surgery. Am J Gastroenterol, 2009; 104(Suppl): S381.
- Janež J, Romih J, Čebron Ž, Gavric A, Plut S, Grosek J. Intraluminal migration of a surgical drain near an anastomosis site after total gastrectomy: A case report. World J Clin Cases, 2025; 13(12): 99229. doi: 10.12998/wjcc.v13.i12.99229.
- Wilmot AS, Levine MS, Rubesin SE, Kucharczuk JC, Laufer I. Intraluminal migration of surgical drains after transhiatal esophagogastrectomy: radiographic findings and clinical relevance. AJR Am J Roentgenol, 2007; 189(4): 780-785. doi: 10.2214/AJR.07.2322.
- Lai PS, Lo C, Lin LW, Lee PC. Drain tube migration into the anastomotic site of an esophagojejunostomy for gastric small cell carcinoma: short report. BMC Gastroenterol, 2010; 10: 50. doi: 10.1186/1471-230X-10-50.
- Alhinho HCAW, Ferreira FC, Medeiros RCL, Ferraz ÁAB, Campos JM. Severe Dehiscence of Gastrojejunal Anastomosis after Gastric Bypass: Its Cure by Using Partially Covered Stent and Avoiding Migration. Obes Surg, 2018; 28(2): 594. doi: 10.1007/s11695-017-3016-8.
- Liscia G, Scaringi S, Facchiano E, Quartararo G, Lucchese M. The role of drainage after Roux-en-Y gastric bypass for morbid obesity: a systematic review. Surg Obes Relat Dis, 2014; 10(1): 171-176. doi: 10.1016/j.soard.2013.09.008.
- Peña ME, Schlottmann F, Laxague F, Sadava EE, Buxhoeveden R. Usefulness of Abdominal Drain in Laparoscopic Roux-en-Y Gastric Bypass: A Randomized Controlled Trial. J Laparoendosc Adv Surg Tech A, 2020; 30(5): 538-541. doi: 10.1089/lap.2019.0783.
- Narendra N, Sandar N, Jhagroo G, Anand S. Intraluminal migration of a Jackson-Pratt drain: an unusual complication of gastric bypass surgery. Am J Gastroenterol, 2009; 104(Suppl): S381.
- Doumouras AG, Maeda A, Jackson TD. The role of routine abdominal drainage after bariatric surgery: a metabolic and bariatric surgery accreditation and quality improvement program study. Surg Obes Relat Dis, 2017; 13(12): 1997-2003. doi: 10.1016/j.soard.2017.08.019.

