Guillain-Barré Syndrome: Diagnostic Challenges and Management Pearls in The Presence of Concomitant Cervical Spondylosis Compression Myelopathy
Omar Alqaisi1,*, Maha Subih1, Lorent Sijarina2, Patricia Tai3 and Aliaa Ezz Eldin Abd Elmoaty4
1Faculty of Nursing, Al-Zaytoonah University, Amman, Jordan
2Faculty of Medicine, University of Prishtina, 45 Rruga Ibrahim Lutfiu, Kosovo
3Department of Oncology, University of Saskatchewan, 105 Administration Place, Saskatoon, SK, S7N 5A2, Canada
4Faculty of Nursing, Helwan University, lecturer in the Nursing Administration department, Cairo, Egypt
Received Date: 30/10/2025; Published Date: 08/12/2025
*Corresponding author: Omar Alqaisi, Faculty of Nursing, Al-Zaytoonah University, Amman, Jordan
ORCID: 0009-0006-9760-651X
Abstract
Background: Guillain-Barré Syndrome (GBS) is an uncommon differential diagnosis of paraplegia. There are controversies in its management.
Clinical Case: A previously healthy man in his 50s presented with progressive ascending weakness, facial paralysis, and autonomic dysfunctions over one week. Initial assessment revealed concurrent cervical spondylosis with compression myelopathy at C4-C5, creating diagnostic complexity. Cerebrospinal fluid analysis demonstrated albumin-cytologic dissociation (protein 167 mg/dL), and nerve conduction studies confirmed bilateral demyelinating polyneuropathy consistent with acute inflammatory demyelinating polyradiculoneuropathy (AIDP). Treatment with immunoglobulin (2g/kg over 5 days) followed by plasma exchange (4 sessions) resulted in significant neurological improvement. Muscle strength increased from grade 2/5 to 4/5 in the upper limbs and 1/5 to 3/5 in the lower limbs. Comprehensive multidisciplinary rehabilitation enabled independent ambulation and the resumption of functional activities.
Conclusion: This case highlights the diagnostic challenges that arise when Guillain-Barré Syndrome (GBS) presents with concomitant cervical spondylosis compression myelopathy. Effective management of GBS requires systematic clinical assessment, evidence-based immunotherapy and coordinated multidisciplinary rehabilitation.
Introduction
Guillain-Barré syndrome (GBS) is an acute autoimmune demyelinating polyneuropathy affecting approximately 1-2 per 100.000 individuals annually worldwide [1,2]. The condition poses significant diagnostic challenges when it occurs concurrently with spinal pathology, as overlapping clinical presentations can obscure the diagnosis and delay critical interventions [3].
This case report demonstrates the diagnostic complexity encountered when GBS presents alongside cervical spine abnormalities, emphasizing the critical importance of systematic clinical evaluation and evidence-based treatment approaches [4,5]. We highlight contemporary management strategies incorporating the latest evidence from 2024-2025 research, particularly focusing on multidisciplinary care, care coordination, and treatment controversies in acute inflammatory demyelinating polyradiculoneuropathy (AIDP) [6,7]. The educational value of this case lies in illustrating how careful clinical judgment, supported by appropriate diagnostic investigations, can differentiate between concurrent pathological processes and guide optimal therapeutic decision-making in complex neurological presentations.
Written informed consent was obtained from the patient for publication of this case report and all accompanying clinical information and images. The patient was provided with a copy of the final manuscript before submission and granted explicit permission for publication. This study was conducted in accordance with the principles of the Declaration of Helsinki. Patient’ confidentiality has been maintained throughout with appropriate anonymization of identifying information. Institutional ethical guidelines for case report publication were followed.
This study adhered to the principles of the Declaration of Helsinki. Patients’ confidentiality and anonymity were rigorously maintained.
Observation
A previously healthy teacher in his 50s presented to the emergency department with a chief complaint of progressive bilateral limb weakness developing over one week [2]. He was previously healthy with well-controlled hypercholesterolemia and chronic hypertension plus occasional episodic asthma. There was no history of recent infections or travel vaccinations. His social history was notable for an active lifestyle, including daily one-hour walks and residence in a three-level house without elevator access.
Differential diagnoses included cervical myelopathy, botulism, Acute Inflammatory Demyelinating Polyradiculoneuropathy (AIDP), and myasthenia gravis. The ascending pattern of weakness generalized areflexia, and Cerebrospinal Fluid (CSF) albuminocytologic dissociation guided the diagnosis towards AIDP, while reflexia contradicted isolated cervical myelopathy.
Clinical findings and timeline
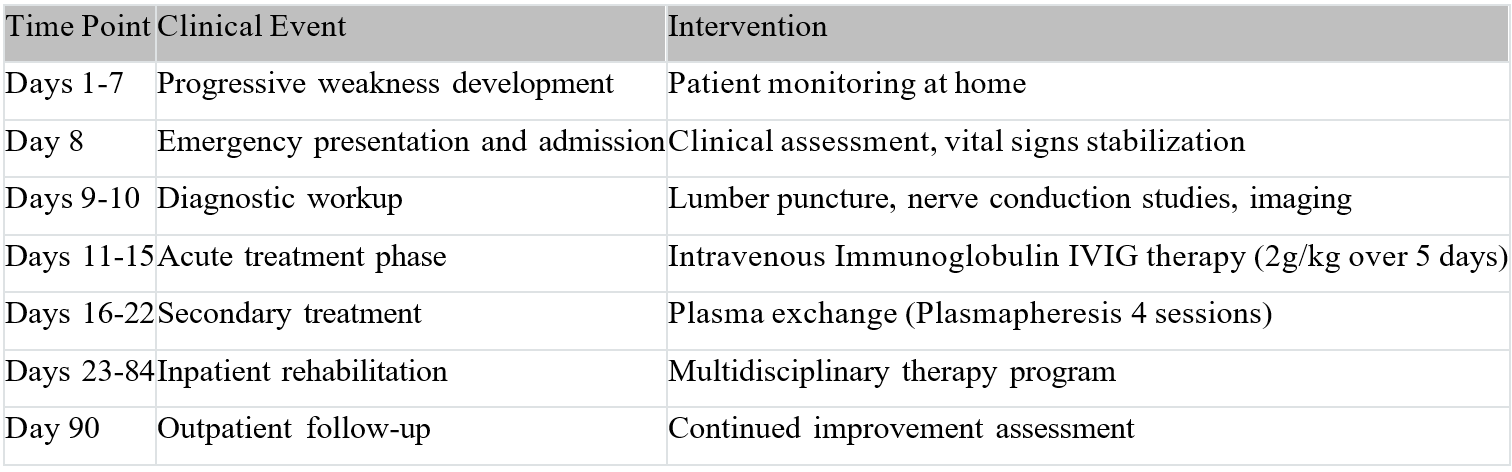
Initial presentation (DAY 1-7)
- Progressive ascending weakness beginning in the lower extremities
- Bilateral upper and lower limb paraesthesia starting distally
- Facial weakness with difficulty speaking and swallowing
- Deep, cramping pain in legs and back, worse at night
- Urinary retention and fluctuating blood pressure
Emergency department assessment (DAY 8)
- Admitted again to our Emergency Department
- Patient alert and oriented, yet requiring wheelchair assistance
- Vital signs: BP 130/70 mmHg, HR 110 bpm, O2 saturation 96%, RR 20/min
- Neurological examination revealed:
- Symmetrical bilateral muscle weakness (lower limbs > upper limbs)
- Diminished deep tendon reflexes throughout
- Bilateral facial muscle weakness
- Distal sensory loss in all four extremities
- Dysarthria and dysphagia
- Normal cognitive function
Timeline of Care
Investigations
Laboratory studies:
Lumber puncture revealed albumin cytologic dissociation: elevated CSF protein (167 mg/dL) with elevated neutrophils (93%; normal <5 cells/µL ) [8,9].
Complete blood count, electrolytes, liver function tests, and creatine phosphokinase: within normal limits
Electrophysiological studies:
Electrophysiological studies revealed bilateral demyelinating polyneuropathy, consistent with AIDP. Lumbar puncture showed albumin cytologic dissociation (protein 167 mg/dl). Cervical MRI showed spondylosis and compression at the C4-5 level, although clinical findings were incongruent with pure myelopathy, favoring multiple diagnoses [9,10].
Imaging studies (Figures 1 and 2 as shown):
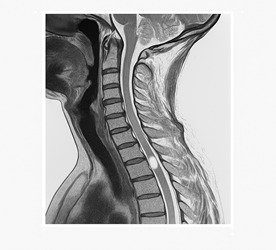
Figure 1: Cervical spine magnetic resonance imaging (MRI) revealed multilevel spondylosis with significant spinal canal stenosis and compression myelopathy at C4-C5 levels, showing hyperintense T2 signal changes indicating myelomalacia.
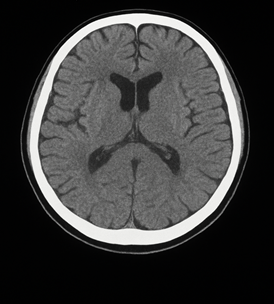
Figure 2: Demonstrated cerebral atrophy which is normal for his age, with no acute pathological changes, thus excluding central nervous system causes.
Differential diagnosis
Given the patient's presentation with progressive weakness, facial paralysis, and autonomic dysfunction, several differential diagnoses were systematically considered:
1. Cervical Myelopathy clinical considerations: initially suspected due to documented cervical spondylosis with compression at C4-C5 levels on MRI imaging [2,6].
Exclusion criteria: the ascending patterns of weakness (starting from lower limbs and progressing upward) were inconsistent with cervical myelopathy, which typically presents with descending weakness. Additionally, the presence of areflexia throughout all limbs contraindicated the expected hyperreflexia below the level of spinal cord compression.
2. Acute Inflammatory Demyelinating Polyradiculoneuropathy (AIDP) – confirmed diagnosis
Supporting evidence: classic ascending weakness patterns, generalized areflexia, facial nerve involvement with bulbar symptoms, and characteristic albumin cytologic dissociation in cerebrospinal fluid (protein 167 mg/dL with elevated neutrophils 93%) [8,9].
Confirmatory findings: nerve conduction studies revealed bilateral demyelinating polyneuropathy affecting both upper and lower limbs, consistent with the AIDP subtype of Guillain-Barré syndrome.
3. Acute Motor Axonal Neuropathy (AMAN)
Clinical considerations: another variant of Guillain-Barré syndrome that could present with similar motor weakness.
Exclusion criteria: electrophysiological studies showed demyelinating rather than axonal patterns, and the patients had significant sensory involvement, which is less common in pure AMAN [10,11].
4. Myasthenia Gravis
Clinical consideration: considered due to the presence of facial weakness, dysphasia, and fluctuating symptoms.
Exclusion criteria: the presence of significant sensory symptoms, generalized areflexia, and characteristic CSF findings with albumin cytologic dissociation are not typical of myasthenia gravis [12,13].
5. Brainstem stroke
Clinical consideration: initially considered due to the acute onset of facial weakness and bulbar symptoms that could suggest brainstem pathology.
Exclusion criteria: normal brain CT scan, excluding acute cerebrovascular events, bilateral symmetrical presentation rather than focal neurological deficits peripheral nerve involvement demonstrated on electrophysiology.
6. Botulism
Clinical consideration: could present with similar descending paralysis and autonomic dysfunction.
Exclusion criteria: Botulism was excluded due to the upward progression pattern and significant sensory involvement. Myasthenia gravis was considered unlikely given the sensory symptoms and electrophysiological findings
The final diagnosis of Guillain-Barré syndrome (AIDP subtype) was confirmed through the combination of characteristic clinical presentation, cerebrospinal fluid analysis demonstrating albumin cytologic dissociation, and nerve conduction studies showing bilateral demyelinating polyneuropathy.
Treatment
Immunotherapy
Intravenous immunoglobulin (IVIG): 2g/kg administered over five consecutive days, providing therapeutic antibodies to modulate the autoimmune response [3,11].
Plasma exchange: four sessions conducted over one week (200-250 ML plasma /Kg body weight per session) to remove pathogenic circulating antibodies [4,12].
Supportive care:
• Nutritional support via nasogastric tube initially due to dysphagia risk
• Indwelling urinary catheter for managing urinary retention, preventing pressure ulcers through frequent repositioning, and utilizing pressure-relieving surfaces.
• Pain management with appropriate analgesics
• Anticoagulation for thromboembolism prevention
• Continuous respiratory monitoring
Multidisciplinary rehabilitation:
Physiotherapy: Twice-weekly sessions focusing on functional mobility, balance training, gait re-education, and progressive strengthening
Occupational therapy: Providing adaptive equipment and training for independence in daily activities
Speech-language pathology: swallowing assessment, diet texture modifications, and speech clarity improvement exercises.
Outcome and follow-up
Inpatient rehabilitation progress (weeks 3-12)
Upper body strength improvement: 2/5 to 4/5
Lower limb strength improvement: 1/5 to 3/5
Progressive sensory recovery
Achieved independence in most daily activities
Transitioned from total dependence to minimal assistance with transfers
Achieved short-distance ambulation with walker assistance
Improved swallowing ability, allowing for soft diet intake
Significant speech clarity improvement
Three-month follow-up
Continued strength and endurance improvement
Occasional neuropathic pain in the feet
Mild residual lower limb weakness
Independent ambulation for longer distances without assistive devices
Minimal foot drop requiring ankle-foot orthosis for extended walking
Enrolled in a long-term outpatient therapy program
Overall prognosis assessed as fair to good, with continued gradual recovery expected
Returned to teaching
Discussion
This case illustrates the diagnostic complexity that arises when GBS presents concurrently with structural spinal pathology [2,6]. The presence of cervical spondylosis with compression myelopathy at C4-C5 levels initially complicated the clinical assessment, as both conditions can present with sensorimotor deficits.
The key distinguishing features that supported GBS diagnosis included:
Patterns of weakness: ascending progression characteristics of GBS versus descending patterns typical of cervical myelopathy
Reflex changes: generalized areflexia in GBS versus hyperreflexia below the level of spinal compression
Electrophysiological findings: demyelinating polyneuropathy patterns consistent with AIDP subtype
CSF analysis: classic albumin cytologic dissociation supporting peripheral nerve inflammation
Early administration of IVIG therapy is aligned with current evidence-based guidelines and has been shown to hasten recovery and reduce disability [3,11]. Subsequent plasma exchange therapy was initiated due to an incomplete initial response, consistent with clinical protocols for severe or refractory cases [4,12].
The comprehensive rehabilitation approach used in this case highlights the vital role of coordinated multidisciplinary care. Early physical therapy, occupational therapy for functional independence, and speech-language pathology for dysphagia management all significantly contribute to the favourable outcomes achieved [14,15].
Subtype-specific management
This case represents the AIDP subtype, characterized by demyelinating changes on nerve conduction studies. Management approaches and considerations for different GBS subtypes include:
AIDP: typically responds well to standard immunotherapy with a favourable but prolonged recovery
Acute Motor Axonal Neuropathy (AMAN): may show faster initial improvement but potentially more residual deficits
Miller Fisher Syndrome often requires less aggressive treatment due to the tendency for spontaneous recovery
Acute Motor-Sensory Axonal Neuropathy (AMSAN): usually requires aggressive treatment with guarded prognosis due to axonal involvement
Contemporary treatment advances
Emerging therapies showing promise in 2024-2025 research include:
Complement inhibitors (e.g., ANX005): targeting the classical complement pathway for more specific immunomodulation
IgG degradation therapy (ImIifidase): offering rapid antibody reduction within hours, biomarker-guided therapy: developing predictive markers for personalized treatment selection
Multidisciplinary care integrations
The comprehensive rehabilitation approaches employed in this case demonstrate the critical importance of coordinated multidisciplinary care. Early physical therapy intervention, occupational therapy for functional independence, and speech-language pathology for dysphagia management significantly contributed to favourable outcomes achieved.
Long-term considerations include monitoring for post-GBS fatigue syndrome, managing neuropathic pain, and addressing potential psychological impacts of acute illness experiences. The patients’ gradual return to functional independence highlights the importance of sustained rehabilitation efforts and realistic outcome expectations.
Learning points
1. Systematic clinical evaluation is essential when multiple pathological processes may coexist [2,6].
2. Early recognition of GBS patterns (ascending weakness, areflexia) can differentiate from the spinal pathology [8,9].
3. Prompt immunotherapy with IVIG or plasma exchange significantly improves functional outcomes [3,11].
4. Multidisciplinary rehabilitation is crucial for optimizing recovery and quality of life [14,15].
5. Patient education regarding prognosis and long-term recovery expectations is crucial for treatment adherence and psychological well-being [13,15].
This case contributed to our understanding of the complexity of GBS management while demonstrating that excellent functional recovery is achievable through evidence-based treatment and comprehensive rehabilitation, even in diagnostically challenging presentations.
Authors’ Contributions: The last author participated in the evaluation and follow-up of the patient. First and second authors wrote the case report. All the authors of the manuscript have read and agreed to its content.
References
- Leonhard SE, Mandarakas MR, Gondim FA, Bateman K, Ferreira ML, Cornblath DR, et al. Diagnosis and management of Guillain–Barré syndrome in ten steps. Nature Reviews Neurology, 2019; 15(11): 671-683.
- Wijdicks EF, Klein CJ, editors. Guillain-barre syndrome. Mayo Clinic Proceedings; 2017: Elsevier.
- Hughes RA, Swan AV, Van Doorn PA. Intravenous immunoglobulin for Guillain‐Barré syndrome. Cochrane Database of Systematic Reviews, 2014; 9.
- Group SG-BST. Randomised trial of plasma exchange, intravenous immunoglobulin, and combined treatments in Guillain-Barré syndrome. The Lancet, 1997; 349(9047): 225-230.
- Van den Berg B, Walgaard C, Drenthen J, Fokke C, Jacobs BC, Van Doorn PA. Guillain–Barré syndrome: pathogenesis, diagnosis, treatment and prognosis. Nature Reviews Neurology, 2014; 10(8): 469-482.
- Yuki N, Hartung H-P. Guillain–barré syndrome. New England Journal of Medicine, 2012; 366(24): 2294-2304. DOI: 10.1056/NEJMra1114525.
- Willison HJ, Jacobs BC, van Doorn PA. Guillain-barre syndrome. The Lancet, 2016; 388(10045): 717-727.
- Verboon C, Doets AY, Galassi G, Davidson A, Waheed W, Péréon Y, et al. Current treatment practice of Guillain-Barré syndrome. Neurology, 2019; 93(1): e59-e76.
- Fokke C, van den Berg B, Drenthen J, Walgaard C, van Doorn PA, Jacobs BC. Diagnosis of Guillain-Barré syndrome and validation of Brighton criteria. Brain, 2014; 137(1): 33-43.
- Moss KR, Bopp TS, Johnson AE, Höke A. New evidence for secondary axonal degeneration in demyelinating neuropathies. Neuroscience Letters, 2021; 744: 135595.
- Ruts L, Drenthen J, Jongen J, Hop W, Visser GH, Jacobs B, et al. Pain in Guillain-Barre syndrome: a long-term follow-up study. Neurology, 2010; 75(16): 1439-1447.
- Ullah W, Ali H, Muhammad Y, Ullah N, Khalil I, Ahmad S, et al. Evaluating the Efficacy and Cost-Effectiveness of Plasmapheresis and Intravenous Immunoglobulin in Acute Guillain-Barre Syndrome Management in Emergency Departments. Cureus, 2024.
- Burns TM, editor Guillain-barré syndrome. Seminars in neurology; Thieme Medical Publishers, 2008.
- Meena A, Khadilkar S, Murthy J. Treatment guidelines for Guillain–Barré syndrome. Annals of Indian Academy of Neurology, 2011; 14(Suppl1): S73-S81.
- Dimachkie MM, Barohn RJ. Guillain-Barré syndrome and variants. Neurologic clinics, 2013; 31(2): 491.

