Technical Validation of Dried Antibody Reagent Tube RE-ALB for B-ALL Minimal Residual Disease Analysis
Dia Mansukhani*, Malini Chawla, Nahid Anis Shaikh and Rania Medhat Seliem
Hematology Laboratory, Department of Laboratory Medicine and Pathology, Rashid hospital, Dubai Health, Dubai
Received Date: 20/09/2025; Published Date: 06/10/2025
*Corresponding author: Dia Mansukhani, Hematology Laboratory, Department of Laboratory Medicine and Pathology, Rashid hospital, Dubai Health, Dubai
Abstract
Background: Flow-cytometric minimal/measurable residual disease (FC-MRD) monitoring is a well-established risk-stratification factor in B-lymphoblastic leukemia (B-ALL) management. It is considered as a basis for deintensification or escalation in treatment protocols. FC- MRD is a high sensitivity assay, which routinely detects 1 diseased cell in 10,000 cells (10-4). Standardizing multi-color flow-cytometry panels for minimal/measurable residual disease (MRD) testing has been difficult due to the complexity of identifying leukemic cells from normal blood cells and ensuring consistent, high-sensitivity results across different labs and protocols. Ready-to-use tubes containing pre-aliquoted, dried antibody reagents for MRD flow cytometry are now available Hence, we decided to evaluate Beckman Coulter Duraclone Rare Events ALB (RE-ALB) for MRD evaluation, and evaluate its applicability in our laboratory. RE-ALB is a single 7-color tube with provision of 3 drop in markers in PE, APC and PB fluorochromes.
Methods: RE-ALB tube was studied for evaluation of minimal residual disease (MRD) in 25 bone marrows (BM) of postinduction phase of B-ALL and compared to the in use 5-color, multiple tubes ,MRD assay. Additionally, spiking studies was performed for lower limit of quantification (LLOQ) and lower limit of detection (LOD) in RE-ALB tubes. RE-ALB was performed in duplicates with drop in marker as CD73C PE, CD123 APC in one tube and CD66c PE in another tube additionally verifying reproducibility of population of interest in all cases. More than 1 million events could be acquired for most of our MRD cases (median events were 1.5 million events with a range from 580085 – 2270364 events). Additionally, College of American Pathologists (CAP) proficiency testing (PT) samples (n-4) were also processed and compared.
Results: MRD was measurable in 4 out of 25 cases in both RE-ALB (0.26%, 0.06% 0.03 % and 0.01% respectively) and 5-color antibody panel (0.26, 0.047, 0.048% and 0.01% respectively). Average CV obtained comparing the percentage of cell of interest obtained on RE-ALB and 5-color panel was 17.02%. Rest of the 21 cases were negative by both RE-ALB and 5-color panel. 2 of 4 samples PT from CAP showed MRD positivity by both RE-ALB tube and 5-color panel.100% diagnostic concordance was found between 5-color and RE-ALB tube.
Conclusion: Ease of validation, acquisition of high number of events, provision of addition of new Leukemia associated immunophenotype (LAIP) markers, differentiating MRD from background hematogones was possible utilizing Duraclone RE-ALB tubes. Additionally, the results obtained using these tubes are easily reproducible. Using these tubes help in better inventory management and increase overall efficiency in our lab.
Keywords: Technical Validation, B-Acute Lymphoblastic Leukemia, Minimal/measurable Residual Disease, Flow Cytometry
Introduction
Flow-cytometric minimal residual disease (FC-MRD) monitoring is a powerful tool for risk stratification in B-lymphoblastic leukemia/lymphoma (B-ALL) and is a basis for modifying treatment protocols [1] Discrimination of abnormal from normal B cells during early stages of development in bone marrow can be achieved through precise assessment of B cell surface antigen expression patterns. Differential densities of CD10, CD19, CD20, CD34, CD38, CD45 and CD58 expression on the surface of B cells, can define normal and abnormal patterns [1].
FC-MRD (FC-MRD) technique is widely applicable, relatively easy to perform and has a rapid turnaround time [1].
FC-MRD analysis allows simultaneous assessment of the normal hematopoietic elements and presence of CD34+ normal myeloid blasts, mast cells, plasma cells represent sample quality for adequacy of bone marrow to avoid false-negative results [1]. While 5-6 color antibody panel are technically easy to standardize and perform in the lab but it has disadvantages as in one has to perform multiple tubes utilizing backbone markers like CD10, CD19, CD34 which may have greater financial implications and increased staff hours to perform and analyze each MRD case.
With availability of highly sophisticated multicolor flow cytometers which can detect up to 8-10 fluorochromes [2] one can routinely achieve the sensitivity of 0.01% (1-in-104) with the acquisition of minimum 500,000 events with a single tube [1]. This allows for the analysis of smaller sample volumes while optimizing time and cost.
Although multicolor single tube MRD assay has many advantages, but increase in the single tube of multiple color complexity is not without challenges and validating this is very laborious. Keeping this in mind, we decided to evaluate performance of Beckman Coulter (BC) dried reagent tube for the detection of B- ALL MRD “Duraclone Rare Events ALB” (RE-ALB). The DURA Clone RE-ALB panel enables sensitive detection of MRD using carefully optimized and titrated antibody combinations. The RE-ALB tube included seven BC, fluorochrome-conjugated dried antibodies along with 3 empty channels (PE, APC, PB) to add drop-in markers basis the Leukemia associated immunophenotype (LAIP) of individual MRD case or as per the Lab policy [2,3].
The aim of this study was to evaluate performance of Duraclone RE-ALB tube as compared to us in use 5-color MRD panel using liquid reagents. Secondary aim was to study applicability of RE-ALB for B –ALL MRD detection in our lab.
Material and Methods
RE-ALB tube was performed on 5 normal BM (lymphoma staging) samples of patients to evaluate marker expressions on hematogones as expected. RE-ALB tube was evaluated in 25 postinduction bone marrow samples (day 29) received in the laboratory for B-ALL MRD testing [7]. Patient age ranged from 3 yrs. - 48 yrs. Additionally RE-ALB tube was also evaluated in 10 diagnostic B-ALL samples (Table 2). Proficiency testing (PT) samples from college of American pathologists (CAP) received for B-ALL MRD analysis were also processed for RE-ALB tubes (n=4). All samples were compared to our routine 5-color panel utilizing liquid reagents.
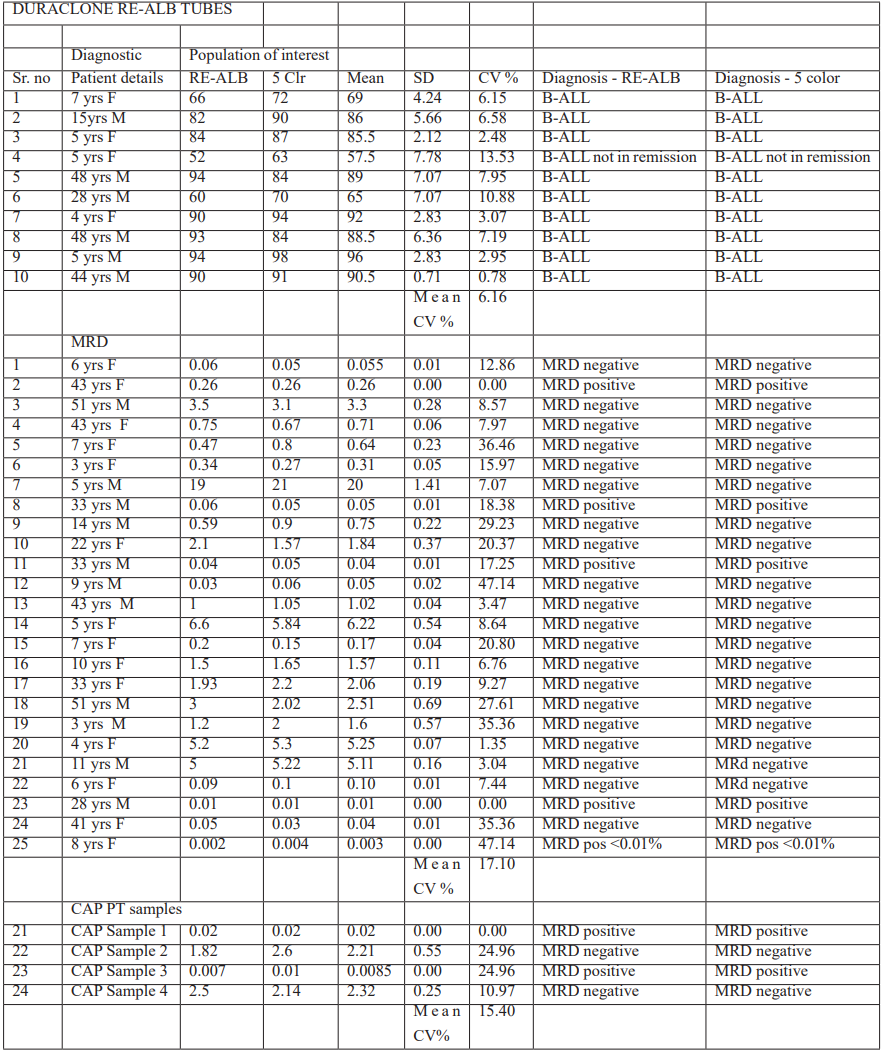
The RE-ALB tube included seven BC, fluorochrome-conjugated dried antibodies: CD45Krome Orange (J.33), CD58FITC (AICD58), CD34 ECD (581), CD10PC5.5 (ALB1), CD19PC7 (J3-119), CD38 AA700 (LS198-4-3), CD20 AA750 (B9.E9 [HRC20]) [3].
All Duraclone RE-ALB tubes were performed in duplicate, with CD73 PE (Clone AD-2), CD123 APC (SSDDCLY107D2, Beckman coulter) in one tube and CD66C PE (clone KOR-SA3544, Beckman coulter) used as drop in marker in the other.
5-color panel used was as in Table 1. All the antibodies were procured from Beckman Coulter.
Sample preparation: RE-ALB tube was processed as per manufacturer’s instruction. Approximate cell concentration was obtained on CBC analyzer by running the flow cytometry sample (EDTA) in 1:10 dilution. Appropriate volume of sample was added to have final volume of 1.5 to 2.0 million leukocytes in each RE-ALB tube. 5-color antibody panel with 7 tubes was processed as stain-lyse-wash protocol using 100 ul as sample. All antibodies were titrated at the time of initial validation of 5-color panel and were within their mentioned expiry.
Flow Cytometer and Quality Control: The instrument used was 10-color, three-laser Navios (Beckman Coulter®, Miami, FL). Daily controls including Flow check and Flow Set Pro Fluorophores™ (Beckman Coulter) were run to monitor the instrument performance. All the samples were processed and analyzed within 24 hours of receiving the sample as per recommendation. All processed tubes were acquired immediately.
Gating Strategy: Initial gating was performed on Time vs SSC and followed by gating on singlets. Then, FSC vs SSC gating was done to remove debris, artifacts that may interfere with high event analysis of MRD and to include only viable events from singlets dot-plots. Subsequently, clean CD19-positive B cells were gated as shown in the figure 1. These cells were evaluated using bivariate dot plots with multiple combinations of markers included in the panel [1,3]. MRD percentage was calculated as MRD positive events in viable cells [6].
Analysis of each case was done using Kaluza software (Beckman Coulter) by two pathologists. Patient and sample characteristics are summarized in table 2.
Table 1: 7 tubes with 5 color panel used for B-ALL MRD detection.
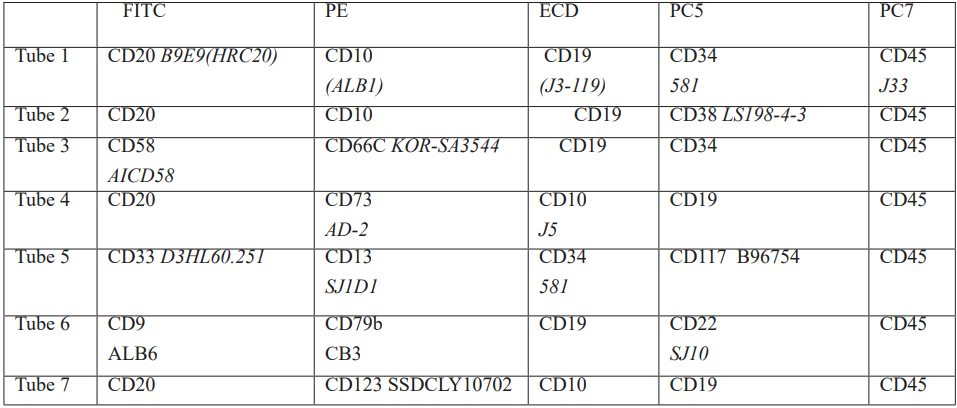
Table 2: Showing comparison of population of interest and final diagnosis for all samples in 5 colors to RE-ALB tube.
Method of Comparison
Normal samples were evaluated for presence of hematogones, plasma cells and evaluate expression of markers on these cells as expected. A concordance in diagnosis of presence/absence of MRD by RE-ALB tube was checked in comparison to in-use 5-color panel. Acceptable criteria are 100% concordance on presence or absence of neoplastic population and on all markers evaluated [4] Secondly, we compared the percentage population of interest, correlation coefficient was done between the new (RE-ALB tube) and in use method, r2 value was obtained for the MRD and diagnostic B-ALL samples [4,5]. The acceptance criteria were CV of <25% and when value is near Lower limit of quantification (LLOQ) acceptance criteria CV < 35% was regarded as acceptable for population of interest between RE-ALB and 5 color panel [4]. All MRD negative samples were compared using percentage of CD19 positive viable cells by both methods. Comparing median fluorescent intensity for markers was not possible as the antibodies were tagged to different fluorochromes however we compared the overall staining intensity (dim, moderate, bright) for the backbone markers like CD19, CD10, CD34, CD38, and CD45.
To verify precision and to check for any interoperator variation, RE-ALB tube was processed three times a day for a patient sample and one normal sample by three different operators each time to verify precision and to account for any interoperator variation. Percent positivity for CD19 positive viable cells and X-median for markers like CD19, CD45, CD10, CD34 was compared between the three tubes and concordance between final diagnosis was noted. LOD was determined by performing serial dilution (5 dilutions) of a BM with B-ALL blasts with another BM which was performed for staging of Lymphoma.Dilutions were performed to obtain 0.001% blasts in the last dilution. For the establishment of LLOQ we spiked leftover nonleukemic uninvolved staging bone marrow aspirates with B-ALL blasts from a diagnostic sample. Limit of blank (LOB) was performed using 4 normal/staging BM samples processed for B-MRD analysis (RE-ALB tube) and about 5,00,000- 7, 00,000 cells were studied [1].
Results
Normal samples showed presence of hematogones with expected marker positivity for CD19, CD10, CD34, CD38 and CD20 and showing classical maturation pattern of hematogones. Plasma cells with bright CD38 positivity, CD19 positivity were noted in these samples as expected constituting ~1% of cells. We found 100% concordance between the 2 methods in reporting diagnostic B-ALL samples (n=10 samples), MRD samples (n=25 samples) and CAP- PT samples (n=4). In addition, staining expression of all the positively expressed antibodies were found to be equivalent. All antibodies reported as negative in 5-color assay was negative in RE-ALB tube. Internal controls identified in each RE-ALB tube run was as expected and satisfactory.
On comparing percent population of interest, percentage CV was found to be at 17.10%. On linear regression analysis, r2 value is 0.99 with intercept at 0.
Precision CV for percent positivity for CD19 viable cells was 6% and for X-median precision mean CV was 4% (range - 1-7%). Final diagnosis was also in concordance during precision evaluation.
LLOQ was found to be 43 events (precision CV of 9.8%) and sensitivity of the assay was 0.005% on acquisition of ~ 1,000,000 events. LOD was found to be 20 events (0.001% MRD). Hence MRD positive events cut off was taken at 50 events.
LOB was found to be two events and this was subtracted from final MRD events.
The BM cellularity was adequate in the majority of samples to obtain more nearly 1 million for MRD analysis. The median events acquired in these 25 MRD assays were 1.5 million events with a range from 580085 – 2270364 events.
It took approximately 40–45 min for processing and 5-7 minutes for the acquisition of one sample for MRD. In comparison, 5 color tubes took 60- 75 mins for processing, 30 minutes for acquisition. RE-ALB tubes reduced 10–15 min for processing and 15–20 min for the acquisition as compared to the in house MRD panel by 5 color tubes.
Of 25 MRD samples, 4 samples were MRD positive. Of which one sample showed MRD positivity only using backbone markers like CD19, CD10, CD38, the other three samples were detected based on LAIPs of additional drop in markers.
MRD levels ranged from (0.04% – 0.26%) (Table 2). With RE- ALB using LLOQ as a cut off level of MRD positivity, sensitivity of at least 0.01% was achieved in all cases.
Presence of hematogones in the background were noted along with MRD in 2 out of 4 samples positive for MRD.
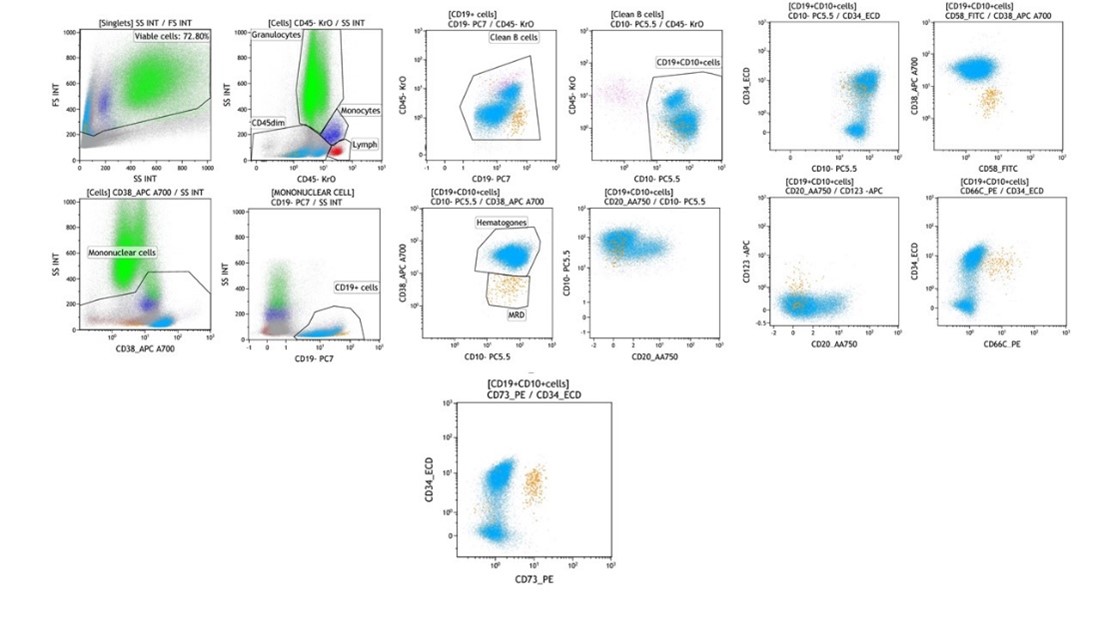
Figure 1: This case showed admixture of hematogones (stage 1 and 2) (aqua) along with MRD (Mustard). Residual blasts showed CD19+(overexpression), CD10+CD34+, CD58 (homogenous), CD38 dim. There is abberant expression of drop in markers CD66C, CD73 and CD123 (dim partial).
Discussion
In the present study, we validated dried reagent tube Duraclone Re-ALB tube, a high-sensitivity FC-MRD assay tube which utilizes bulk-lysis method for sample processing and hence allowing acquisition of 1.5 million events in the majority of samples (71%) in our lab, which allowed us to detect low-level MRD with the highest sensitivity of 0.005% (5-in-105) utilizing lesser volume of BM sample as compared to multiples tubes while using 5 color MRD assay to achieve the recommended assay sensitivity. The assay sensitivity parameters, and LOD, LLOQ were determined using spiking and dilution experiments. These results are almost same as defined by recently published highly sensitive B-ALL FC-MRD assay by Euroflow Consortium (i.e., LOD at 10 events and LLOQ at 40 events) [1] To increase the applicability and accuracy of FC-MRD, it is desirable to incorporate the markers that allow the highest discrimination of residual blasts in the background of hematogones and other hematopoietic cellular elements. Elaine Coustan-Smith et al. introduced 22 new markers potentially useful for B-ALL FC-MRD evaluation [8]. Our FC-MRD assay included the new markers like CD73, CD66c, CD123, CD58 along with six backbone markers (CD10, CD19, CD20, CD34, CD38, and CD45) leaving one drop in channel still vacant.
Similarly, Prashant et al showed findings suggested that simultaneous use of new markers along with backbone markers in 8–10 color FC assay may have better MRD detection power even at standard sensitivity (>0.01%) as compared to standard 4–6 color FC assay. (1) LAIP markers can be added to RE-ALB tube as per laboratory policy.
Additionally, this method keeps the advantage of FC-MRD of short turnaround time. It takes 45 min for processing and acquisition of one sample FC-MRD, which can be reduced further using batch processing. In the previous year, we performed multiple tubes of 5-color assay for our MRD samples for which at least 4-5 hours would go into processing, acquisition and analyzing one case of MRD.
Dried, room-temperature tubes offer advantages over liquid format, as they are stable for at least 12 months and, unlike liquid reagents, are made with a single lot for each antigen. In our laboratory, using dried format tubes effectively eliminates the need to individually titrate, pipette, create cocktails, and performing lot validation for individual antibodies. Other advantages are APC tandem dyes are relative unstable which limits the stability for liquid format cocktails containing them, which does not happen with the dried reagent tubes [5].
In this study, we have demonstrated complete equivalency between a novel dried reagent combination and liquid format tubes in sample interpretation, staining percentages, and intensity expression of positive markers in a variety of sample types with diagnoses including B-Acute lymphoblastic leukemia samples and MRD–BALL. MRD could be identified clearly from background hematogones as was seen in 2 out of 4 MRD positive patient cases. We found 100% concordance in final diagnosis in all the patient samples studied including CAP- PT samples for MRD evaluation.
There is only one other study performed by Lakhdar Bouriche et al on Duraclone RE-ALB tube which found that these tubes were highly comparable with the liquid-antibody tube [3]. One interesting point is our demonstration of validation journey of the tube in our laboratory with patient samples, CAP-PT samples and applicability of this tube in our laboratory thus proving that a laboratory could easily upgrade the B-ALL MRD assay from 5 color to 10 color allowing reproducibility, while reducing on processing and analyzing times and allowing better inventory management.
To conclude, use of this tube helps improve workflow, overall efficiency and performance of our laboratory. Use of this tube can also help standardize results between various labs and is easily reproducible.
Ethics declarations
Patient consent statement: This study was done by utilizing left over sample which arrived in the laboratory. The patient details are anonymized to protect patients’ privacy, hence patient consent is not required.
Ethics statement: The medical ethics committee of Mohamed Rashid bin university declared that a technical validation in the laboratory is not medical research and therefore does not fall within the scope of ethics committee.
Conflicts of interest: The authors declare ‘no conflicts of interests.
Funding: The authors have no relevant financial or non -financial interests to disclose.
No funding received by any author.
References
- Tembhare PR, Subramanian PG, Ghogale S, Chatterjee G, Patkar NV, et al. A High-Sensitivity 10-Color Flow Cytometric Minimal Residual Disease Assay in B-Lymphoblastic Leukemia/Lymphoma Can Easily Achieve the Sensitivity of 2-in-106 and Is Superior to Standard Minimal Residual Disease Assay: A Study of 622 Patients. Cytometry Part B, 2020; 98B: 57– 67.
- Sonal Jain, Anurag Mehta, Dushyant Kumar. Evaluating New Markers for Minimal Residual Disease Analysis by Flow Cytometry in Precursor B Lymphoblastic Leukemia. ndian J Hematol Blood Transfus, 2018; 34(1): 48–53. doi: 10.1007/s12288-017-0845-5.
- Bouriche L, Bernot D, Nivaggioni V, Arnoux I, Loosveld M. Detection of Minimal Residual Disease in B Cell Acute Lymphoblastic Leukemia Using an Eight-Color Tube with Dried Antibody Reagents. Cytometry B Clin Cytom, 2019; 96(2): 158-163. doi: 10.1002/cyto.b.21766.
- Clinical and Laboratory Standards Institute (CLSI). Validation of Assays Performed by Flow Cytometry. 1st ed. CLSI guideline H62. Clinical and Laboratory Standards Institute, USA, 2021.
- Hedley BD, Keeney M, Popma J, Chin-Yee I. Novel lymphocyte screening tube using dried monoclonal antibody reagents. Cytometry B Clin Cytom, 2015; 88(6): 361-370. doi: 10.1002/cyto.b.21251.
- Pulkit Rastogi, Man Updesh Singh Sachdeva. Flow Cytometric Minimal Residual Disease Analysis in Acute Leukemia: Current Status. Indian J Hematol Blood Transfus, 2020; 36(1): 3–15.
- Owens MA, Vall HG, Hurley AA, Wormsley SB. Validation and quality control of immunophenotyping in clinical flow cytometry. J Immunol Methods, 2000; 243(1-2): 33-50. doi: 10.1016/s0022-1759(00)00226-x.
- Coustan-Smith E, Song G, Clark C, Key L, Liu P et al. New markers for minimal residual disease detection in acute lymphoblastic leukemia. Blood, 2011; 117(23): 6267– 6276.

