Rhabdomyomatous Mesenchymal Hamartoma: An Acquired Plaque-Type Variant Case Report
Robyn Okereke1,*, Anthony Linfante2
1Oregon Health & Sciences University, Department of Dermatology, Portland, Oregon, United States
2University of Texas Medical Branch at Galveston, Galveston, Texas, United States
Received Date: 02/08/2025; Published Date: 05/09/2025
*Corresponding author: Robyn Okereke, Oregon Health & Sciences University, Department of Dermatology, Portland, Oregon, United States
Abstract
Rhabdomyomatous Mesenchymal Hamartoma (RMH) is a rare, typically congenital, benign hamartomatous tumor of the dermis and subcutaneous tissue. The lesions consist of disordered collections of mature skeletal muscle and varying amounts of additional mesenchymal-derived tissue. RMH is usually a solitary, polypoid, midline lesion of the head or neck in an infant or child, which may occur in conjunction with multiple ectodermal/mesodermal abnormalities. More recently, patterns of RMH occurring in adults have been described. Herein, we present an adult patient who presented with an RMH on the chin, followed by a clinical course and histologic evaluation. This case demonstrates an uncommon variant of an acquired, plaque-type RMH with unique histologic properties.
Keywords: Primary Cutaneous Cribriform Apocrine Carcinoma; Primary; Cutaneous; Apocrine; Cribriform
Introduction
Rhabdomyomatous Mesenchymal Hamartoma (RMH) is a rare cutaneous lesion first described in the literature in 1989. As of 2024, there have been over 80 cases reported in the literature since the initial case in 1986 [1-3]. Few cases (20.8%) of RMH occur with uncommon medical syndromes [4]. RMH classically occurs in infants and children (80%), with a minority (20%) of cases in literature occurring in adults [5]. On histopathological analysis, RMH classically displays mesenchymal tissue (with a predominance of skeletal muscle) within the reticular dermis. However, in adult-onset RMH, the sole dermal proliferation of skeletal muscle fibers has been observed on histology [5]. Upon reviewing cases in the literature, we apprise a unique case of a rare phenomenon, RMH, presenting as an indurated plaque on the chin in an adult, and further describe observed histological deviations.
Case Presentation
A 40-year-old African-American male presented with a skin lesion on his right chin for many years; the patient was unsure if it had been present since his childhood. On physical exam, a 3 cm, subcutaneous indurated plaque with central dell was observed on the right chin. No intraoral connection was noted. The clinical diagnosis was an epidermal inclusion cyst (EIC). The patient underwent surgical excision with 3 mm margins and the specimen was sent for histopathologic analysis. At the time of this manuscript writing, no clinical recurrence has occurred.
Histopathology of the specimen was notable for abundant haphazard bundles of mature striated muscle admixed with adipose tissue, sclerotic collagen bundles, nerve bundles, and prominent folliculosebaceous adnexa within the reticular dermis and subcutaneous tissue (Figures 1, 2). There was no evidence of cystic elements or malignancy on histopathologic evaluation. Thus, the histopathologic findings were consistent with a diagnosis of RMH.
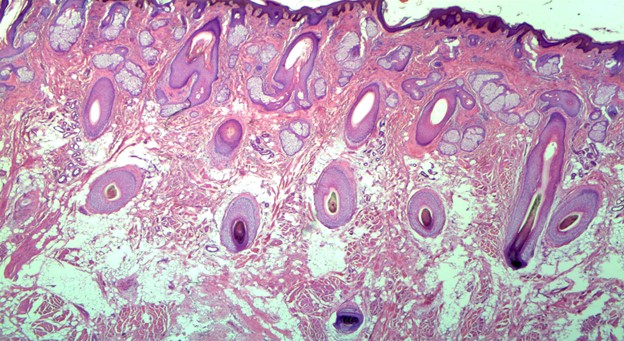
Figure 1: Hematoxylin & Eosin Stain displaying abundant haphazard bundles of mature striated muscle admixed with adipose tissue, sclerotic collagen bundles, nerve bundles, and prominent folliculosebaceous adnexa within the reticular dermis and subcutaneous tissue (20x).
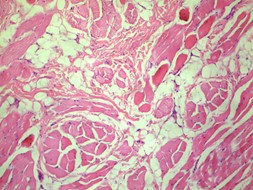
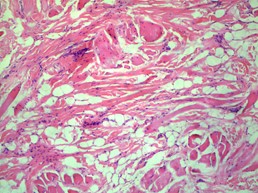
Figure 2(a,b): Proliferation of well differentiated skeletal muscle within the dermal and subcutaneous layers can be appreciated at high power fields (100x).
Discussion
While the definitive etiology is unknown, RMH is believed to be derived from aberrant embryonic migration of mesodermal tissue and the presence of a disorganization gene found in mouse models [2,5]. The striated muscle component is believed to be comprised of superficial muscular aspects originating from the second branchial arch which underwent erroneous migration, resulting in characteristic striated muscle bundles just below the epidermis [3,7]. More recently, elongation hyperplasia of striated muscles has been described as a potential etiology of adult-onset RMH.
RMH usually presents as a flesh-colored, pedunculated lesion or nodule, but can also be seen as a papule that may be subcutaneous or sessile. Most (88.2 %) of the reported lesions have been localized to areas with immediately underlying striated muscle on the head and neck region [7]. At least 14 cases in the literature have specified RMH to have occurred on the chin specifically, as observed in our patient [3,6]. The proximity of the mentalis and platysma muscles in the mandible to the subcutaneous tissue, leading to direct confluence with the dermis likely offers anatomical predisposition to the development of RMH [3,6]. Other lesions in the literature have been detected on the sacral area, hallux, digits, orbit, anus, chest, back, and vagina [1,2,4,8].
More recently, the literature has made efforts to classify RMH according to age demographics; congenital type (in those under 18 years of age) or acquired type (in those above 18 years of age). A nodular-type lesion morphology predominates in congenital RMH (74%), while plaque-type composition (43.8%) is more common in acquired RMH [3]. Among reported cases in the literature, it is becoming evident that the chin is a common location for acquired RMH (43.8%) [3].
In addition to distinguishing clinical morphologies in congenital and acquired RMH, more recently, differentiating histopathologic patterns have also been recognized. All RMH tumors display a histologically normal epithelium and papillary dermis, with a disorganized reticular dermis. In congenital RMH, the reticular dermis and subcutaneous layers often (80%) contain a predominance of highly differentiated skeletal muscle, mesenchymal elements (adipose tissue, sclerotic collagen bundles, elastic fibers, and blood vessels), and occasional neural elements. Acquired plaque type usually features solely skeletal muscle fiber dermal proliferation, with no other mesenchymal tissue propagation.3 A minority (33%) of acquired RMHs have been observed to display hamartomatous proliferation with alternative mesenchymal elements in addition to striated skeletal muscle [3]. However, hamartomatous proliferation of multiple types of mesenchymal elements has only been observed in acquired nodular RMH (75%) [3]. Therefore, our case represents a unique variant of acquired plaque type RMH with hamartomas proliferation of multiple mesenchymal elements in addition to skeletal muscle fibers. This is notable, as the acquired plaque type is expected to solely comprise striated skeletal muscle on histology, and a heterogenous mesenchymal composition is also less commonly observed in adult-onset RMH [3,6].
RMH has a favorable prognosis in adults and no risks for complications following treatment in these patients. Although treatment is not necessary unless lesions become symptomatic, surgery is the main treatment for RMH, though laser treatments have been described [9]. Some RMH lesions spontaneously regress, and treated lesions show no recurrence [2]. It is recommended to observe subcutaneous and plaque‐type variants where removal is not easily executed [6].
Conclusion
Considering its rare prevalence in literature, and compounding, its atypical occurrence in adults, we have drawn further attention to the increasing prevalence of acquired plaque-type RMH in adults, which should be considered in the differential diagnosis for lesions of the head and neck. We described the first case of acquired plaque-type RMH, which uniquely displayed additional mesenchymal elements in addition to characteristic dermal skeletal muscle fiber proliferation, in contrast to previous cases, which have described sole skeletal muscle proliferation on histology.
Limitations: Our patient could not specifically quantify the duration of the lesion. Therefore, we recognize that we cannot completely exclude the possibility that the mass was present in childhood, and may have gone unnoticed. Additionally, the subcutaneous location of the mass may have resulted in delayed appreciation by the patient, as well as subsequent presentation. Finally, a clinical photograph of the lesion was not collected, as it was initially believed to be an EIC.
Conflict of interest statement: The authors have no conflict of interest nor funding sources to disclose.
References
- Nguyen CP, Aboutalebi S JM, Stetson CL. Incidental rhabdomyomatous mesenchymal hamartoma. Proceedings (Baylor University. Medical Center), 2020; 34(1), 161–162. https://doi.org/10.1080/08998280.2020.1801087.
- Rosenberg AS, Kirk J, Morgan MB. Rhabdomyomatous mesenchymal hamartoma: an unusual dermal entity with a report of two cases and a review of the literature. Journal of Cutaneous Pathology, 2002; 29: 238-243. https://doi.org/10.1034/j.1600-0560.2002.290407.x.
- Ehara Y, Yoshida Y, Kataoka S, Yoshioka, et al. Rhabdomyomatous Mesenchymal Hamartoma: A New Proposed Clinical Classification of Adult Onset Acquired Type. Acta dermato-venereologica, 2022; 102: adv00627. https://doi.org/10.2340/actadv.v101.170.
- Yang SH, McCalmont TH, Ahn GG, et al. Supraorbital cutaneous fetal rhabdomyoma of intermediate type: a case report. The American Journal of dermatopathology, 2014; 36(5): e93–e96. https://doi.org/10.1097/01.dad.0000441941.83273.c5.
- Sampat K, Cheesman E, Siminas S. Perianal rhabdomyomatous mesenchymal hamartoma. Annals of the Royal College of Surgeons of England, 2017; 99(6): e193–e195. https://doi.org/10.1097/01.dad.0000441941.83273.c5.
- Lee YH, Yao XF, Wu YH. Plaque-Type Variant of Acquired Rhabdomyomatous Mesenchymal Hamartoma on the Chin: A Case Series. The American Journal of dermatopathology, 2021; 43(12): 908–912. https://doi.org/10.1097/DAD.0000000000002016.
- Sayan A, Diniz G, Mert M, et al. Rhabdomyomatous mesenchymal hamartoma developed at an unexpected location. Hamartoma mesenquimal rabdomiomatoso en una ubicación inesperada. Archivos argentinos de pediatria, 2019; 117(5): e519–e522. https://doi.org/10.5546/aap.2019.eng.e519.
- Fontecilla NM, Weitz NA, Day C, et al. Rhabdomyomatous mesenchymal hamartoma presenting as a skin tag in a newborn. JAAD case reports, 2016; 2(3): 222–223. https://doi.org/10.7181/acfs.2017.18.4.292.
- Kim H, Chung JH, Sung HM, et al. Rhabdomyomatous Mesenchymal Hamartoma Presenting as a Midline Mass on a Chin. Archives of craniofacial surgery, 2017; 18(4): 292–295. https://doi.org/10.5546/aap.2019.eng.e519.

