Giant Polycystic Liver Disease Revealed by Abdominal Mass: A Case Report and Review of the Literature
Bachar Amine, Benzidane Kamal*, Brahmi Soufiane, Essaidi Zakaria, El Abbassi Taoufik and Bensardi Fatima Zahra
Hassan II University of Casablanca, Morocco Institution: Department of Visceral Surgery, Ibn Rochd University Hospital, Casablanca, Morocco
Received Date: 14/07/2025; Published Date: 12/08/2025
*Corresponding author: Benzidane Kamal, Hassan II University of Casablanca, Morocco Institution: Department of Visceral Surgery, Ibn Rochd University Hospital, Casablanca, Morocco
Abstract
Background: Polycystic Liver Disease (PLD) is a rare inherited disorder characterized by the presence of multiple hepatic cysts. While often asymptomatic, large or numerous cysts may lead to abdominal distension, gastrointestinal symptoms, or mass effect complications.
Case Presentation: We report the case of a 59-year-old female with no significant medical history, presenting with progressive abdominal distension and right hypochondrial pain. Imaging revealed a massively enlarged liver with numerous cysts, the largest measuring over 20 cm, consistent with PLD. Surgical fenestration of the hepatic cysts was performed, resulting in symptomatic relief and favorable postoperative evolution.
Discussion: PLD is most often associated with autosomal dominant polycystic kidney disease, but isolated forms exist. The diagnosis relies heavily on imaging, particularly CT and MRI. Symptomatic patients may benefit from surgical interventions such as cyst fenestration, hepatectomy, or, in advanced cases, liver transplantation.
Conclusion: Although usually benign and asymptomatic, PLD can present as a giant abdominal mass with significant clinical impact. Early recognition and appropriate surgical management are essential in improving outcomes and quality of life.
Keywords: Polycystic liver disease; Hepatic cysts; Cyst fenestration; Gigot classification; Schnelldorfer classification
Introduction
Polycystic Liver Disease (PLD) is a rare genetic condition characterized by the development of multiple fluid-filled hepatic cysts. It can occur in isolation or more commonly in association with polycystic kidney diseases, particularly the autosomal dominant form (ADPKD). While most cases remain asymptomatic, some patients may develop symptoms due to mass effect, including abdominal pain, distension, early satiety, and gastrointestinal dysfunction. Advanced cases may present with severely enlarged liver volumes, leading to a significant decline in quality of life. Diagnosis is primarily radiological, based on ultrasound, Computed Tomography (CT), or magnetic resonance imaging (MRI). Treatment options range from conservative monitoring to surgical cyst fenestration or liver transplantation, depending on the severity and symptomatology. We report a case of symptomatic PLD in a woman presenting with a giant abdominal mass and discuss the diagnostic and therapeutic strategies.
Case Report
59-year-old female patient with no significant personal or familial past medical history. The history of the illness dates back 18 years, with the gradual onset of a dull, pressure-like pain in the right hypochondrium, without any associated digestive symptoms. Over the past five years, the clinical picture was marked by the appearance of a progressively enlarging mass in the right hypochondrium, eventually occupying almost the entire abdomen. This was associated with dyspepsia and bowel habit disturbances in the form of constipation, all evolving in a context of preserved general health.
Clinical examination revealed a patient in good general condition, with a performance status (PS) of 0 and normally colored conjunctivae. Abdominal examination showed a multilobulated mass occupying nearly all abdominal quadrants, measuring 25 cm in its longest axis, painless, firm, mobile with respiration but fixed to deep planes, and without signs of overlying inflammation. Pelvic examinations revealed no abnormalities.
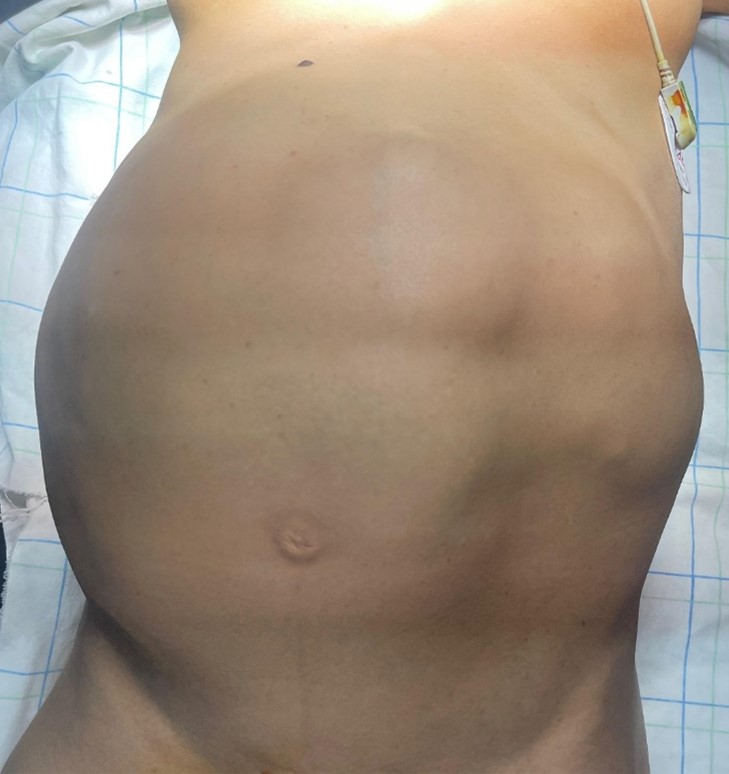
Figure 1: image of the patient abdomen, showing a multilobulated mass occupying nearly all abdominal quadrants. The patient presented with an abdominopelvic CT scan showing.
An enlarged liver (measuring 20 cm in craniocaudal height), with irregular contours and homogeneous enhancement after contrast administration. The liver harbored multiple cystic lesions (more than 10), involving all hepatic segments. These lesions were round to oval, fluid-filled, with thin walls, no post-contrast enhancement. Some contained septations, and others showed peripheral calcifications. No communication with the biliary tract was noted.
The largest cyst had an exophytic growth pattern, located in the right lobe of the liver, measuring 211 x 118 mm, with a height of 235 mm.
Complementary ultrasound confirmed that these cysts had thin walls. Some contained finely echogenic material, and others had internal septations.
The kidneys were of normal size, with regular contours and good vascular nephrography. Multiple bilateral renal cysts were observed, the largest being located at the lower pole of the right kidney, measuring 24 mm in diameter, with a simple appearance, classified as Bosniak I.
On conclusion, the imaging features of both hepatic and renal cystic involvement are highly suggestive of hepatorenal polycystic disease, with the presence of a large compressive hepatic cyst.
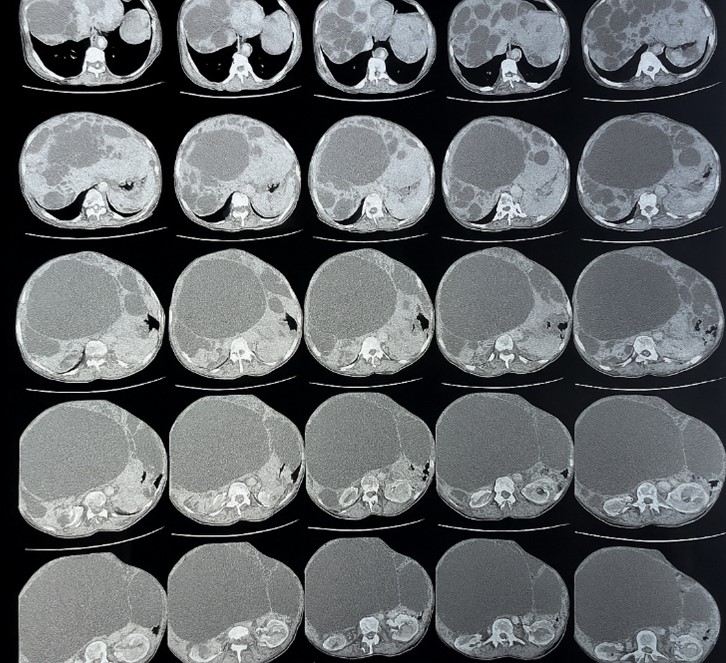
Figure 2: CT scan image showing multiple hepatic and renal cysts suggesting a Polycystic liver disease.
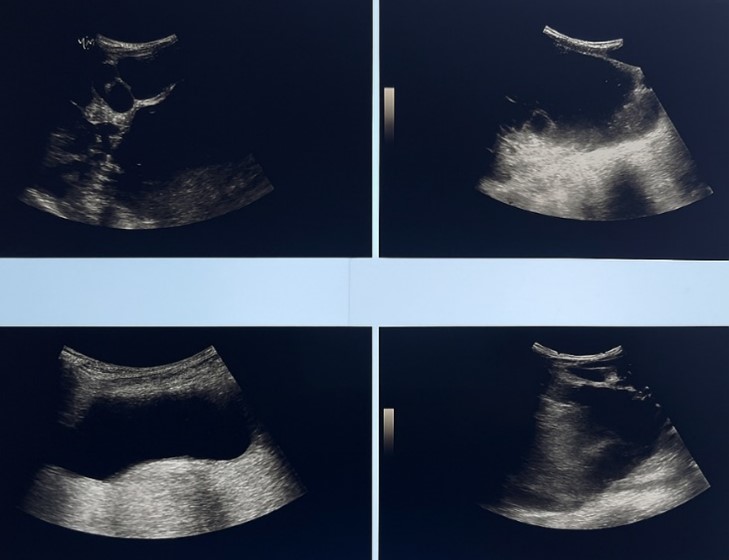
Figure 3: ultrasound images showinga multiple hepatic cysts containing finely echogenic material, and others had internal septations.
Biologically, the patient presented with:
- Negative hydatid serology
- Normal liver function tests, with AST/ALT levels at 83/80 IU/L
- Normal renal function
- Normal cholestasis panel
The patient was considered for simultaneous surgical management of both hepatic and renal cysts. However, given the normal renal function, a conservative approach was decided for the renal cysts, with no surgical intervention indicated.
The patient was admitted to the operating room. Surgical access was achieved through a midline incision extending above and below the umbilicus.
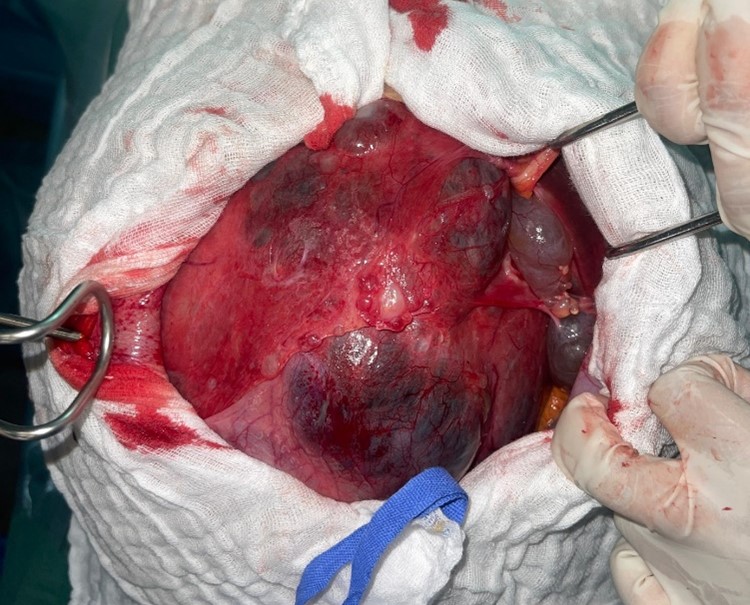
Figure 4: Per operative image after making the incision.
Intraoperative exploration revealed an enlarged liver extending to the left hypochondrium, containing multiple cysts, the largest of which occupied the entire right lobe of the liver, displacing the digestive structures (stomach, duodenopancreatic loop, transverse colon, and small intestine) towards the left side. The spleen appeared normal.
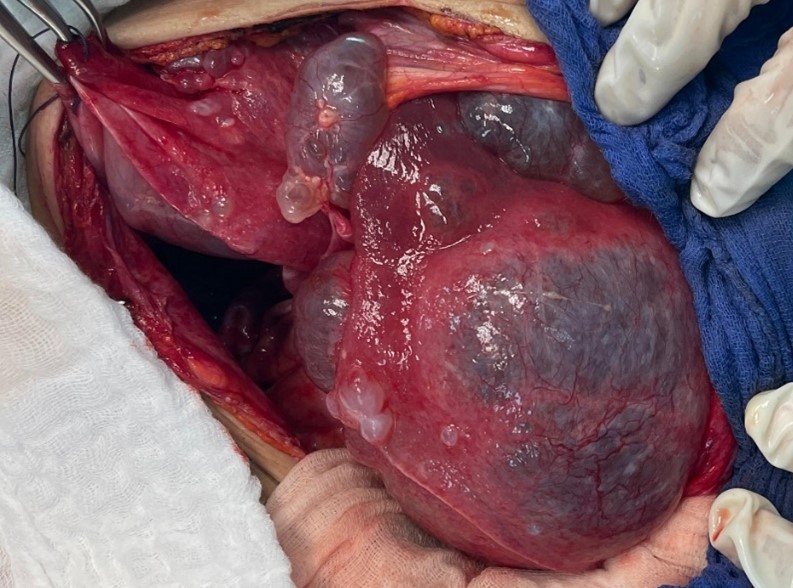
Figure 5: Per operative image showing a larger hepatic cyst located in the left liver.
The surgical procedure consisted of a fenestration of nearly all hepatic cysts, filled with transparent fluid, with wide drainage of both the peritoneal cavity and the residual cavity of the right hepatic cyst, using Salem tubes.
Biopsies of the cyst walls were sent to the pathology laboratory and were consistent with polycystic liver disease, with no signs of malignancy. Cytological analysis of the cyst fluid revealed no cellular atypia, and bacteriological and parasitological studies showed no infectious agents, particularly no evidence of Echinococcus granulosus.
Postoperative course was marked by the drainage of a total of 700 cc of serous fluid during the first three days, followed by a gradual decrease in volume until the 6th postoperative day, at which point only a few cc of serous fluid was collected. The drains were removed, and the patient was discharged on postoperative day 7.
Follow-up at 1 week, 15 days, and 1 month postoperatively revealed no abnormalities.
Discussion
Polycystic Liver Disease (PLD) is a clinical condition characterized by the presence of more than 20 cysts in the liver [1]. it is an uncommon disorder that manifests in association with three inherited conditions: Autosomal Dominant Polycystic Liver Disease (ADPLD), autosomal dominant polycystic kidney disease (ADPKD), and Autosomal Recessive Polycystic Kidney Disease (ARPKD) [2]. PLD can present either as an autosomal dominant form or as an isolated condition without associated kidney involvement [1].
Although the genetic basis and pathophysiological mechanisms of PLD are not yet fully elucidated, the condition is mainly attributed to abnormalities in ciliogenesis and deficiencies in Endoplasmic Reticulum (ER) quality control. In ADPKD and ARPKD, mutations that result in loss of function in genes directly implicated in cilia formation—such as Pkd1, Pkd2, Pkhd1, and Dzip1l—can drive the development of cysts in both the liver and kidneys. Furthermore, mutations in genes responsible for ER-associated processes including protein folding, trafficking, and maturation—such as PRKCSH, SEC63, ALG8, ALG9, GANAB, and SEC61B—can disrupt the synthesis or function of polycystin-1 (PC1) and polycystin-2 (PC2), or accelerate their degradation, thereby contributing to isolated liver cysts or combined hepatic and renal cyst formation. More recently, defects in LRP5, which impairs the canonical Wnt signaling pathway, have also been linked to hepatic cyst development [2].
The condition is characterized by the gradual formation of renal cysts, resulting in kidney enlargement and, ultimately, progression to renal failure [3]. Liver cysts typically follow a progressive course, enlarging in both size and number over time, which can result in massive and debilitating hepatomegaly with usually not impaired liver function [1,2]. Key factors contributing to the progression of polycystic liver disease include older age, female gender, estrogen exposure (such as from oral contraceptives or hormone replacement therapy), the degree of renal impairment, and the overall burden of renal cysts [4]. Regardless of the specific genes involved in its pathogenesis, the clinical progression of different forms of polycystic liver disease generally follows a similar pattern, characterized by a steady increase in both the number and size of hepatic cysts [5].
Despite an average liver volume increase of approximately 1.8% every 6 to 12 months in PLD patients [5], around 95–98% of individuals with Polycystic Liver Disease (PLD) remain clinically asymptomatic [4]. However, a small subset experiences debilitating symptoms and a marked decline in quality of life. The clinical presentation largely depends on the number, size, location, and distribution of hepatic cysts, with most symptoms resulting from mass effect or compressive phenomena due to hepatomegaly.
Abdominal distension is commonly seen in cases of significant liver enlargement, while tension on Glisson’s capsule may lead to abdominal pain. When cysts compress the stomach or duodenum, patients may report early satiety, postprandial fullness, gastroesophageal reflux, or intolerance to food. These manifestations can lead to a reduced food intake, ultimately causing weight loss and, in severe cases, malnutrition. Notably, malnutrition often goes unrecognized initially, as the increased abdominal girth and liver volume can obscure body weight loss.
Cysts located in the hepatic dome can elevate the diaphragm, restricting thoracic expansion and causing dyspnea. Some patients also report lower back pain, which is likely due to the pressure exerted on nearby structures. When symptoms are pronounced, they can severely impact daily functioning and self-image. The increasing abdominal volume may hinder simple tasks such as bending forward and can contribute to reduced self-esteem.
Although vascular compression by liver cysts is uncommon, it can occur. For instance, cysts in the hepatic hilum may compress the portal vein, potentially leading to Portal Hypertension (PH). While cirrhosis is rare, complications of PH, including splenomegaly, ascites, variceal bleeding, and hepatic encephalopathy, may arise in advanced cases with fibrosis [4].
Inferior Vena Cava (IVC) compression is often seen but seldom results in hemodynamic compromise. In contrast, hepatic vein compression is more frequent and can result in the development of intraparenchymal and subcapsular veno-venous collateral circulation, increasing the risk of bleeding during surgery. While intrahepatic bile ducts are commonly compressed, extrahepatic bile duct involvement is rare. Nevertheless, bile duct obstruction may cause jaundice in certain cases. Hepatic insufficiency is uncommon and typically reflects an advanced disease stage, liver function tests are typically within normal ranges in most PLD patients, as the hepatic parenchyma remains largely preserved [4,5].
Polycystic Liver Disease (PLD) is typically diagnosed when more than 20 hepatic cysts are present. However, in individuals with a family history of isolated Polycystic Liver Disease (PCLD), the diagnosis can be established with as few as five cysts. Distinguishing between the various forms of PLD can be challenging. For example, patients with PCLD may also develop renal cysts, while those with autosomal dominant or recessive polycystic kidney disease (ADPKD or ARPKD) may present with hepatic cysts as a predominant feature. In the absence of a clear family history, differentiation between these entities often requires genetic testing [5].
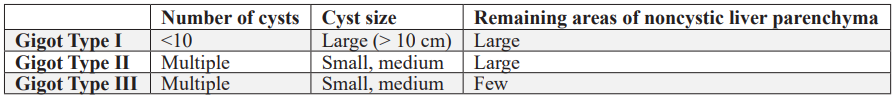
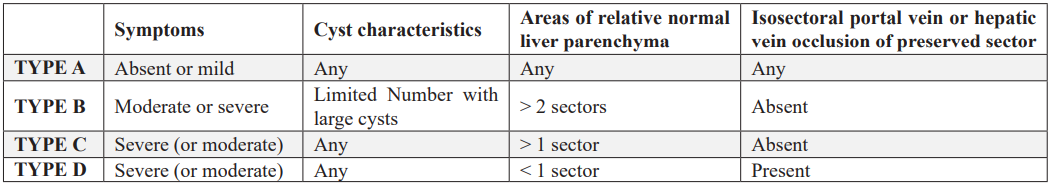
Currently, two main clinical classification systems are used to assess PLD: the Gigot classification [6] (Table 1) and the Schnelldorfer classification [7] (Table 2). Both systems categorize the disease based on the number and size of hepatic cysts as well as the volume of preserved liver parenchyma. The Schnelldorfer classification further incorporates the functional inflow and outflow of the remaining liver segments, making it particularly helpful in guiding therapeutic decisions.
According to Gigot’s classification, Polycystic Liver Disease (PLD) is categorized into three types. Type I involves fewer than ten hepatic cysts, each exceeding 10 cm in diameter. Type II is characterized by numerous cysts interspersed with substantial regions of preserved, noncystic liver tissue. Type III presents with predominantly small- to medium-sized cysts and minimal remaining unaffected parenchyma [6].
Table 1: Gigot's classification [6].
On the other hand, the Schnelldorfer classification outlines four distinct types. Type A includes patients with no or only mild symptoms, regardless of cyst size. Type B features moderate to severe clinical symptoms with a mixture of large and small cysts and significant preserved parenchymal regions. In type C, symptoms are severe, with large and small cysts present and a marked reduction in healthy liver tissue. Type D also involves severe symptoms, but with extensive cystic involvement and very limited remaining normal parenchyma [7].
Table 2: Schnelldorfer's classification [7].
For patients with Polycystic Liver Disease (PLD) and preserved renal function, contrast-enhanced Computed Tomography (CT) is the primary imaging modality for radiologic evaluation. Hepatic cysts typically appear as well-defined, fluid-filled lesions with thin, non-enhancing, avascular walls on CT, unless complicated by intracystic hemorrhage. In rare cases—less than 2%—fine calcifications may be seen, often as a sequela of bleeding within the cysts [1]. In most instances, CT findings are sufficient to guide therapeutic decision-making. When evaluating diffuse PLD, critical parameters include the size and distribution of cysts, as well as both the qualitative and quantitative assessment of the remaining functional liver tissue. Particular attention should be paid to relatively preserved liver regions that might still be affected by venous outflow obstruction due to hepatic vein compression. Important aspects to assess in patients with PLD include the presence of compression at the confluence of the hepatic veins, the existence or absence of intrahepatic collateral circulation, and any parenchymal heterogeneity observed after contrast injection in areas with compromised venous drainage. Additionally, the identification of hypervascular lesions may suggest the presence of Regenerative Nodular Hyperplasia (RNH). CT imaging also enables evaluation of potential compression of portal and biliary structures at the hepatic hilum, detection of ascites, and assessment of the patient's nutritional status by measuring muscle mass, particularly the thickness of the anterior abdominal wall and the psoas muscles.
Magnetic Resonance Imaging (MRI) is particularly valuable for evaluating PLD-related abdominal pain, especially when intracystic hemorrhage is suspected—best visualized using T1-weighted sequences. In rare cases, MRI with T2-weighted imaging can help detect hilar biliary strictures. However, interpreting T2 images may be challenging due to the multitude of cysts, and the use of hepatobiliary contrast agents may improve diagnostic clarity in complex presentations [1].
The treatment options for PLD depend on the severity of disease. Individuals without symptoms undergo conservative management (i.e., watchful waiting). Treatment of symptomatic patients includes nonsurgical and surgical options. Classification systems by Gigot and Schnelldorfer are commonly used to characterize the severity of PLD and to outline a potential treatment.
In patients with Gigot type I PLD, cyst aspiration and sclerosis are the recommended treatment. The success of the procedure correlates with the cyst size. Laparoscopic fenestration of hepatic cysts (a less common procedure since the results are often short lived) is used in patients with multiple cysts in Gigot types I and II or Schnelldorfer type B PLD. In individuals with Gigot type II or Schnelldorfer type C, the treatment includes combined hepatic resection and fenestration. A liver transplantation is reserved for patients with impaired liver function or for whom a liver resection is not feasible (Gigot type III or Schnelldorfer type D) [8].
Patients with Polycystic Liver Disease (PLD) often undergo a variety of therapeutic interventions, frequently requiring repeated treatments over the course of the disease. Currently, the only available pharmacologic approach for symptomatic individuals in whom surgery is either not feasible or not warranted is the off-label use of somatostatin analogs. Advances in the understanding of the molecular pathways involved in PLD pathogenesis have facilitated the identification of several therapeutic targets, which are now being investigated in both preclinical studies and clinical trials such as mammalian target of rapamycin (mTOR) and Ursodeoxycholic acid (UDCA) [8].
For many years, surgical fenestration—performed either via laparotomy or laparoscopy—was considered the primary therapeutic option for polycystic liver disease (PLD) [1]. The procedure involves resecting the majority of the cyst wall to allow the cystic fluid to drain into the peritoneal cavity, where it can be reabsorbed. Deeper cysts can often be reached through previously opened ones, allowing for sequential fenestration of multiple cysts. The technique carries a low operative risk, with postoperative ascites and biliary fistulas being the main complications, particularly if a bile duct is inadvertently transected during cyst wall resection [1].
This approach is most beneficial for patients who have a limited number of large, well-localized cysts. However, its effectiveness diminishes when cysts are located in hard-to-reach areas—such as the hepatic dome—where diaphragmatic adhesion to the fenestrated surface may lead to the formation of a new cyst-like wall. It is also less effective in cases with numerous, diffusely distributed small cysts, especially because preoperative CT imaging may overestimate cyst size due to the inability to visualize thin internal septations [1].
Although laparoscopic fenestration is generally less effective than the open approach, it offers the advantage of minimizing adhesions, thus facilitating future re-interventions, which are often necessary. It is usually preferred in patients with predominantly large cysts in a limited number and preferably situated in the anterior segments of the liver or the left lobe [9,10].
Extensive cyst fenestration has been shown to effectively alleviate symptoms in patients with autosomal dominant Polycystic Liver Disease (APLD). However, this aggressive approach to reducing liver volume carries risks, including bleeding and biliary complications. In type I APLD, the procedure can be safely performed laparoscopically. For patients with type II APLD, longer-term follow-up is necessary to assess the sustained benefit of the intervention. In contrast, type III APLD is associated with a higher risk of disease progression, with studies indicating significant worsening in approximately 40% of cases over time. These findings suggest that fenestration may not be the optimal long-term treatment strategy for all APLD subtypes [6]. Recent studies have demonstrated that partial hepatectomy combined with cyst fenestration (PHCF) offers a substantial and lasting reduction in liver volume and can significantly alleviate symptoms and enhance quality of life in patients with severe symptomatic polycystic liver disease (PLD) [11,12].
Conclusion
Polycystic liver disease, although uncommon, should be considered in the differential diagnosis of large abdominal masses, especially in patients without a history of liver disease. A thorough radiological evaluation is crucial for diagnosis and treatment planning. Surgical fenestration remains an effective option for symptom relief in selected patients with large, compressive cysts. Given the progressive nature of the disease and its potential complications, timely surgical intervention can significantly enhance patient outcomes and quality of life.
References
- Aussilhou B, Dokmak S, Dondero F, Joly D, Durand F, Soubrane O, et al. Treatment of polycystic liver disease. Update on the management. J Visc Surg, 2018; 155(6): 471–481.
- Mahboobipour AA, Ala M, Safdari Lord J, Yaghoobi A. Clinical manifestation, epidemiology, genetic basis, potential molecular targets, and current treatment of polycystic liver disease. Orphanet J Rare Dis, 2024; 19(1): 175.
- Borghol AH, Bou Antoun MT, Hanna C, Salih M, Rahbari-Oskoui FF, Chebib FT. Autosomal dominant polycystic kidney disease: an overview of recent genetic and clinical advances. Ren Fail, 2025; 47(1): 2492374.
- Norcia LF, Watanabe EM, Hamamoto Filho PT, Hasimoto CN, Pelafsky L, de Oliveira WK, et al. Polycystic Liver Disease: Pathophysiology, Diagnosis and Treatment. Hepatic Med Evid Res, 2022; 14: 135–161.
- Zhang ZY, Wang ZM, Huang Y. Polycystic liver disease: Classification, diagnosis, treatment process, and clinical management. World J Hepatol, 2020; 12(3): 72–83.
- Gigot JF, Jadoul P, Que F, Van Beers BE, Etienne J, Horsmans Y, et al. Adult polycystic liver disease: is fenestration the most adequate operation for long-term management? Ann Surg, 1997; 225(3): 286–294.
- Schnelldorfer T, Torres VE, Zakaria S, Rosen CB, Nagorney DM. Polycystic liver disease: a critical appraisal of hepatic resection, cyst fenestration, and liver transplantation. Ann Surg, 2009; 250(1): 112–118.
- Masyuk TV, Masyuk AI, LaRusso NF. Polycystic Liver Disease: Advances in Understanding and Treatment. Annu Rev Pathol, 2022; 17: 251–269.
- Kabbej M, Sauvanet A, Chauveau D, Farges O, Belghiti J. Laparoscopic fenestration in polycystic liver disease. Br J Surg, 1996; 83(12): 1697–1701.
- Pantè S, Di Dio V, Putorti A, Salvo A, Barbera N, Catalfamo G, et al. Laparoscopic cyst fenestration in the treatment of polycystic liver disease. Ann Ital Chir, 2014; 85(3): 298–303.
- Bernts LHP, Neijenhuis MK, Edwards ME, Sloan JA, Fischer J, Smoot RL, et al. Symptom relief and quality of life after combined partial hepatectomy and cyst fenestration in highly symptomatic polycystic liver disease. Surgery, 2020; 168(1): 25–32.
- Boillot O, Cayot B, Guillaud O, Crozet-chaussin J, Hervieu V, Valette PJ, et al. Partial major hepatectomy with cyst fenestration for polycystic liver disease: Indications, short and long-term outcomes. Clin Res Hepatol Gastroenterol, 2021; 45(3): 101670.

