Rosai-Dorfman Disease: A Rare Pediatric Case with Bilateral Temporal Lobe Lesions and Right-Sided Hemiparesis
Areeba Tariq1,*, Qurat-ul-Ain Tariq2, Saad-bin-Anis3, Muhammad Omar Ahmad4, Irfan Yousaf5 and Fauzan Alam Hashmi6
1Fellow Surgical NeuroOncology, Shaukat Khanum Memorial Cancer Hospital and Research Centre, Lahore, Pakistan
2Senior Instructor Surgical NeuroOncology, Shaukat Khanum Memorial Cancer Hospital and Research Centre, Pakistan
3Consultant Neurosurgeon, Shaukat Khanum Memorial Cancer Hospital and Research Centre, Pakistan
4Resident medical officer, Shaukat Khanum Memorial Cancer Hospital and Research Centre, Pakistan
5Consultant Neurosurgeon, Shaukat Khanum Memorial Cancer Hospital and Research Centre, Pakistan
6Consultant Neurosurgeon, Shaukat Khanum Memorial Cancer Hospital and Research Centre, Pakistan
Received Date: 06/05/2025; Published Date: 29/07/2025
*Corresponding author: Dr. Saad-bin-Anis, Consultant Neurosurgeon, Shaukat Khanum Memorial Cancer Hospital and Research Centre, Lahore, Pakistan
Abstract
Rosai-Dorfman Disease (RDD) is a rare, idiopathic histiocytic disorder, typically presenting with massive cervical lymphadenopathy. Central Nervous System (CNS) involvement is uncommon, occurring in less than 5% of cases, with isolated intracranial presentations being extremely rare, particularly in children.
We present the case of a 14-year-old girl with progressive right-sided hemiparesis. MRI revealed bilateral temporal lobe lesions, midline shift, and hydrocephalus—initially suggestive of low-grade gliomas. The patient underwent a neuronavigation-guided left temporal craniotomy for lesion resection. Histopathology showed S100-positive histiocytes, negative for GFAP, Olig2, and SSTR2, confirming RDD. PET-CT revealed no systemic disease, and management proceeded with a multidisciplinary team.
RDD should be considered in the differential diagnosis of atypical CNS masses in children. Early recognition, collaborative care, and long-term follow-up are crucial for favorable outcomes.
Keywords: Rosai-Dorfman Disease; Central Nervous System; Pediatric neurology; Intracranial mass; Temporal lobe lesion; Histiocytosis
Introduction
Rosai-Dorfman Disease (RDD), also called Sinus Histiocytosis and Massive Lymphadenopathy (SHML), is a rare, benign and idiopathic histiocytic proliferative disorder [1]. The most common clinical presentation is painless bilateral cervical lymphadenopathy [2,3]. The most common sites of extranodal involvement are the skin, paranasal sinuses, soft tissues, bone, and central nervous system [4,5]. Extranodal disease can occur in 43%, however, in less than 5% of cases, the disease has been reported in the central nervous system and in the majority with lymph node involvement, with reports of isolated intracranial involvement being exceptional [6,7]. Central Nervous System (CNS) involvement occurs with 75% occurring as intracranial and 25% as spinal lesions. Neurologic RDD has been reported in >300 cases [8] and usually occurs in older patients and without lymphadenopathy [9]. Symptoms include headaches, seizures, gait difficulty, motor or sensory abnormalities, and cranial nerve deficits, usually evolving over weeks or months [8].
The most common radiographic appearance of intracranial RDD is a solitary extraaxial, homogeneously enhancing dural mass mimicking a meningioma [10,11]; although RDD can cause diffuse pachymeningitis. Parenchymal lesions are frequently infratentorial (brainstem and pons) [12], whereas supratentorial, intraventricular, and multifocal lesions are rare [13,14].
Bilateral temporal lobe involvement presenting with neurological deficits, such as hemiparesis, is exceptionally rare. This case emphasizes the significance of recognizing and managing atypical CNS presentations of RDD, particularly in pediatric patients.
Case Presentation
A 14-year-old, previously well, girl presented with right sided hemiparesis which was insidious in onset and slowly progressed over the 4 months. There was no associated fever, headaches, vomiting, weight loss, visual disturbances, facial weakness, dysarthria, paresthesias and ataxia. Vitals were stable. On the Higher Mental Function examination, she was awake, alert, well oriented in time, place and a person with mild dysphasia and no memory deficit. CNS examination showed right sided hemiparesis with normal bulk, increased tone, reflexes 3+ and positive Babinski sign. Power was 4/5 in all the muscle groups. Cranial nerves and sensory system were intact. Speech was scanning. Pupils were bilaterally equal and reactive. All other systems were normal and there was no lymphadenopathy.
MRI outside hospital was done which was re-reported in our centre as Bilateral temporal lobe enhancing lesions. The left temporal lobe mass was larger in size and causing right-sided midline shift causing compression of the third ventricle with upstream hydrocephalus and transependymal seepage. These masses were reported as of neoplastic etiology, likely low-grade gliomas. Then MRI Brain with contrast was repeated which showed Bilateral intraventricular masses which are more or less stable in sizes causing hydrocephalus and compression effect at the level of the third ventricle towards the right side. Differentials included intraventricular tumors. No adjacent invasion is seen.
The patient was started on Dexamethasone 2mg three times a day with Famotidine alongwith Levitericitam 250mg two times daily. MRI spine with contrast was done which showed No drop metastasis. The case was discussed in the weekly board meeting where the initial assessment was that the lesion is likely a glioma/PNET.Bilateral surgical resection was decided to be done and if the lesion turned out to be vascular then planned for biopsy with shunt placement. Patient was admitted and patient was scheduled for neuronavigational guided Bilateral surgical resection, however patient underwent neuronavigational guided left craniotomy and excision of tumor only.
On the day of surgery, the patient was held in Supine Position, head rotated to the right side. The head was fixed in a three pin mayfields head clamp. Hair shaved and Left "?" shaped incision in front of left ear. Temporalis fascia and muscle cut. Burr holes were made using the burr and craniotomy performed using a craniotome. Dura was opened in a "C" shaped fashion. On raising the bone flap, Tense dura identified. the temporal lobe was bulging. A very hard, encapsulated, solid, calcified tumor in its entirety with no areas of softness identified.The core was avascular and the tumor was partly intraventricular, medially compressing the brainstem which was very gliotic. The brain parenchyma all around the tumor was also gliotic. Main arterial feeders were coming from the medial side from the left posterior cerebral artery and main venous drainage was into the left vein of rosenthal. Corticectomy done over the inferior temporal gyrus. Tumor was identified. Frozen Section was sent which came out to be lesional tissue, (inflammatory vs very low-grade glioma).
The specimen was taken for histopathology. Tumor was debulked. It was a very hard and gritty tumor. The tumor was removed in total.
After surgery the patient was shifted to the Intensive care unit for a day for monitoring followed by ward stay and later discharge. Post op patient developed right sided ophthalmoplegia (right eye: abducens and trochlear palsy, vision decreased to finger counting) (left eye: left abducens palsy, vision normal) right facial weakness grade 3. On motor examination of extremities, there was right hemiparesis 2/5 in arms, 2/5 in leg.
Post op MRI was done after 24 hours and it showed post-surgical changes of left temporal craniotomy with left temporal and subarachnoid hemorrhage. Pneumocephalus was noted along the left subarachnoid space and left frontal lobe, causing mild effacement of the ipsilateral gyri. Moreover, there was a redemonstration of enhancing right temporal lobe mass with enhancing lesion is noted in the left lateral ventricle suggestive of residual disease.
Patient was followed on after 1 month in clinic with complains of facial weakness but some improvement. Pupils were reactive, left sided cranial nerve palsy static (lower half), right sided weakness with clawing and wasting right hand, power ⅕ in right arm and leg.
Histopathology showed sections revealing brain parenchyma with a lesion composed of abundant histiocytes with round to oval nuclei and abundant pale cytoplasm. IMMUNO/HISTOCHEMICAL/IN SITU HYBRIDIZATION (ISH) STAIN(S) were run on sample showing: SSTR2: Negative, GMS: Negative for fungus, Olig2:Negative , GFAP: Negative, S100: Positive. Fungal cultures also came out to be negative. The final diagnosis was Rosai Dorfman disease.
The patient's case was discussed in National Multidisciplanry meeting and they advised PET CT and started prednisolone. The patient was started on prednisolone 40 mg/m2 /day for 4 weeks followed by weaning in next 2 weeks with proton pump inhibitor and MRI brain in 6 weeks.PET-CT showed Hypermetabolic enhancing intraventricular lesions likely involved by primary disease process. Post craniotomy extra-axial non-avid fluid density noted in the left frontoparietal region. A few mildly avid non-enlarged bilateral cervical level II nodes. No other evidence of hypermetabolic nodal or visceral disease involvement. Physiotherapy was continued and Peds oncology follow up clinic was scheduled.
Case was later rediscussed in National Pediatric tumor board at 3 months post-surgery and it was recommended to follow up patient 3 monthly as she is clinically well and Sirloimus or XRT can be offered in case of disease progression.
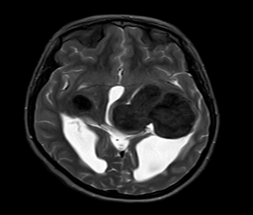
Figure 1: Lesion on T2 axial brain MRI.
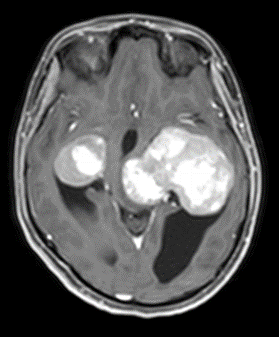
Figure 2: Lesion on T1 post contrats axial brain MRI.
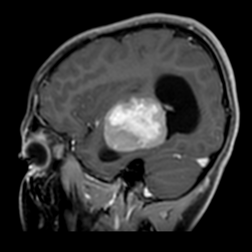
Figure 3: Lesion on T1 post contrast sagittal brain MRI.
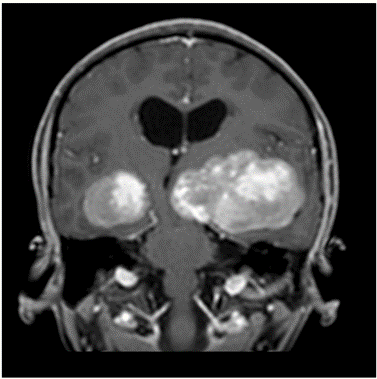
Figure 4: Lesion on T1 post contrast coronal brain MRI.
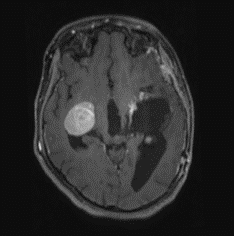
Figure 5: Post op MRI T1 post contrast axial image.
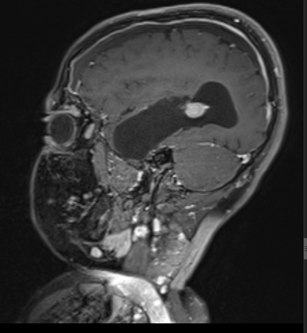
Figure 6: Post op MRI T1 post contrast sagittal image.
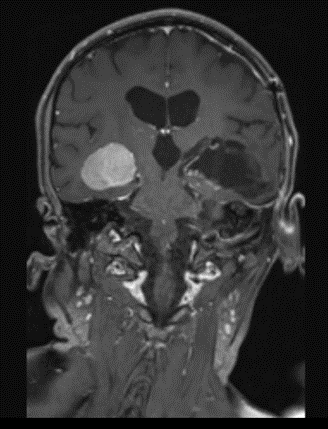
Figure 7: Post op MRI T1 post contrast coronal image.
Discussion
RDD is a well described entity, the etiology of which is still unknown. It often occurs in the setting of nonspecific immune dysfunction with many cases occurring after a viral illness [15]. Levine et al recommended that the human herpes virus 6 and to a lesser extent Epstein Barr virus may be involved in the aetiology [16].
The mean age of onset with nodal disease is 20.6 years with a male to female ratio of 1.4:1 [6,17]. Patients who develop intracranial involvement, however, become symptomatic at a mean age of 34.9 years, with a strong male preponderance. In intracranial RDD, the most involved structures are the suprasellar region, cerebral convexity, parasagittal region, cavernous sinus, and petroclival region [18,19]. Radiologically, the typical MRI findings include isointense to hypointense T1-weighted signals and hyperintense T2-weighted signals with homogeneous enhancement post-contrast. Bitemporal lesions often exhibit a dural-based, meningioma-like appearance [20]. Intracranial RDD is commonly confused with meningioma and requires tumor histopathology and immunohistochemistry for its definitive diagnosis.
Our case shows intraxial, multifocal and supratentorial,intraventricular involvement of CNS in a young fourteen year old child, which is rare.
Bilateral temporal lobe involvement with mass effect, brainstem compression and obstruction of the ventricular system at the level of the third ventricle causing hydrocephalus is exceptional. Differential diagnosis includes several diseases (such as bilaterla glioma, PNET) in this case. A precise diagnosis could not be reached on clinical grounds alone.
A definitive diagnosis of RDD relies on biopsy. Histologically RDD histiocytes are positive for CD163, CD68, and S100 antigens, which are found on macrophages and dendritic cells. RDD is CD1a and langerin negative, which distinguishes it from Langerhans cell histiocytosis (LCH). Like LCH, RDD is a clonal proliferative disease, characterized by the overrepresentation of blood cells derived from a single clone. Characteristically RDD presents with emperipolesis, where intact lymphocytes or erythrocytes are found within histiocyte cytoplasm [6].
In our case tissue histology of resected specimen revealed brain parenchyma with a lesion composed of abundant histiocytes with round to oval nuclei with abundant pale cytoplasm and immunohistochemistry positive for S100 and negative for SSTR2, GMS, Olig2 and GFAP stains.
Management strategies for RDD are guided by the extent of disease and symptoms. In isolated CNS cases, surgical resection remains the mainstay of treatment, particularly for symptomatic mass lesions [21]. Gross total resection (GTR) is associated with favorable outcomes, though Subtotal Resection (STR) may suffice if critical structures are involved(6). Adjuvant therapies, including corticosteroids, radiation, and chemotherapy, have been employed in refractory cases or those with systemic involvement [22]. A consensus report suggests that targeted therapies, such as MEK inhibitors, show promise in patients with activating mutations in the MAPK/ERK pathway [22]. Pediatric cases with bitemporal lesions treated surgically demonstrated significant symptomatic relief and no recurrence during follow-up [16]. Long-term follow-up with regular imaging is recommended to monitor for recurrence. Cases of bitemporal lesions reviewed in the literature exhibited good neurological outcomes post-treatment, though some patients required prolonged corticosteroid therapy for residual inflammation [17].
Conclusion
Rosai-Dorfman Disease (RDD) remains a rare and diagnostically challenging histiocytic disorder, particularly when it presents as isolated intracranial involvement in pediatric patients. This case underscores the clinical complexity of RDD, especially with bilateral temporal lobe and intraventricular lesions causing mass effect, brainstem compression, and hydrocephalus—features that closely mimic high-grade glioma or PNETs on imaging. Definitive diagnosis requires histopathologic confirmation, with immunohistochemistry playing a key role in distinguishing RDD from other neoplastic and histiocytic disorders.
In our case, early surgical intervention facilitated both tissue diagnosis and symptomatic relief, though postoperative neurologic deficits highlighted the delicate balance between tumor resection and preservation of function. Subsequent corticosteroid therapy and follow-up imaging were essential components of management. Given the rarity and potential for recurrence, particularly in CNS-involved RDD, a multidisciplinary approach and long-term surveillance remain crucial. This case adds to the limited literature on pediatric intracranial RDD and emphasizes the importance of considering RDD in the differential diagnosis of atypical CNS masses in children.
References
- Rosai J, Dorfman RF. Sinus histiocytosis with massive lymphadenopathy: A pseudolymphomatous benign disorder. Analysis of 34 cases. Cancer. 1972;30(5):1174–88. doi:10.1002/1097-0142(197211)30:5<1174::AID-CNCR2820300507>3.0.CO;2-S.
- Foucar E, Rosai J, Dorfman RF. Sinus histiocytosis with massive lymphadenopathy (Rosai–Dorfman disease): review of the entity. Semin Diagn Pathol. 1990;7:19–73.
- Rosai J. Rosai and Ackerman’s Surgical Pathology. 10th ed. New York: Mosby; 2011.
- Simos M, Dimitrius P, Philip T. A new clinical entity mimicking meningioma diagnosed pathologically as Rosai-Dorfman disease. Skull Base Surg. 1998;8(2):87–92.
- Andriko JA, Morrison A, Colegial CH, Davis BJ, Jones RV. Rosai-Dorfman disease isolated to the central nervous system: a report of 11 cases. Mod Pathol. 2001;14(3):172–8.
- Konishi E, Ibayashi N, Yamamoto S, Scheithauer BW. Isolated intracranial Rosai-Dorfman disease (sinus histiocytosis with massive lymphadenopathy). AJNR Am J Neuroradiol. 2003;24(3):515–8.
- Gupta K, Bagdi N, Sunitha P, Ghosal N. Isolated intracranial Rosai-Dorfman disease mimicking meningioma in a child: a case report and review of the literature. Br J Radiol. 2011;84(1003):e138–41.
- Sandoval-Sus JD, Sandoval-Leon AC, Chapman JR, et al. Rosai-Dorfman disease of the central nervous system: report of 6 cases and review of the literature. Medicine (Baltimore). 2014;93(3):165–75.
- Taufiq M, Khair A, Begum F, Akhter S, Farooq MS, Kamal M. Isolated intracranial Rosai-Dorfman disease. Case Rep Neurol Med. 2016;2016:1972594.
- Nalini A, Jitender S, Anantaram G, Santosh V. Rosai-Dorfman disease: case with extensive dural involvement and cerebrospinal fluid pleocytosis. J Neurol Sci. 2012;314(1–2):152–4.
- Varan A, Şen H, Akalan N, Oğuz KK, Sağlam A, Akyüz C. Pontine Rosai-Dorfman disease in a child. Childs Nerv Syst. 2015;31(6):971–5.
- Morandi X, Godey B, Riffaud L, Heresbach N, Brassier G. Isolated Rosai-Dorfman disease of the fourth ventricle. Case illustration. J Neurosurg. 2000;92(5):890.
- Antuña Ramos A, Alvarez Vega MA, Alles JV, Antuña Garcia MJ, Meilán Martínez A. Multiple involvement of the central nervous system in Rosai-Dorfman disease. Pediatr Neurol. 2012.
- Goyal G, Ravindran A, Young JR, Shah MV, Bennani NN, Patnaik MM, et al. Rosai-Dorfman disease of bone and soft tissue: a rare entity with distinct clinical and pathologic features. Arch Pathol Lab Med. 2022;146(1):40–6. doi:10.5858/arpa.2020-0489-OA.
- Levine PH, Jahan N, Murari P, Manak M. Detection of human herpesvirus 6 in tissues involved by sinus histiocytosis with massive lymphadenopathy (Rosai-Dorfman disease). J Infect Dis. 1992;166(2):291–5. doi:10.1093/infdis/166.2.291.
- Abla O, Jacobsen E, Picarsic J, et al. Consensus recommendations for the diagnosis and clinical management of Rosai-Dorfman-Destombes disease. Blood. 2018;131(26):2877–90. doi:10.1182/blood-2018-03-839753.
- Zumaeta J, Palacios F, Anicama W, Burgos C. Isolated intracranial Rosai-Dorfman disease: case report. Peru J Neurosurg. 2020;2(1):15–21.
- Symss NP, Cugati G, Vasudevan MC, Ramamurthi R, Pande A. Intracranial Rosai-Dorfman disease: report of three cases and literature review. Asian J Neurosurg. 2010;5(2):19–30.
- Forest F, N’guyen AT, Fesselet J, et al. Meningeal Rosai-Dorfman disease mimicking meningioma. Ann Hematol. 2014;93(6):937–40.
- Cooper SL, Jenrette JM. Rosai-Dorfman disease: management of CNS and systemic involvement. Clin Adv Hematol Oncol. 2012;10(3):199–202.
- Raslan O, Ketonen LM, Fuller GN, Schellingerhout D. Intracranial Rosai-Dorfman disease with relapsing spinal lesions. J Clin Oncol. 2008;26:3087–9.
- McAlister WH, Herman T, Dehner LP. Sinus histiocytosis with massive lymphadenopathy (Rosai-Dorfman disease). Pediatr Radiol. 1990;20:425–32.

