Effective Synergy of Fractional CO2 and 675 nm Laser for Moderate to Severe Acne Vulgaris
Domenico Piccolo1, Irene Fusco2,* and Tiziano Zingoni2
1Novea Skin Center - Dermo Aesthetic Laser Centers, Avezzano, Pescara and L'Aquila, Italy
2El.En. Group, 50041 Calenzano, Italy
Received Date: 19/06/2025; Published Date: 25/07/2025
*Corresponding author: Irene Fusco, El.En. Group, 50041 Calenzano, Italy
ORCID: 0000-0001-7264-8808
Abstract
Background: Acne vulgaris is a common inflammatory skin condition that can lead to long-term physical and psychological effects. Traditional therapies may fall short in addressing both active lesions and residual scarring.
Objective: This study evaluates the clinical efficacy of a combined treatment using a mild ablative CO₂ fractional laser and a non-ablative 675 nm laser system in patients with moderate to severe acne vulgaris.
Methods: Twelve patients (mean age 19.8 ± 3.4 years; 83% women) with Fitzpatrick skin types II–III and moderate to severe acne were treated in a single session with both CO₂ fractional laser (TetraPRO) and 675 nm non-ablative laser (RedTouchPRO). Acne severity was assessed using Acne Severity Grading Criteria before treatment and at a 3-month follow-up.
Results: The combined laser treatment resulted in a visible and measurable reduction in acne severity, fibrosis, and inflammation. Clinical imaging confirmed an improvement in skin texture and lesion appearance, with no reported adverse effects.
Conclusion: The synergistic use of ablative CO₂ and non-ablative 675 nm lasers appears to be an effective and well-tolerated treatment option for moderate to severe acne vulgaris, offering promising results in both lesion reduction and overall skin texture improvement.
Clinical Images
A total of 12 subjects (83% women and 17% men, mean age 19.8±3.4 years), with Fitzpatrick II-III skin types (66% type II, 34% type III) affected by moderate to severe acne vulgaris (Papulopustular 83%, Nodulocystic 17%), were synergistically treated with a mild CO2 ablative fractional Laser (TetraPRO, Deka M.E.L.A, Italy) combined with a non-ablative 675 nm laser system (RedTouchPRO, Deka M.E.L.A, Italy) in the same session.
The efficacy of the treatment was assessed using the Acne Severity Grading Criteria, assessed before treatment and at 3 months follow-up after the second session. The graph in Figure 1 shows a decrease in severities, fibrosis, inflammation, indicating an improvement of skin texture. Some clinical cases are shown in Figures 2 and 3.
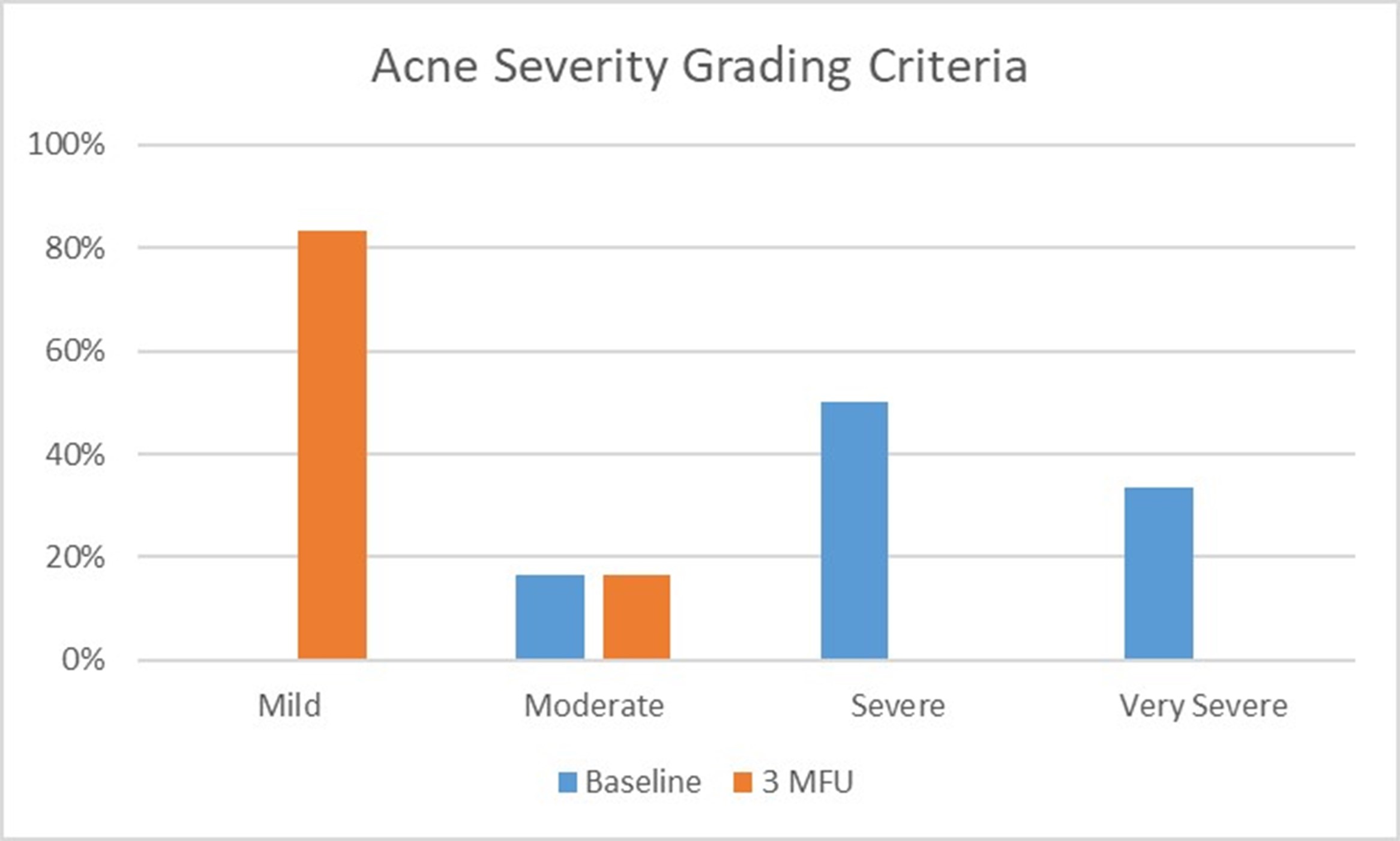
Figure 1: Acne Severity Grading Criteria, before and at 3 Months follow-up.
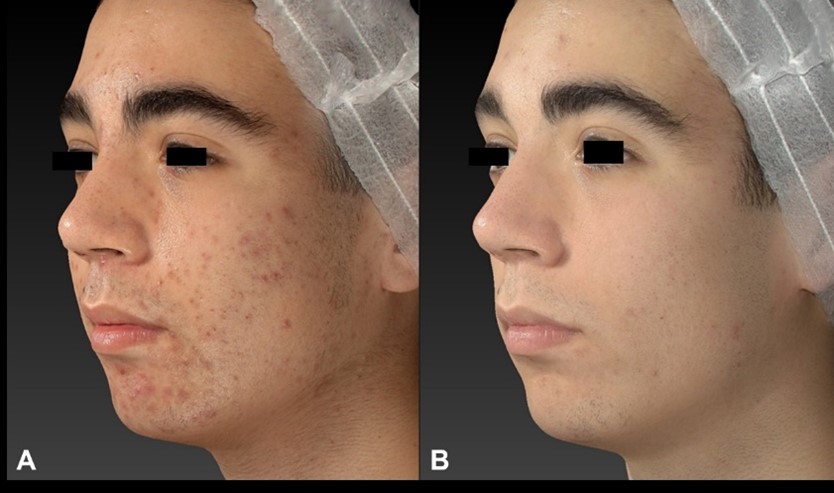
Figure 2: Quantificare Images, before (A) and at 3 Months follow-up (B). Fractional CO2 laser (10W Power, 1000 µs Pulse duration, SP pulse, Stack 1, 500 µm spacing) followed by the 675 nm laser (standard mode, 10W Power, 150 ms Pulse duration, Stack 1, 1500 µm spacing).
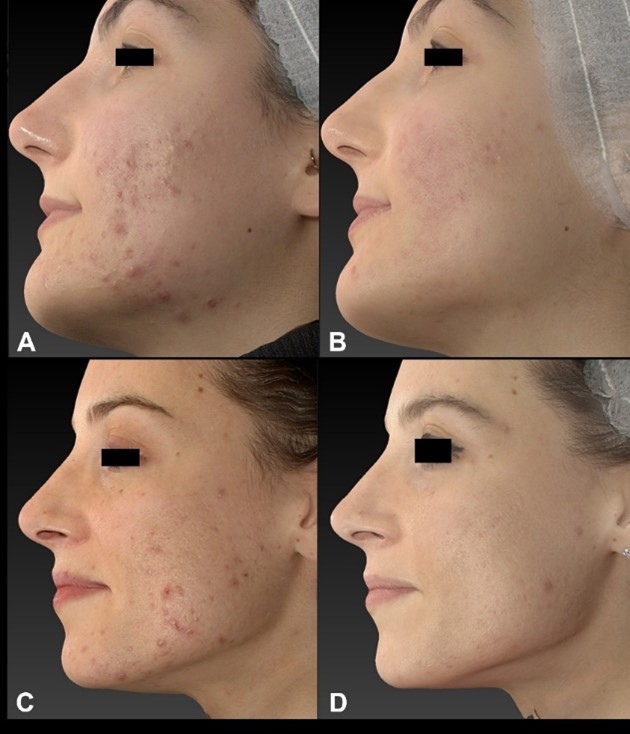
Figure 3: Upper panels: Quantificare Images of a female patient, before (A) and at 3 Months follow-up (B). Fractional CO2 laser (10W Power, 1000 µs Pulse duration, SP pulse, Stack 1, 500 µm spacing) followed by the 675 nm laser (standard mode,10W Power, 150 ms Pulse duration, Stack 1, 1500 µm spacing). Low panels: Quantificare Images of another female patient, before (C) and at 3 Months follow-up (D). Fractional CO2 laser (10W Power, 1000 µs Pulse duration, SP pulse, Stack 1, 500 µm spacing) followed by the 675 nm laser (standard mode,10W Power, 150 ms Pulse duration, Stack 1, 1500 µm spacing).
Conclusion
The combined application of fractional CO₂ and non-ablative 675 nm laser systems demonstrated a synergistic effect in reducing the severity of moderate to severe acne vulgaris. Improvements were observed in inflammation, fibrosis, and overall skin texture at 3 months post-treatment, with high patient satisfaction and no adverse effects reported. These findings suggest that the integrated use of ablative and non-ablative laser modalities may offer a safe, effective, and innovative therapeutic approach in the clinical management of acne, particularly in cases resistant to conventional therapies. Further studies with larger cohorts and longer follow-ups are recommended to validate these preliminary outcomes.
Statements
Funding: This research received no external funding.
Institutional Review Board Statement: the article is in accordance with the Declaration of Helsinki on Ethical Principles for Medical Research involving human subjects. Ethical approval is not necessary as the study devices are already CE marked since 2023-2024.
Informed Consent Statement: Written informed consent has been obtained from the patients to publish this paper.
Data Availability Statement: Data that support the study findings are available on request from the corresponding author (IF).
Conflicts of Interest: Authors TZ and IF were employed by El.En. Group. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.

