The Impact of Carbohydrate Derived Energy Sources and Metabolism on Cognition
Xin Qi* and Richard Tester
Glycologic Limited, Lambs Farm Business Park, UK
Received Date: 13/06/2025; Published Date: 16/07/2025
*Corresponding author: Dr. Xin Qi, Glycologic Limited, Lambs Farm Business Park, Basingstoke Road, Reading, RG7 1PQ, UK
Abstract
The brain has limited energy supply options, where glucose is the primary energy source of choice although it may also use lactate and ketone bodies for this purpose. Glucose is consumed in the diet in many forms which can be absorbed/digested to provide energy, including the free monosaccharide, within the common disaccharides lactose, maltose and sucrose, in dextrins and starch. The brain needs a managed and controlled energy supply. Key to this control is regulation of the diet so that calories are not consumed in excess of need and that foods providing a steady nutrient release within the gut are selected. People tend not to consider how glucose provision to the body affects mental performance - they tend to think in terms of physical impact alone. This short review teases out the various health-related issues that are associated with consuming glucose in different forms within the diet and in particular, the impact on cognition.
Keywords: Blood glucose; Brain; Cognition; Energy; Health
Introduction
Overview
The physiological basis of blood glucose concentration ‘glucostat’ is the matter of great debate - in particular the role of the pancreas versus the brain within the control mechanism [1-2]. The brain uses only a very select group of nutrients as a source of energy where glucose is the most important. The utilisation of energy by the brain has been discussed by many authors where there are a few key elements in this respect as highlighted below [3-15]:
- The adult brain represents about 2% of the body weight but utilises 20% of the oxygen inhaled and 20-25% of the glucose consumed;
- Glucose is the main energy source of/for the brain where adenosine triphosphate (ATP) is generated from adenosine diphosphate (ADP) plus inorganic phosphate (Pi) by glycolysis and oxidative phosphorylation;
- Neuron metabolism is mainly oxidative, for astrocytes (a sub-group of glial cells) mainly glycolytic;
- Ketone ‘bodies’ (β-hydroxybutyrate, acetoacetate, and acetone) derived from the oxidation of fat in the liver, provide an additional fuel source for the brain through oxidative phosphorylation - up to about 70% of total energy if necessary;
- About 10-12% of glucose is converted by glycolysis to lactic acid even though oxygen is not limiting - referred to often as ‘aerobic glycolysis’ or the ‘Warburg effect’;
- Lactic acid may be used readily as an energy source in the brain;
- Neurones consume 75-80% of energy produced in the brain to (a) transmit information through action potentials along the nerves and across synapses and (b) to reconfigure the neural pathways - especially synaptic linkages;
- Glycogen deposits exist in astrocytes in the brain and provide energy reserve depots and;
- The brain itself contributes to the regulation of glucose homeostasis in the body. There are regions in the brain’s hypothalamus and specialised neurones which sense glucose, fatty acids, insulin and leptin. These metabolic signals converge in the hypothalamus and brain stem and support the overall energy usage of the body and brain.
Glucose homeostasis is critical for supporting the energy needs of normal mental functionality/cognition (the mental ability to gain knowledge and understanding through thought processes, experiences and through input from the various senses). Without this control, the ability to function correctly is compromised.
Dietary carbohydrates
The average human diet is very complex where carbohydrates are key components. The carbohydrates may be digestible and enter the blood steam in the small intestine as monosaccharides or be indigestible and more-or-less fermented in the colon of man. Many different monosaccharides structures are consumed in different forms where fructose, galactose and glucose are most common [16-17]. These sugars a provided frequently in the form of the common disaccharides lactose (galactose + glucose), maltose (glucose + glucose) and sucrose (fructose + glucose) as discussed elsewhere [16-17].
Glucose exists in many different molecular forms in foods and drinks that are more-or-less accessible to the human digestive processes/enzymes. These forms of glucose include sugars (see above), oligosaccharides and starch derived polysaccharides (amylose and amylopectin) from a range of different plant sources and tissues. Rarely is glucose consumed as the free monosaccharide although honey (rich in inverted sucrose, that is fructose and glucose) is the exception.
The α-(1-4) and α-(1-6) bonded dietary glucose sources, namely (but not exclusively) amylose and amylopectin within starch granules and starch derivatives, may be digested to glucose by humans. Digestion is slow in the case of starches in their native granular form [18-21], although the granules are digested more easily by the concerted action of α-amylase, maltase-glucoamylase (‘maltase’ commonly and ‘maltase-𝛼-glucosidase’) and sucrase-isomaltase (‘debranching enzyme’ or ‘isomaltase’) when damaged/cooked or hydrolysed to dextrins [17,19-23].
Once within the blood stream, the body works hard using a number of interlinked physiological mechanisms (including the opposing actions of glucagon and insulin) to manage continuously the blood glucose concentration (glucose homeostasis) - for what is a critical energy supply for both the brain and body. The glucose entering the blood stream from the small intestine providing a source to be managed to optimise health in concert with glucose stored in the storage depots (discussed below). During physical exertion the demand for glucose within the body will increase where in common with rest, provision must be made for both the body’s and brain’s needs. States of glucose imbalance (discussed below) challenge the body’s capacity to function as the deficiency and excess are not compatible with health. Hence, there are a complex array of mechanisms to maintain blood glucose concentrations within a desirable operational range.
Blood Glucose and Brain Health
The impact of blood glucose concentration on cognitive health
Localisation: There are suggestions in the literature that not only does glucose enhance cognition when delivered to the brain via different routes but, that mental performance in specific areas of the brain can be enhanced by increased local blood glucose supply [24-26]. Thus, glucose is utilised according to local need. Fructose, although not a key brain fuel, exerts impact on cognition although differently from glucose [27].
Hyper- and hypoglycaemia: Overall, dysregulated glucose supply to the brain causes trauma. In terms of physiological health impacts on brain function, both low (hypo-) and high (hyper-glycaemia) blood glucose concentrations are detrimental. Cognitive decline is associated with diabetes - and hence with high blood glucose concentrations especially [28]. Hyperglycaemia associated with both type I or type II diabetes causes cognitive dysfunction, as discussed by Sommerfield et al. [29], Cox et al. [30] and Weinstein et al. [31].
There may well be a direct relationship between hyperglycaemia and dementia [32-34]. However, and perhaps perceived as an opposite effect for the brain (tissue) itself, reduced glucose uptake in the brain is an early biomarker of Alzheimer’s disease [35]. A maternal history of Alzheimer disease predisposes normal individuals to progressive glucose metabolism issues in Alzheimer disease vulnerable brain regions, which may reflect a greater risk for developing the disease [36].
In general, it is difficult to elicit from the literature if (a) a congenital/induced detrimental change in the brain affecting glucose uptake by cells and/or metabolism (glycolysis and oxidative phosphorylation) is responsible (at least in part) for Alzheimer disease development [37-41] or (b) if high circulating glucose concentrations (as in for example diabetes) is toxic to the brain and is (at least in part) responsible for the disease development [42-43]. However, according to Johnson et al. [44] who worked on Alzheimer brain tissue and cerebrospinal fluid, ‘A protein network module [domains that are found repeatedly in a range of proteins] linked to sugar metabolism emerged as one of the modules most often associated with Alzheimer’s disease pathology and cognitive impairment.’
High ‘normal’ blood glucose concentrations are like hyperglycaemic events equally, a risk factor for cognitive decline [45-47]. One could argue, therefore, that a lot more must be done to regulate energy consumption leading to regularly elevated blood glucose concentrations in the western diet. Liu et al. [48] however reported that in non-diabetic older adults lower concentrations of the anti-aging protein with neural protective effects called ‘Klotho’ are associated with poorer cognitive performance.
In type I diabetes, in both young and old people, hypoglycaemia is associated with a deleterious impact on cognition [49] too. The impact of hypoglycaemia on brain function and cognition has been considered by some professionals, however, to be controversial [50].
Hawks et al. [51] reported in a type I diabetic study review that by minimising glucose concentration fluctuations, the brain’s processing speed is optimised. In terms of cognitive vulnerability to this blood glucose concentration impact, they reported that the main relevant factors were age, time in hypoglycaemia, life time severe hypoglycaemic events, microvascular complications, glucose variability, fatigue and neck circumference.
In terms of cognition in healthy volunteers versus diabetes (type I), hypoglycaemia (induced via hyperinsulinaemic glucose clamps) causes cognition to be impaired in both groups [26,52]. This implies that the impact is a function of the low blood glucose directly - independently of any underlying health condition(s). In an elderly age group with reasonably good blood glucose control, it is apparent that a glucose source in the diet does improve mental performance [53]. Younger healthy people too, find cognition difficult when they are made hypoglycaemic [54]. Hence, the hypoglycaemic state impacts upon all age groups regardless of underlying health conditions to diminish cognitive functions.
Brain injury linked to hyper- and hypoglycaemia: Brain injury linked to acute and chronic hyperglycaemia is associated with impaired attention and memory capability - reflecting an early manifestation of impaired glucose metabolism [31, 55]. Hyperglycaemia may exacerbate dementia-related neuropathology - although the direct impact on cognitive decline or dementia risk is difficult to quantify [56]. Hypoglycaemia causes/results in functional impairment too although this state may be corrected by increasing the blood glucose concentration [57].
Glucose transport into the brain: Overall, with respect to brain glucose metabolism, the concentration dependent glucose transporter GLUT 3 transports glucose into brain cells [58]. Berg et al. [59] discussed how the low KM (1.6 mM) for glucose of GLUT3 means it is usually saturated and thus provides the brain with a continuous source of glucose. The glucose concentration in the brain is roughly 1 mM when plasma is 4.7 mM. They also indicated that when the pervading glucose concentration approaches the KM of hexokinase (50µM, the enzyme which in effects retains glucose in the cell) glycolysis slows down, but approaches a dangerous point when plasma glucose concentration drops below 2.2mM (close to the KM of GLUT3).
Glucose ‘dosage rate’ from the gut: The interesting question with respect to blood glucose concentration is, ‘is it better to receive a slow entrance of glucose into the blood stream over a long time period for cognition optimisation or can the brain adapt to highs and lows of glucose exposure?’. This has three elements essentially: (i) the composition of the diet, eating habits and overall exposure to digestible carbohydrates; (ii) physiological status of the body/brain impacting on blood glucose supply to the brain and; (iii) cognition in health and disease. This is against the backdrop of the relative importance of the brain and body in managing glucose homeostasis [60]. In general terms, calorie control/restriction is beneficial with respect to cognition [61]. However, it depends on how calorie availability is managed actually. Dias et al. [61] indicated that calorie restriction can improve cognition, when managed moderately early in life but if applied more intensely later in life, it may be deleterious. Clearly a balance of calories entering the body versus those utilised by the body is relevant to both physical and cognitive health. Apparently, excessive sugar intake is associated with dementia [62].
The dietary sources of glucose impact on cognitive health
As discussed above, glucose sources are common in nature both as sugars and within starch molecules (Table 1) and are important components of the diet [16-17,63-66]. Providing sweetness, bulk, structure, texture and calories. Dextrins (unlike indigestible dextrans) are generated by starch hydrolysis and are usually amorphous, readily dispersed/solubilised and unmodified chemically. Consequently, dextrins are hydrolysed in the digestive tract of man very rapidly to glucose which is then absorbed [63,67]. Amorphous starch - especially cooked or gelatinised starch - is also hydrolysed rapidly to glucose and absorbed [64-66]. Hence, the body is very efficient at liberating glucose from digestible sources.
Table 1: Glucose sources in the diet.

Glucose, glucose-containing digestible sugars and amorphous starch cause a rapid peak of blood glucose concentration (in parallel with increasing insulin concentration) around thirty to forty-five minutes post ingestion. This is followed by a slow decline to baseline and then usually undershoots the baseline as blood glucose concentration is managed within the body - utilising what it needs for energy during the process, storing some as glycogen and alternatively as fat. There is a general recognition that a slow flat post prandial blood glucose profile is desirable for health - which can be achieved by consuming slowly digestible starch [64-66]. This reduces the ‘pressure’ on the physiological responses around the body to manage peaks and troughs associated with the consumption. Overall energy consumption - including sugars and starches - must also be controlled to avoid physical diseases caused by excess (see below). These include obesity, diabetes, cardiovascular disease, metabolic syndrome etc. Usually, only physical manifestations of calorie excess, including glucose containing food sources are considered in health-related dialogue. However, there is an equally important cognitive related element too.
As discussed above, amorphous starch in the form of high molecular weight dextrins is hydrolysed and absorbed as glucose from the small intestine of man at about the same rate as glucose monosaccharide [63,67]. In rats at least, excess maltodextrin and sucrose consumption provide comparable harmful metabolic and cognitive impact [68]. Diets comprising glucose, maltodextrin and starch, high protein and high fat diets were fed to healthy male students by Fischer et al. [69]. They stated that ‘good and stable cognitive performance is related to a balanced glucose metabolism and metabolic activation state’ although it is not clear what impact the various carbohydrates had separately in their work. The starting point for glucose regulation from the gut then lies in if starch is amorphous or not [65]. This raises a question. Is it possible using slowly digested starch, providing slow energy release during digestion [66], that cognition is optimised over time? This where glucose is consumed with less peaks and troughs than yo-yo consumption of drinks and foods providing rapidly digestible glucose.
According to some authors [70], the impact of slowly digestible starch on cognition is uncertain. This applies to the so called ‘functional foods’ too [71] which were developed initially for physical rather than cognitive wellbeing. However, there is a growing view that controlled steady glucose presentation and usage by the body, is beneficial for both physical and mental health [72-73]. Thus, (i) the amount of glucose consumed and (ii) in what form glucose is consumed, impacts on all areas of wellbeing- physical and cognitive.
According to Nilsson et al. [74] and Sünram-Lea and Owen [75], blood glucose ‘spikes’ can provide short term cognitive benefits although flatter sustained stable blood glucose concentrations, avoiding peaks and troughs, provide better cognitive function and reduced risk of longer-term impairment. Hence, the control of blood glucose is not just important for physical health, but mental welfare too. Highs and lows of blood glucose concentration do not promote optimal cognitive performance and overall brain wellbeing. Mattson [76] makes an interesting overall view about why excess calories dull cognition. The author suggests that ‘…the reason that overconsumption of energy-rich foods negatively impacts cognition is that signalling pathways that evolved to respond adaptively to food scarcity are relatively disengaged in the setting of continuous food availability’. Obesity impairs cognition and increases the risk for some psychiatric disorders and dementias [76]. It is an interesting concept that has the evolutionary basis in the food gatherer lifestyle that humans followed during development/evolution.
The Role of Glycogen in Brain Health
In humans, glycogen is the main storage form of glucose - primarily in the liver but in muscles, the kidneys and the brain too [77-81]. These glycogen depots (Figure 1) provide an energy storage reserve where the muscles and liver are most important for the body in terms of amount stored. However, the brain depots may well prove to be more important in time than realised currently. The glycogen depots receive glucose from the blood stream post-prandial under the control of insulin in the most part, to provide an energy reserve and maintain a normal blood glucose concentration.
The glycogen within tissues occurs within granules or particles - which are smaller than plant starch granules (about one log scale) although there are many analogies. The heterogeneity of glycogen structures and their more readily digestible nature lends them more to be called particles than granules as for starch. The relative inner diameter of brain, skeletal muscle and liver glycogen particle sizes are 10-30, 10-40 and 110-290nm respectively [78]. In the body, glycogen particles comprise protein-glycogen forms within three types of structures - α- (mainly liver, comprising β-particles, relatively slow energy release), β- (individual particles, relatively rapid energy release, as found in the brain astrocytes, comprising γ-particles) and γ-particles. Liver α-particles contain about twenty-thirty β-particle subunits [80].
The structure of glycogen molecules is similar to amylopectin within starch. It contains about 92-93% α-(1-4) and 7-8% α-(1-6) glucose bonds; the latter creating the branches [77,80]. The exterior chain branches of α-(1-4) linked glucose residues, are, on average, thirteen residues long with three-four glucose residues between the branches [80]. A fully formed glycogen molecule within a β-particle (which is apparently just one molecule per particle) contains about 55,000 glucose residues [77,80] which equates to a molecular weight of about 8.91 million Daltons. Similar molecular weights of 1x106-1x107 Daltons have been reported by Brewer and Gentry [80] for these molecules. So called pro-glycogen and macro-glycogen are probably intermediate forms of β-granule synthesis [80].
Abnormal glycogen synthesis and cognitive health
Polyglucosan molecules, unlike glycogen, are water-insoluble, phosphorylated, contain long glucose chains and range from 1-2 to 35 or 40 µm [82-84]. The intracellular accumulation of polyglucosan deposits known as Lafora bodies in the brain and other tissues is associated with Lafora disease which is a fatal childhood epilepsy.
In humans, it is the loss-of-function mutations in laforin and malin proteins which suppress glycogen synthesis, that induces large numbers of these insoluble polyglucosan molecules to be deposited in neuronal cells [85]. Excessive glycogen accumulation in this context, with the associated dysregulated metabolism, may be responsible for epilepsy and neurodegeneration [82-84]. Therapies that reduce glycogen synthesis or target Lafora bodies are being developed as an approach to treat the condition [84]. There is a connection between affliction with epilepsy and cognitive impairment [86].
Properly regulated glycogen synthesis in the brain can protect against seizures [87]. The lack of glycogen increasing the susceptibility to epileptic seizures [87]. Neuronal accumulation of excess glycogen, however, is associated with/contributes to aging processes and may be a key factor regulating progressive neurological decline in humans [85]. Clearly the control of glucose management and utilisation in the brain is critical for good health.
Other forms of polyglucosan molecules have been found commonly in the aging brain (especially but not necessarily where Alzheimer’s disease is present) and include Corpora amylacea (corpus = body, amylaceous = starch like), Bielschowsky bodies and amylopectin bodies [88-93]. The Corpora amylacea are hyaline - glycoprotein-based structures. Their cause of occurrence and brain related impact is not at all certain.
It is interesting that many people suffering with epilepsy benefit from the high fat low carbohydrate/protein diet - the so-called keto diet - where ketone bodies are utilised by the brain for energy rather than glucose [94]. Brain reactive astrocytosis, synonymous with epileptic tissue, is associated with loss of glutamine synthetase (catalyses the condensation of glutamate with ammonia to form glutamine) activity, compromised glutamate-glutamine cycle and an associated increase in extracellular and astrocytic glutamate concentration [95]. The authors indicate that chronically elevated intracellular glutamate concentrations in astrocytes leads to alterations in the metabolism of glycogen and the synthesis of polyglucosans. Inaccessibility of glycogen-metabolising enzymes to these (unlike glycogen proper) insoluble molecules, affects the glycogenolysis driven reuptake of extracellular potassium ions by astrocytes. This then leads to an increased extracellular potassium ion concentration and associated membrane depolarisation. Further, DiNuzzo et al. [95] indicate that the loss of structural homogeneity of glycogen particles is associated with the disruption of brain potassium ion homeostasis and increased consequently susceptibility to seizures. Thus, the critical proper functionality of brain glycogen deposits prevents uncontrolled neurological activity via this mechanism.
Glycogen buffer and ascocytes in the brain
Gibson [96] discussed the apparent dichotomy of the role of glucose concentration in the blood on cognition, where neither high nor low concentrations are desirable, whilst exploring the glucose ‘buffering’ capability of ascocytes and the glycogen deposited within. The author reported that overall brain activity is relatively unaffected by blood glucose concentrations other than with respect to autonomic activity controlling glucose homeostasis. The astrocytes regulate glucose concentration in the extracellular space of the brain including the intracellular deposition and mobilisation of glycogen. The astrocyte- based glycogen is described sometimes as an emergency energy reserve [97]. This glycogen is metabolised via glycogenolysis/glycolysis to lactic acid which is a neuronal energy source that is transported by mono-carboxylate transporters [98]. Glycogen derived lactate is a critical energy source for brain energy demands - both for neuronal function and for survival needs [98-100]. Brain located glycogen supports learning and memory as it provides lactate to the neurons during the conditioning phase of in vitro long-term potentiation, an experimental equivalent of learning [100]. Within the hippocampus, glycogen plays a critical role supplying neurones with lactate during memory formation. This role of glycogen indicates that it provides a critical role in supporting brain functions [101-102].
Hypoglycaemia protection imparted by brain localised glycogen
When the brain is exposed to hypoglycaemia, a number of metabolic/physiological responses are activated via autonomic processes as discussed by others [103-109]:
- Insulin secretion is inhibited;
- Glucagon, adrenaline, noradrenaline, cortisol (ACTH) and growth hormone are secreted;
- Brain located glycogen provides energy store during hypoglycaemia;
- Multiple metabolic pathways are modified to protect the brain;
- After hypoglycaemia, the brain glycogen depots are increased - often described as ‘rebound’;
- Exercise can also condition the brain (like the body) to store greater glycogen deposits.
Hence, a great deal of responses is activated, to protect the physiological/cognitive processes involving many different mechanisms. The cognitive health of the body as critical and the physiological health.
The consequences of hypoglycaemia are: headache, seizure, stroke-like episodes, cognitive dysfunction and coma because the amount of glycogen stored in the brain, between 0.5 and 1.5g, or about 0.1% of total brain weight, can only protect from the hypoglycaemia for a relatively short time [106,110]. Although brain glycogen is depleted during hypoglycaemia, doubts have been expressed about its exact role in brain glucose supply [107]. Apparently, moderate and repeated hypoglycaemia can train the brain to adapt to more extreme hypoglycaemic situations [111].
Hyperglycaemia protection imparted by brain localised glycogen
In terms of hyperglycaemia responsiveness of the brain, Eelco and Ryan [112] have discussed how they believe uncontrolled blood glucose concentrations may damage the brain. Where there is a long history of poor glycaemic control, especially when accompanied by micro- and macrovascular complications, there is an increased risk of cognitive decline. More specifically, in type I diabetics, events like elevated blood glucose concentrations (glucose ‘neuro-toxicity’) and diabetic keto acidosis may alter the integrity of the blood-brain barrier. As a consequence, ‘toxic agents’ are able to enter the central nervous system and affect neurodevelopmental processes. This happens especially during the first several years of life, where there is particular active brain development. They did not really associate this with any defined protective role of brain glycogen, however. Presumably any control that brain-based glycogen could exert on the local glucose concentration to nullify hyperglycaemia is simply overwhelmed.
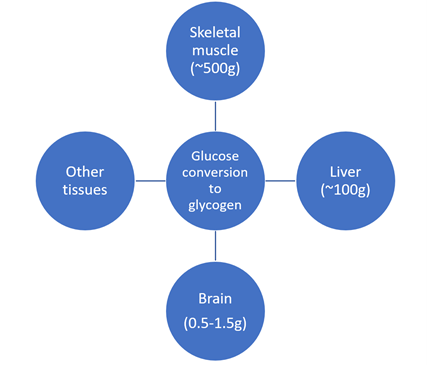
Figure 1: Human glycogen depots [106,110,117].
Glycated Haemoglobin
Glycated haemoglobin (HbA1c) provides an indication of average (and thus including impaired) blood glucose concentration up until the test point reflecting the blood glucose profiles in humans during the past few months before testing. The HbA1c concentrations are associated with cognitive decline in older people (healthy individuals) and those diagnosed with type II diabetes. Altschul et al. [113] discussed how HbA1c concentrations reflect also: (i) micro- and macrovascular complications, (ii) reduction in brain volume and (iii) dementia. They indicated further that the HbA1c - cognitive function relationship, rather than being parallel events, may in fact be revealing causative factors for brain damage. The toxic generation of free radicals accompanying increased HbA1c concentrations may cross over into the brain, affecting cognition [113].
Li et al. [114] have discussed glucose neurotoxicity in more detail than most authors and discussed the overall impact on brain glycogen metabolism. They indicate that indeed hyperglycaemia might affect astrocyte energy metabolism and function. This area requires further investigation [28] but does indicate why hyperglycaemia - in diabetes especially - is very damaging to the brain and supports the discussion that the consumption of glucose that is released slowly into the blood stream rather than in periodic spikes provides cognitive and health advantages.
Lifestyle Changes to Impact on Cognition
As discussed above, both excess calories in the diet with repeated highs and lows of blood glucose concentration, are not conducive to good health. Slowly released glucose from the gut, provided by slowly digestible starch is a much healthier physiological state.
It is reported often that sugars, especially in products like sugary drinks, are detrimental to the body in terms of tooth decay and excess calories leading to obesity and further physiological decline [16-17]. What is harder to define, however, is how exactly these products may be associated with or even cause cognitive impairment/decline [47,115-116]. These types of drinks contain more than just sugar and water and hence, other components may be detrimental to cognitive health. Setting that argument aside, however, it does add further support to the debate on the need to minimise concentrated bursts of sugar ingestion, leading to peaks and troughs in blood glucose, not just for physical health but cognition too.
Although attention is usually paid to the positive role of sustained energy release for physical activity, less is said about how this supply may benefit the functionality of the brain too. As the brain utilises glucose extensively for its activities, it would seem entirely logical that accessibility to a sustained release of glucose (gut via slow digestion and from glycogen stores) would optimise the performance of the brain. This supply not just a control of periods when food is consumed but the choice of energy source and its sustained impact on blood glucose concentration with associated stable impact on insulin concentrations.
Conclusion
People tend to associate low sugar intake with tiredness and high sugar intake with excess calories, tooth decay, obesity, type II diabetes, cardiovascular disease etc. However, less is said about cognition and in particular how avoiding too many highs and lows of blood glucose provides a cognitive benefit. Glycogen stores of glucose, distributed within organs around the body and in the brain, are relevant to overall glucose management for physical energy. However, with respect to cognition, the exact role of glycogen in the brain is still to be defined. There is little doubt that the brain needs to be protected from hypo- and hyperglycaemia to allow normal neurological activity.
With the exception perhaps of honey in only some geographic locations, most evolving humans would have been exposed to sugar within fruit and vegetables with a maximum concentration before drying of about 15% and perhaps up to around 75% for some dried fruit (e.g. pineapple). As cooking skills developed and cooking pots became more readily available, starchy crops as a source of polymerised glucose would have been harvested (eventually farmed) and cooked, allowing the body’s enzymes greater capacity to digest the starch. At the present time, we live in a world of pre-processed foods rich with calories - many from sugars and starches. All this excess energy intake has been coupled with less physical activity and a gradual negative health impact too. Not only has this change impacted on the physical body, cognition has become an ever increasingly important issue with respect to optimal brain food - especially glucose with respect to calories.
It does seem to be the case that the brain operates most efficiently when it is supported by a steady glucose supply. It is difficult to make many cognitive measurements linked to normal rather than abnormal physiological events - e.g. blood glucose concentration versus blood alcohol concentration - for many different reasons. However, there is very good reason to promote good health and mental wellbeing based on a good balanced, energy-controlled diet which promotes a healthy blood glucose profile.
Acknowledgement: No financial and material support were obtained.
Disclosure of interest: The authors report there are no competing interests to declare.
Authorship: Xin Qi: Writing - review and editing; Richard Tester: Writing - original draft.
References
- Rodriguez-Diaz R, Molano RD, Weitz JR, Abdulreda MH, Berman DM, Leibiger B, et al. Paracrine interactions within the pancreatic islet determine the glycaemic set point. Cell Metab, 2018; 27: 549-558.
- Lundqvist MH, Almby K, Abrahamsson N, Eriksson JW. Is the Brain a Key Player in Glucose Regulation and Development of Type 2 Diabetes? Front Physiol, 2019; 10: 457. doi: 10.3389/fphys.2019.00457.
- White H, Venkatesh B. Clinical review: Ketones and brain injury. Crit Care, 2011; 15: 219.
- Mergenthaler P, Lindauer U, Dienel GA, Meisel A. Sugar for the brain: The role of glucose in physiological and pathological brain function. Trends Neurosci, 2013; 36: 587-597.
- Falkowska A, Gutowska I, Goschorska M, Nowacki P, Chlubek D, Baranowska-Bosiacka I. Energy metabolism of the brain, including the cooperation between astrocytes and neurons, especially in the context of glycogen metabolism. Int J Mol Sci, 2015; 16: 25959-25981.
- Magistretti PJ, Allaman I. A cellular perspective on brain energy metabolism and functional imaging. Neuron, 2015; 86: 883-901.
- Roh E, Song DK, Kim M-S. Emerging role of the brain in the homeostatic regulation of energy and glucose metabolism. Exp Mol Med, 2016; 48: e216. doi:10.1038/emm.2016.4.
- Camandola S, Mattson MP. Brain metabolism in health, aging and neurodegeneration. EMBO J, 2017; 36: 1474-1492.
- Puchalska P, Crawford PA. Multi-dimensional roles of ketone bodies in fuel metabolism, signalling, and therapeutics. Cell Metab, 2017; 25: 262-284.
- Sonnay S, Gruetter R, Duarte JMN. How energy metabolism supports cerebral function: Insights from 13C magnetic resonance studies in vivo. Front Neurosci, 2017; 11: 288. doi: 10.3389/fnins.2017.00288.
- Watts ME, Pocock R, Claudianos C. Brain energy and oxygen metabolism: Emerging role in normal function and disease. Front Mol Neurosci, 2018; 11: 216. doi: 10.3389/fnmol.2018.00216.
- Bordone MP, Salman MM, Titus HE, Amini E, Andersen JV, Chakraborti B, et al. The energetic brain - A review from students to students. J Neurochem, 2019; 151: 139-165.
- Cali C, Tauffenberger A, Magistretti P. The strategic location of glycogen and lactate: From body energy reserve to brain plasticity. Front Cell Neurosci, 2019; 13: 82. doi: 10.3389/fncel.2019.00082.
- Dienel GA. Brain glucose metabolism: Integration of energetics with function. Physiol Rev, 2019; 99: 949 -1045.
- Steiner P. Brain fuel utilisation in the developing brain. Ann Nutr Metab, 2019; 75 (Suppl 1): 8-18.
- Qi X, Tester RF. Fructose, galactose and glucose - In health and disease. Clin Nutr ESPEN, 2019; 33: 18-28.
- Qi X, Tester RF. Lactose, maltose, and sucrose in health and disease. Mol Nutr Food Res, 2020; 64(8): Article 1901082.
- Tester RF, Qi X, Karkalas J. Hydrolysis of native starches with amylases. J Animal Feed Sci Technol, 2006; 130: 39-54.
- Qi X, Tester RF. Effect of native starch granule size on susceptibility to amylase hydrolysis. Starch/Stärke, 2016; 68: 807-810.
- Qi X, Tester RF. Effect of composition and structure of native starch granules on their susceptibility to hydrolysis by amylase enzymes. Starch/Stärke, 2016; 68: 811-815.
- Qi X, Tester RF. Heat and moisture modification of native starch granules on susceptibility to amylase hydrolysis. Starch/Stärke, 2016; 68: 816-820.
- Qi X, Tester RF. Impact of starch gelatinisation on digestibility and human health. Starch/Stärke, 2023; Article 2200195. doi 10.1002/star.202200195.
- Qi X, Tester RF. Nutritional uses of starch to provide blood glucose control. Nutrire, 2023; 48: Article 14 doi.org/10.1186/s41110-023-00198-8.
- McNay EC, McCarthy RC, Gold PE. Fluctuations in brain glucose concentration during testing: Dissociations between brain areas and between brain and blood. Neurobiol Learn Mem, 2001; 75: 325-327.
- McNay EC, Gold PE. Food for thought: Fluctuations in brain extracellular glucose provide insight into the mechanisms of memory modulation. Behav Cogn Neurosci Rev, 2002; 1: 264-280.
- Feldman J, Barshi I. The effects of blood glucose levels on cognitive performance: A review of the literature. NASA/TM–2007-214555, 2007.
- Zanchi D, Meyer-Gerspach AC, Schmidt A, Suenderhauf C, Depoorter A, Drewe J, et al. Acute effects of glucose and fructose administration on the neural correlates of cognitive functioning in healthy subjects: A pilot study. Front Psychiatry, 2018; 9: 71. doi: 10.3389/fpsyt.2018.00071.
- Zheng F, Yan L, Yang Z, Zhong B, Xie W. HbA1c, diabetes and cognitive decline: The English longitudinal study of aging. Diabetologia, 2018; 61: 839-848.
- Sommerfield AJ, Deary IJ, Frier BM. Acute hyperglycemia alters mood state and impairs cognitive performance in people with type 2 diabetes. Diabetes Care, 2004; 27: 2335-2340.
- Cox DJ, Kovatchev BP, Gonder-Frederick LA, Summers KH, McCall A, Grimm KJ, et al. Relationships between hyperglycaemia and cognitive performance among adults with type 1 and type 2 diabetes. Diabetes care, 2005; 28: 71-77.
- Weinstein G, Maillard P, Himali JJ, Beiser AS, Au R, Wolf PA, et al. Glucose indices are associated with cognitive and structural brain measures in young adults. Neurol, 2015; 84: 2329-2337.
- Crane PK, Walker R, Hubbard RA, Nathan DM, Zheng H, Haneuse S, et al. Glucose levels and risk of dementia. N Engl J Med, 2013; 369: 540-548.
- Mushtaq G, Khan JA, Kamal MA. Impaired glucose metabolism in Alzheimer’s Disease and diabetes. Enzym Eng, 2015; 4: Article 1000124.
- Calsolaro V, Edison P. Alterations in glucose metabolism in Alzheimer’s Disease. Recent Pat Endocr Metab Immune Drug Discov, 2016; 10: 31-39.
- Duran-Aniotz C, Hetz C. Glucose metabolism: A sweet relief of Alzheimer’s Disease. Curr Biol, 2016; 26: R794-R815.
- Mosconi L, Mistur R, Switalski R, Brys M, Glodzik DL, Rich K, et al. Declining brain glucose metabolism in normal individuals with a maternal history of Alzheimer disease. Neuro, 2009; 72: 513-520.
- Mosconi L, Pupi A, Mony De Leon J. Brain Glucose Hypometabolism and Oxidative Stress in Preclinical Alzheimer’s Disease. Ann N Y Acad Sci, 2008; 1147: 180-195.
- Cunnane SC, Nugent S, Roy M, Courchesne-Loyer A, Croteau E, Tremblay S, et al. Brain fuel metabolism, aging and Alzheimer’s Disease. Nutrition,2011; 27: 3-20.
- Iadecola C. Sugar and Alzheimer’s disease: A bittersweet truth. Nat Neurosci, 2015; 18: 477-478.
- Yang A, Varma VR, Varma S, Casanova R, Dammer E, Pletnikova O, et al. Evidence for brain glucose dysregulation in Alzheimer’s disease. Alzheimers Dement, 2018; 14: 318-329.
- Kuehn BM. In Alzheimer research, glucose metabolism takes centre stage. JAMA, 2020; 323: 297-299.
- Xu W, Qiu C, Winblad B, Fratiglioni L. The effect of borderline diabetes on the risk of dementia and Alzheimer’s Disease. Diabetes, 2007; 56: 211-216.
- Lee HJ, Seo HI, Cha HY, Yang YJ, Kwon SH, Yang SJ. Diabetes and Alzheimer's Disease: Mechanisms and Nutritional Aspects. Clin Nutr Res, 2018; 7: 229-240.
- Johnson ECB, Dammer EB, Duong DM, Ping L, Zhou M, Yin L, et al. Large-scale proteomic analysis of Alzheimer’s disease brain and cerebrospinal fluid reveals early changes in energy metabolism associated with microglia and astrocyte activation. Nat Med, 2020; 26: 769-780.
- Mortby ME, Janke AL, Anstey KJ, Sachdev PS, Cherbuin N. High ‘normal’ blood glucose is associated with decreased brain volume and cognitive performance in the 60s: The PATH through life study. PLoS ONE, 2013; 8(9): e73697. doi:10.1371/journal.pone.0073697.
- Anstey KJ, Sargent-Cox K, Eramudugolla R, Magliano DJ, Shaw JE. Association of cognitive function with glucose tolerance and trajectories of glucose tolerance over 12 years in the AusDiab study. Alzheimer's Res Ther, 2015; 7: 48. DOI 10.1186/s13195-015-0131-4.
- Chong CP, Shahar S, Haron H, Che Din N. Habitual sugar intake and cognitive impairment among multi-ethnic Malaysian older adults. Clin Interv Aging, 2019; 14: 1331-1342.
- Liu X, Li Y, Chen X, Yin H, Li F, Chen N, et al. Revisiting the mechanisms linking blood glucose to cognitive impairment: new evidence for the potential important role of klotho. Front Endocrinol, 2024; 15, Article 1323407. doi: 10.3389/fendo.2024.1323407.
- Lacy ME, Gilsanz P, Eng C, Beeri MS, Karter AJ, Whitmer RA. Severe hypoglycaemia and cognitive function in older adults with Type 1 diabetes: The study of longevity in diabetes (SOLID). Diabetes Care, 2020; 43: 541-548.
- McNay EC, Cotero VE. Mini-review: Impact of recurrent hypoglycaemia on cognitive and brain function. Physiol Behav, 2010; 100: 234-238.
- Hawks ZW, Beck ED, Jung L, Fonseca LM, Sliwinski MJ, Weinstock RS, et al. Dynamic associations between glucose and ecological momentary cognition in Type 1 Diabetes. NPJ Digit Med, 2024; 7, Article 59. doi.org/10.1038/s41746-024-01036-5.
- Graveling AJ, Deary IJ, Frier BM. Acute hypoglycaemia impairs executive cognitive function in adults with and without type 1 diabetes. Diabetes care, 2013; 36: 3240-3246.
- Kaplan RJ, Greenwood CE, Winocur G, Wolever TMS. Cognitive performance is associated with glucose regulation in healthy elderly persons and can be enhanced with glucose and dietary carbohydrates. Am J Clin Nutr, 2000; 72: 825-836.
- Rosenthal JM, Amiel SA, Yaguez L, Bullmore E, Hopkins D, Evans M, et al.The effect of acute hypoglycaemia on brain function and activation. A functional magnetic resonance imaging study. Diabetes, 2001; 50: 1618-1626.
- Hamed SA. Brain injury with diabetes mellitus: Evidence, mechanisms and treatment implications. Expert Rev Clin Pharmacol, 2017; 10: 409-428.
- Kirvalidze M, Hodkinson A, Storman D, Fairchild TJ, Bała MM, Beridze G, et al. The role of glucose in cognition, risk of dementia, and related biomarkers in individuals without type 2 diabetes mellitus or the metabolic syndrome: A systematic review of observational studies. Neurosci Biobehav Rev, 2022; 135: Article 104551. doi.org/10.1016/j.neubiorev.2022.104551.
- Cryer PE. Hypoglycaemia, functional brain failure and brain death. J Clin Investig, 2007; 117: 868-870.
- Simpson IA, Dwyer D, Malide D, Moley KH, Travis A, Vannucci SJ. The facilitative glucose transporter GLUT3: 20 years of distinction. Am J PhysiolEndocrinolMetab, 2008; 295: E242-E253.
- Berg JM, Tymoczko JL, Stryer L. Each Organ Has a Unique Metabolic Profile (Section 30.2). New York: W H Freeman. Biochemistry (5th Edn), 2002.
- Deem JD, Muta K, Scarlett JM, Morton GJ, Schwartz MW. How should we think about the role of the brain in glucose homeostasis and diabetes? Diabetes, 2017; 66: 1758-1765.
- Dias IR, Santos CD, Magalhaes COD, de Oliveira LRS, Peixoto MFD, De Sousa RAL, et al. Does calorie restriction improve cognition? IBRO Reports, 2020; 9: 37-45.
- Zhang S, Xiao Y, Cheng Y, Ma Y, Liu J, Li C, et al. Associations of sugar intake, high-sugar dietary pattern, and the risk of dementia: a prospective cohort study of 210,832 participants. BMC Med, 2024; 22: Article 298. doi.org/10.1186/s12916-024-03525-6.
- Qi X, Tester RF. Is starch or maltodextrin ‘glucose?’ Starch/Staerke, 2018; 70: Article 1700304.
- Qi X, Tester RF. Nutritional uses of starch to provide blood glucose control. Nutrire, 2022; 48: 14. https://doi.org/10.1186/s41110-023-00198-8.
- Qi X, Tester RF. Impact of starch gelatinisation on digestibility and human health. Starch, 2022; 75: 2200195. https://doi.org/10.1002/star.202200195.
- Qi X, Ta NM, Tester RF. Savoury cracker development for blood glucose control and management. Bioact Carbohydr Diet Fibre, 2020; 24: Article 100249.
- Tester RF, Qi X. β-limit Dextrin - Properties and applications. Food Hydrocoll, 2011; 25: 1899-1903.
- Kendig MD, Lin CS, Beilharz JE, Rooney KB, Boakes RA. Maltodextrin can produce similar metabolic and cognitive effects to those of sucrose in the rat. Appetite. 2014;77: 1-12.
- Fischer K, Colombani PC, Langhans W, Wenk C. Cognitive performance and its relationship with postprandial metabolic changes after ingestion of different macronutrients in the morning. Br J Nutr, 2001; 85: 393-405.
- Lehmann U, Robin F. Slowly digestible starch - its structure and health implications: A review. Trends Food Sci Technol. 2007;18: 346-355.
- Blundell J, Gumaste D, Handley R, Dye L. Diet, behaviour and cognitive functions: A psychobiological view. Scand J Food Nutr, 2003; 47: 85-91.
- Brouns F, Dye L. Starch: Physical and mental performance. In: Eliasson AC, editor. Starch in Food - Structure, Function and Applications. Cambridge: Woodhead Publishing Series in Food Science, Technology and Nutrition, 2004; p. 505-540.
- Hendrich S. Starch: Physical and mental performance and potential health problems. In: Sjoo M, Nilsson L, editors. Starch in Food - Structure, Function and Applications. 2nd ed. Cambridge: Woodhead Publishing Series in Food Science, Technology and Nutrition, 2018; p. 855-871.
- Nilsson A, Radeborg K, Bjorck I. Effects on cognitive performance of modulating the postprandial blood glucose profile at breakfast. Eur J Clin Nutr, 2012; 66: 1039-1043.
- Sünram-Lea SL, Owen L. The impact of diet-based glycaemic response and glucose regulation on cognition: evidence across the lifespan. Proc Nutr Soc, 2017; 76: 466-477. doi:10.1017/S0029665117000829.
- Mattson MP. An evolutionary perspective on why food overconsumption impairs cognition. Trends Cogn Sci, 2019; 23: 200-2012.
- Bezborodkina NN, Chestnova AY, Vorobev ML, Kudryavtsev BN. Spatial structure of glycogen molecules in cells. Biochem (Mosc), 2018; 83: 467-482.
- Prats C, Graham TE, Shearer XJ. The dynamic life of the glycogen granule. J Biol Chem, 2018; 293: 7089-7098.
- Ellingwood SS, Cheng A. Biochemical and clinical aspects of glycogen storage diseases. J Endocrinol, 2018; 238: R131-R141.
- Brewer MK, Gentry MS. Brain glycogen structure and its associated proteins: Past, present and future. Adv Neurobiol, 2019; 23: 17-81.
- Swanson RA. A thermodynamic function of glycogen in brain and muscle. Prog Neurobiol, 2020; 189: Article 101787.
- Turnbull J, Striano P, Genton P, Carpenter S, Ackerley CA, Minassian BA. Lafora disease. Epileptic Disord, 2016; 18 (Suppl 2): 38-62. doi:10.1684/epd.2016.0842.
- Gentry MS, Guinovart JJ, Minassian BA, Roach PJ, Serratosaa JM. Lafora disease offers a unique window into neuronal glycogen metabolism. J Biol Chem, 2018; 293: 7117-7125.
- Brewer MK, Uittenbogaard A, Austin GL, Segvich DM, DePaoli-Roach A, Roach PJ, et al. Targeting pathogenic Lafora bodies in Lafora disease using an antibody-enzyme fusion. Cell Metab, 2019; 30: 689-705.
- Sinadinos C, Valles-Ortega J, Boulan L, Solsona E, Tevy MF, Marquez M, et al. Neuronal glycogen synthesis contributes to physiological aging. Aging Cell, 2014; 13: 935-945.
- Novak A, Vizjak K, Rakusa M. Cognitive impairment in people with epilepsy. J Clin Med, 2022; 11: Article 267. doi.org/10.3390/jcm11010267.
- López-Ramos JC, Duran J, Gruart A, Guinovart JJ, Delgado-García JM. Role of brain glycogen in the response to hypoxia and in susceptibility to epilepsy. Front Cell Neurosci, 2015; 9: article 431. doi:10.3389/fncel.2015.00431.
- Rohn TT. Corpora amylacea in neurodegenerative diseases: Cause or effect? Int J Neurol Neurother, 2015; 2(3): 031. doi: 10.23937/2378-3001/2/2/1031.
- Pisa D, Alonso R, Rábano A, Carrasco L. Corpora amylacea of brain tissue from neurodegenerative diseases are stained with specific antifungal antibodies. Front Neurosci, 2016; 10: 86. doi: 10.3389/fnins.2016.00086.
- Augé E, Cabezón I, Pelegri C, Vilaplana J. New perspectives on corpora amylacea in the human brain. Sci Rep, 2017; 7: 41807. doi: 10.1038/srep41807
- Augé E, Duran J, Guinovart JJ, Pelegri C, Vilaplana J. Exploring the elusive composition of corpora amylacea of human brain. Sci Rep, 2018; 8:13525. doi:10.1038/s41598-018-31766-y.
- Navarro PP, Genoud C, Castano-Diez D, Graf-Meyer A, Lewis AJ, deGier Y, et al. Cerebral Corpora amylacea are dense membranous labyrinths containing structurally preserved cell organelles. Sci Rep, 2018; 8: 18046. doi:10.1038/s41598-018-36223-4.
- Riba M, Augea E, Campo-Sabariza J, Moral-Antera D, Molina-Porcel L, Ximelis T, et al. Corpora amylacea act as containers that remove waste products from the brain. PNAS, 2019; 116: 26038-26048.
- Qi X, Tester RF. The ‘epileptic diet’- ketogenic and/or slow release of glucose intervention: A review. Clin Nutr, 2020; 39: 1324-1330.
- DiNuzzo M, Mangia S, Maraviglia B, Giove F. Does abnormal glycogen structure contribute to increased susceptibility to seizures in epilepsy? Metab Brain Dis, 2015; 30: 307-316.
- Gibson EL. Carbohydrates and mental function: feeding or impeding the brain? Nutr Bull, 2007; 32 (suppl 1): 71-83.
- Cai Y, Guo H, Fan Z, Zhang X, Wu D, Tang W, et al. Glycogenolysis is crucial for astrocytic glycogen accumulation and brain damage after reperfusion in ischemic stroke. iScience, 2020; 23: Article 101136. doi.org/10.1016/j.isci.2020.101136.
- Matsui T, Omuro H, Liu Y-F, Soya M, Shima T, McEwen BS, et al. Astrocytic glycogen-derived lactate fuels the brain during exhaustive exercise to maintain endurance capacity. PNAS, 2017; 114: 6358–6363.
- Ӧz G, Seaquist ER, Kumar A, Criego AB, Benedict LE, Rao JP, et al. Human brain glycogen content and metabolism: implications on its role in brain energy metabolism. Am J Physiol Endocrinol Metab, 2007; 292: E946-E951. doi:10.1152/ajpendo.00424.2006.
- Waitt AE, Reed L, Ransom BR, Brown AM. Emerging roles for glycogen in the CNS. Front Mol Neurosci, 2017; 10: 73. doi: 10.3389/fnmol.2017.00073.
- Duran J, Gruart A, Varea O, Lopez-Soldado I, Delgado-Garcia JM, Guinovart JJ. Lack of neuronal glycogen impairs memory formation and learning-dependent synaptic plasticity in mice. Front Cell Neurosci, 2019; 13: 374. doi: 10.3389/fncel.2019.00374.
- Rich LR, Harris W, Brown AM. The role of brain glycogen in supporting physiological function. Front Neurosci, 2019; 13: 1176. doi: 10.3389/fnins.2019.01176.
- Choi I-Y, Seaquist ER, Gruetter R. Effect of hypoglycaemia on brain glycogen metabolism in vivo. J Neurosci Res, 2003; 72: 25-32.
- Ӧz G, Kumar A, Rao JP, Kodl CT, Chow L, Eberly LE, et al. Human brain glycogen metabolism during and after hypoglycemia. Diabetes, 2009; 58: 1978-1985.
- Tesfaye N, Seaquist ER. Neuroendocrine responses to hypoglycaemia. Ann N Y Acad Sci, 2010; 1212: 12-28.
- Neil WP, Hemmen TM. Neurologic manifestations of hypoglycaemia. In: Rigobelo E, editor. Diabetes - Damages and Treatments. London: IntechOpen Limited, 2011; p. 259-274.
- McCrimmon RJ. Update in the CNS response to hypoglycaemia. J Clin Endocrinol Metab, 2012; 97: 1- 8.
- Rehni AK, Dave KR. Impact of hypoglycaemia on brain metabolism during diabetes. Mol Neurobiol, 2018; 55: 9075-9088.
- Soya M, Matsui T, Shima T, Jesmin S, Omi N, Soya H. Hyper-hippocampal glycogen induced by glycogen loading with exhaustive exercise. Sci Rep, 2018; 8: 1285. DOI:10.1038/s41598-018-19445-4.
- Nilsson M, Jensen N, Gejl M, Bergmann ML, Storgaard H, Zander M, et al. Experimental non-severe hypoglycaemia substantially impairs cognitive function in type 2 diabetes: a randomised crossover trial. Diabetologia, 2019; 62: 1948-1958.
- Puente EC, Silverstein J, Bree AJ, Musikantow DR, Wozniak DF, Maloney S, et al. Recurrent moderate hypoglycaemia ameliorates brain damage and cognitive dysfunction induced by severe hypoglycaemia. Diabetes, 2010; 59: 1055-1062.
- Eelco van D, Ryan CM. Diabetes mellitus in the young and the old: Effects on cognitive functioning across the life span. Neurobiol Dis, 2020; 134: Article 104608.
- Altschul DM, Starr JM, Deary IJ. Cognitive function in early and later life is associated with blood glucose in older individuals: Analysis of the Lothian Birth Cohort of 1936. Diabetologia, 2018; 61: 1946-1955.
- Li W, Choudhury GR, Winters A, Prah J, Lin W, Liu R, et al. Hyperglycaemia alters astrocyte metabolism and inhibits astrocyte proliferation. Aging Dis, 2018; 9: 674-684.
- Ye X, Gao X, Scott T, Tucker KL. Habitual sugar intake and cognitive function among middle-aged and older Puerto Ricans without diabetes. Br J Nutr, 2011; 106: 1423-1432.
- Crichton GE, Elias MF, Torres RV. Sugar-sweetened soft drinks are associated with poorer cognitive function in individuals with type 2 diabetes: The Maine–Syracuse Longitudinal Study. Br J Nutr, 2016; 115: 1397-1405.
- Jensen J, Rustad PI, Kolnes AJ, Lai Y-C. The role of skeletal muscle glycogen breakdown for regulation of insulin sensitivity by exercise. Front Physiol, 2011; 2: Article 112.

