Anti-SRP Myositis: Poor Response and Therapeutic Challenges
Huynh Thi Thuy Trang1, Vo Lam Binh1, Pham Ngoc Quoc My1, Duong Minh Tri1 and Vu Hoang Phuong Thu2,*
1Department of Rheumatology, Nhan dan Gia Dinh Hospital, Ho Chi Minh city, Vietnam
2Pham Ngoc Thach University of Medicine, Ho Chi Minh city, Vietnam
Received Date: 06/06/2025; Published Date: 09/07/2025
*Corresponding author: Vu Hoang Phuong Thu, Pham Ngoc Thach University of Medicine, Ho Chi Minh city, Vietnam
Abstract
Background: Anti-SRP (signal recognition particle) myopathy is a rare and severe subtype of immune-mediated necrotizing myopathy (IMNM), characterized by rapidly progressive proximal muscle weakness, markedly elevated creatine kinase (CK) levels, and limited response to conventional immunosuppressive therapies. Early diagnosis and effective treatment are crucial to prevent long-term disability.
Case Presentation: We report the case of a 55-year-old woman with a three-year history of progressive proximal lower limb weakness, initially misdiagnosed as compression neuropathy. Despite treatment with corticosteroids and multiple immunosuppressive agents—including methotrexate, mycophenolate mofetil, and cyclophosphamide—the patient showed no significant improvement. Diagnostic workup revealed high anti-SRP antibody levels, consistent with IMNM. Due to poor response to conventional therapy, rituximab, a B-cell depleting agent, was introduced. Following two cycles of rituximab, the patient demonstrated marked improvement in muscle strength, daily functioning, and a significant reduction in CK levels.
Conclusion: This case highlights the diagnostic challenges of Anti-SRP myopathy and the potential efficacy of rituximab in treatment-resistant cases. It underscores the importance of early recognition and supports B-cell depletion therapy as a promising option for managing refractory Anti-SRP myopathy.
Keywords: Anti-SRP myopathy; Immune-mediated necrotizing myopathy (IMNM); Autoimmune myositis; Proximal muscle weakness; Rituximab; B-cell depletion therapy; Refractory myopathy; Creatine kinase elevation; Autoantibodies; Immunosuppressive treatment
Introduction
Immune-mediated necrotizing myopathy (IMNM) is a subgroup of autoimmune myositis. This condition is divided into three main types: Anti-SRP (signal recognition particle) myopathy, anti- HMG-CR (HMG-CoA reductase) myopathy, and seronegative immune-mediated necrotizing myopathy. Anti-SRP Autoantibody-Positive Myopathy is approximately 3–6% of all patients with idiopathic inflammatory myopathies. It is rapidly progressive symmetrical proximal muscle weakness, severe myalgias [11], markedly elevated CK levels and the rashes of DM are notably absent. Cardiopulmonary complaints are common, including dyspnea on exertion, and ILD has been reported in up to 25% of patients [12]. SRP-IMNM is thought to be one of the most incapacitating IIMs, and despite treatment, patients frequently experience poor muscle recovery [13]. According to a previous study, after four years, immunotherapy produces satisfactory results for 50% of patients with SRP-IMNM, and the majority of patients' serum CK levels do not return to normal [14].
We report a case of SRP myopathy with progressive proximal muscle weakness over the past three years, which showed poor response to combination therapy with corticosteroids and conventional immunosuppressive drugs. After being treated with B-cell depletion therapy using rituximab, the patient experienced significant improvement in muscle strength, ability to perform daily activities, and disease activity.
Case Report
A 55-year-old female with no significant medical history and no previous internal or autoimmune diseases was examined at the Musculoskeleton Clinic due to progressive leg weakness over the last 3 years. In March 2022, she started experiencing fatigue and mild weakness along with numbness in both legs, without any lower back pain, and was still able to work and perform her usual activities. She visited the clinic and was diagnosed with uterine fibroids that were compressing the nerve and muscle, leading to her undergoing a hysterectomy. Nevertheless, she continued to have leg weakness after the surgery. When the patient arrived at the musculoskeletal clinic, she had to place both hands on the floor in order to stand up because of weakness in both lower limbs (Figure 1). The progression of the patient's illness is as follows in Table 1.
The patient did not agree to perform a muscle biopsy.
Lumbar spine MRI: lumbar spine degeneration - disc protrusion L2/3, L3/4, L4/5, no spinal stenosis, no root compression
Liver and renal function test results were normal.
EULAR/ACR 2017 criteria (without muscle biopsy): 5.8 score
The patient was treated for nearly 2 years with corticosteroids, Methotrexate, Mycophenolate mofetil, Cyclophosphamide and Rituximab (Table 1, Figure 3).
Table 1: The progression of the patient's illness (symptoms, laboratory, diagnosis and treatment) in 2 years.
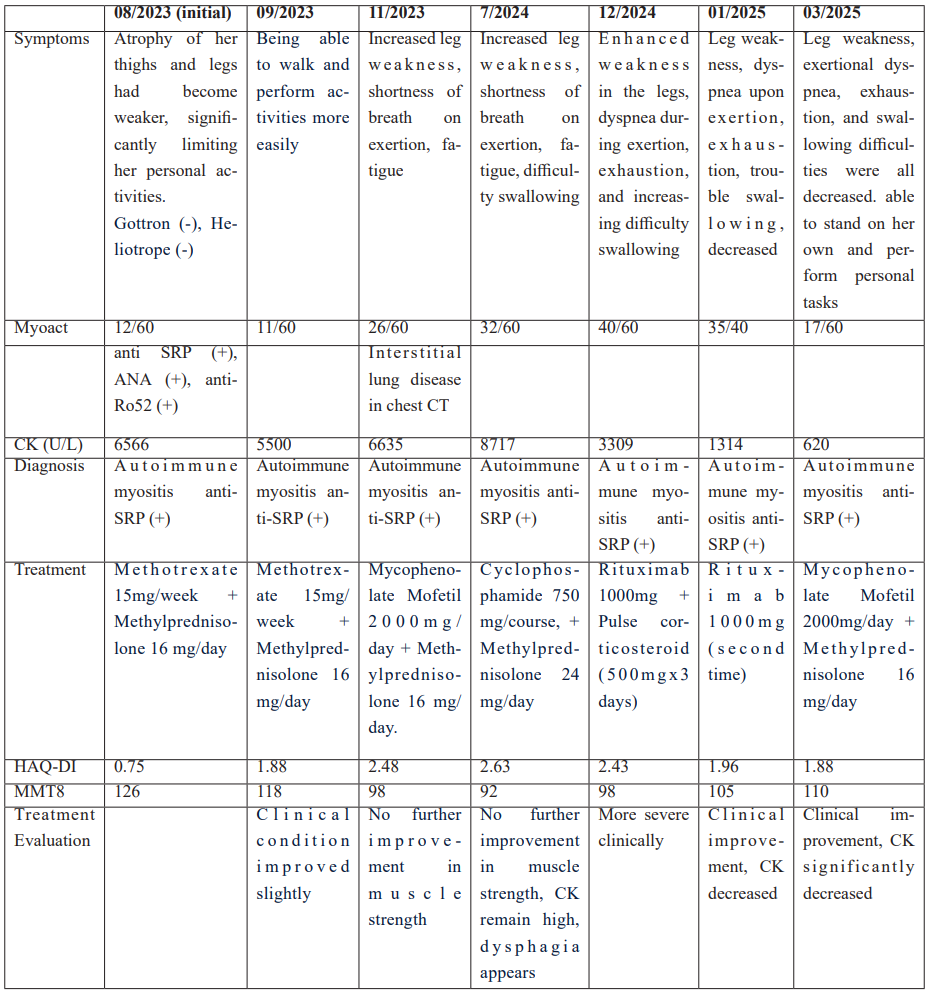

Figure 1: The patient had weak muscles in the limbs, difficulty standing up, and need to place both hands on the ground to stand.

Figure 2: After using Rituximab for two months, the patient was able to walk.
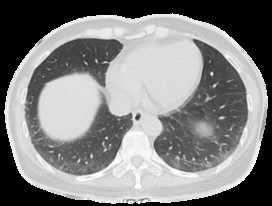
Figure 3: Peripheral reticular interstitial lesions of the lower lobes of lungs.
Discussion
In this case report, we describe the clinical features, paraclinical findings, and treatment response to various immunosuppressive agents in a patient diagnosed with Anti-SRP myopathy. Anti-SRP myopathy is a subtype of immune-mediated necrotizing myopathy (IMNM), characterized histopathologically by muscle fiber necrosis at different stages, scattered distribution, sparse lymphocytic infiltration, and deposition of C5b-9 membrane attack complexes on the sarcolemma [1,4]. This is a rare disease, first described in 2004, accounts for approximately 3–6% of patients with IIM. According to the 2017 EULAR/ACR classification criteria, Anti-SRP autoantibodies are not included, and IIM is divided into four major subgroups: polymyositis, dermatomyositis, amyopathic dermatomyositis, and inclusion body myositis. IMNM is not classified as a separate entity but is instead included under polymyositis, which is problematic due to the distinct histopathology of IMNM compared to polymyositis. A study by Shogo Komaki et al on the clinical features of IMNM indicated that approximately 20% of Anti-SRP myopathy patients are not classified as autoimmune myositis based on the EULAR/ACR 2017 criteria [5]. These findings highlight the diagnostic challenges clinicians face when identifying and diagnosing Anti-SRP myopathy. Indeed, before a definitive diagnosis was made, our patient had been experiencing leg weakness since March 2022, but was initially diagnosed with compression neuropathy due to uterine fibroids and underwent surgery. Furthermore, when applying the EULAR/ACR 2017 criteria, the patient’s total score was 5.8, classifying the case only as probable IIM, due to the lack of Anti-SRP autoantibody in the criteria. Recognizing IMNM as a distinct disease entity, the European Neuromuscular Centre (ENMC) issued diagnostic guidelines in 2018 with three main criteria: elevated muscle enzymes, proximal muscle weakness, and positive anti-SRP antibodies [1]. Our patient met the ENMC diagnostic criteria.
Our female patient presented with progressive proximal lower limb muscle weakness over 3 years, impaired activities of daily living, elevated muscle enzymes, and thigh muscle atrophy developing within only 7 months after disease onset. Throughout the disease, the patient did not experience fever or signs of infection. Regarding her medical history, she had not used any medications affecting muscle function over the past 3 years, did not consume alcohol, and had no exposure to toxins. Paraclinical investigations, including lumbar MRI, excluded spinal cord and lumbar root pathology. Electromyography of the lower limbs showed no peripheral neuropathy. Electrolyte levels, thyroid function tests, and blood glucose were all within normal limits. Autoimmune tests revealed a high positivity for anti-SRP antibodies. Therefore, the clinical and paraclinical features of our case are consistent with those reported in the literature [1,2]. Anti-SRP myopathy is more common in women than in men (with a 3:1 ratio) and typically affects middle-aged individuals. These patients usually present with rapid, symmetrical proximal muscle weakness, severe muscle pain, significantly elevated muscle enzymes, and ultimately muscle atrophy and disability if not treated promptly. Furthermore, these patients tend to recover poorly, with only half achieving near-normal or full muscle strength after 4 years of immunosuppressive therapy. Approximately 19% of patients with Anti-SRP myopathy develop pulmonary involvement on chest CT, primarily exhibiting a non-specific interstitial pneumonia (NSIP) pattern [6,7]. While most of these cases present with mild to moderate symptoms and slow disease progression, this patient cohort frequently experiences a higher incidence of interstitial lung disease and respiratory failure compared to those with Anti-HMG-CR myopathy [6]. This is particularly true when diagnosis and initiation of immunosuppressive therapy are delayed. For our patient, the extended duration of the diagnostic workup and the subsequent delay in treatment initiation directly contributed to the rapid development of exertional dyspnea and significant lung lesions on chest CT within merely four months of a definitive diagnosis. Therefore, Anti-SRP myopathy remains one of the most debilitating autoimmune myopathies, which underscores the importance of early diagnosis and intervention.
In this case, muscle biopsy of the proximal lower limbs was not performed due to the patient concerns about potential complications. It is important to note that while histopathological findings can be suggestive, they are not entirely specific for anti-SRP myopathy [1,3]
In our patient, treatment was initiated with a combination of prednisolone (1 mg/kg) and methotrexate, starting at 10 mg/week and increasing to 15 mg/week due to the severity and rapid progression of muscle weakness. Subsequently, the treatment regimen was switched to mycophenolate mofetil and cyclophosphamide (5 cycles), respectively, as there was no improvement in muscle strength, mobility, and muscle enzyme levels continued to rise. The challenges in treating Anti-SRP myopathy have been documented in other reports [8,9]. In a study of 100 patients in Japan, half of the patients were refractory to treatment, and the other half relapsed when corticosteroid or conventional immunosuppressive doses were reduced. Since Anti-SRP myopathy is a rare disease, there have been no randomized controlled trials conducted to date, and no official treatment guidelines exist for Anti-SRP myopathy.
However, there has been increasing interest in the role of B lymphocytes, given the success of Rituximab, a monoclonal antibody targeting CD20 on B cells, in treating refractory autoimmune diseases [4]. Rituximab initiates B cell lysis through complement activation and antibody-dependent cell-mediated cytotoxicity. Furthermore, several features of Anti-SRP myopathy suggest a role for humoral immunity in its pathogenesis, including deposition of membrane attack complexes on sarcolemma and small blood vessels in muscle fascicles, non-inflammatory small vessel damage, and the correlation between autoantibody levels and disease activity. These findings imply that humoral immune responses, with B cells as the primary cellular component, play a significant role in the disease's pathogenesis. Indeed, in a case series by Ritu Valiyil et al, eight patients with Anti-SRP myopathy who did not respond to initial treatment with corticosteroids and conventional immunosuppressive agents showed significant improvement in muscle strength and enzyme levels following rituximab infusion [10]. In our case, after discussion with the patient and obtaining consent, rituximab was administered in combination with corticosteroid pulse therapy, given over two cycles, 2 weeks apart. The follow-up visit revealed significant clinical improvement (in muscle strength and HAQ-DI score) and a decrease in muscle enzyme levels (Figure 2).
Conclusion
Anti – SRP myopathy is a distinct and severe subtype of immune-mediated necrotizing myopathy, characterized by rapidly progressive proximal muscle weakness, early muscle atrophy, and poor recovery. Early recognition and aggressive immunosuppressive therapy are crucial to prevent long-term disability. We report a case of Anti-SRP myopathy with subacute muscle weakness, initially unresponsive to corticosteroids and conventional immunosuppressive drugs. Rituximab was subsequently introduced, resulting in clinical improvement and disease stabilization. This case underscores the importance of early diagnosis and supports B-cell depletion therapy as an effective option in refractory or severe Anti-SRP myopathy.
References
- Allenbach Y, Mammen AL, Benveniste O, Stenzel W. Immune-Mediated Necrotizing Myopathies Working Group. 224th ENMC International Workshop: Clinico-sero-pathological classification of immune-mediated necrotizing myopathies. Neuromuscul Disord, 2018; 28(1): 87-99.
- Adler B, Christopher-Stine L. Immune-Mediated Necrotizing Myopathy (IMNM). In: Aggarwal R, Oddis CV. Managing Myositis: A Practical Guide. Switzerland, Springer Cham, 2020; p. 225-235.
- Pinal-Fernandez I, Parks C, Werner JL, Albayda J, et al. Longitudinal Course of Disease in a Large Cohort of Myositis Patients with Autoantibodies Recognizing the Signal Recognition Particle. Arthritis Care Res (Hoboken), 2017; 69(2): 263-270.
- Nagaraju K, Aggarwal R, Lundberg IE. Inflammatory Diseases of Muscle and Other Myopathies. In: Firestein GS, McInnes IB, Koretzky GA, Mikuls TR, Neogi T, O’Dell JR. Firestein & Kelley’s Textbook of Rheumatology, 12th ed. Philadelphia, Elsevier, 2025; p. 1533-1562.
- Komaki S, Kubota A, Kitamura A, Maeda M, et al. Clinicopathological features of anti-HMGCR and anti-SRP myopathies that do not satisfy the EULAR/ACR criteria of inflammatory myopathies. Rheumatology (Oxford), 2025. doi:10.1093/rheumatology/keaf183.
- Watanabe Y, Uruha A, Suzuki S, Nakahara J, et al. Clinical features and prognosis in anti-SRP and anti-HMGCR necrotising myopathy. J Neurol Neurosurg Psychiatry, 2016; 87(10): 1038-1044.
- Qureshi A, Brown D, Brent L. Anti-Signal Recognition Particle Antibody-Associated Severe Interstitial Lung Disease Requiring Lung Transplantation. Cureus, 2020; 12(5): e7962. doi: 10.7759/cureus.7962.
- Suzuki S, Nishikawa A, Kuwana M, Nishimura H, et al. Inflammatory myopathy with anti-signal recognition particle antibodies: case series of 100 patients. Orphanet J Rare Dis, 2015; 10: 61.
- Miller T, Al-Lozi MT, Lopate G, Pestronk A. Myopathy with antibodies to the signal recognition particle: clinical and pathological features. J Neurol Neurosurg Psychiatry, 2002; 73: 420–428.
- Valiyil R, Casciola-Rosen L, Hong G, Mammen A, et al. Rituximab therapy for myopathy associated with anti-signal recognition particle antibodies: a case series. Arthritis Care Res (Hoboken), 2010; 62(9): 1328-1334.

