Unmasking a Mild Phenotype in A Compound Heterozygote For Βeta-Thalassemia: The Diagnostic and Reproductive Implications of Co-Inherited Alpha-Globin Variants
Stefania Mariani1,*, Serena Bucossi1, Rosanna Squitti1,2,#, Adelina Franciulli1, Mauro Ciro Antonio Rongioletti1,# and Gioia Mastromoro1,#
1Department of Laboratory Science, Research and Development Division, Ospedale Isola Tiberina Gemelli Isola Rome, Italy
2eCampus University, Como, Novedrate, Italy, Department of Theoretical and Applied Sciences, Viale Massenzio Masia, 26, 22100 Como, Italy
Received Date: 28/05/2025; Published Date: 04/07/2025
*Corresponding author: Mariani Stefania Squitti Rosanna, Department of Laboratory Science, Research and Development Division, Ospedale Isola Tiberina Gemelli Isola Rome, Italy
#Equally contributing authors
Abstract
Beta-thalassemia is a hereditary hemoglobinopathy due to HBB gene variants, resulting in reduced or absent beta-globin synthesis. Accurate molecular diagnosis is crucial during reproductive planning.
We present a 33-year-old man referred for genetic counselling during his partner’s pregnancy. Laboratory tests showed microcytosis and hyperferritinemia (ferritin: 866.8 µg/L; transferrin saturation: 55.87%). Common HFE variants for hereditary hemochromatosis were excluded. Sequencing of HBB, HBA1, and HBA2 revealed three heterozygous pathogenic variants: c.118C>T and c.6259_5720del (Sicilian variant) in HBB, and c.427T>A in HBA2. Segregation confirmed compound heterozygosity in HBB. His partner carried a HBB variant of uncertain significance, implying reproductive risk.
Although the genotype suggested beta-thalassemia major, the proband exhibited only mild hematologic findings, consistent with thalassemia intermedia. The co-inherited HBA2 variant likely modulated the phenotype.
This case highlights the need for comprehensive molecular testing in individuals with atypical hematologic parameters, enabling accurate diagnosis, personalized management, and informed reproductive risk assessment.
Keywords: Alpha-thalassemia; Beta-thalassemia; HBA1; HBA1; HBB
Introduction
Thalassemia represents a group of inherited hemoglobin disorders caused by variations that impair the synthesis of one or more globin chains, leading to variable degrees of anemia. The condition is broadly classified into alpha-thalassemia (MIM #604131) and beta-thalassemia (MIM #613985), depending on whether the alpha or beta globin chains are affected. Alpha-thalassemia results from deletions or point mutations in the HBA1 (HEMOGLOBIN-ALPHA LOCUS 1; HBA1, MIM *141800) and the HBA2 (HEMOGLOBIN-ALPHA LOCUS 2; HBA2, MIM *141850) genes located on chromosome 16, whereas beta-thalassemia (BT, MIM# 613985) is caused by pathogenic variants in the HBB gene on chromosome 11, resulting in either a partial (β⁺) or complete (β⁰) deficiency of beta-globin chain production [1].
β-thalassemia major (Cooley’s anemia) typically presents in early infancy with transfusion-dependent microcytic anemia and complications such as iron overload and splenomegaly. β-thalassemia intermedia encompasses a heterogeneous spectrum of disease severity often associated with compound heterozygosity of causative variants in HBB or can be the result of the co-inheritance of pathogenic alterations with other modifying genes [2].
β-thalassemia minor is the condition usually detected in asymptomatic individuals carrying a pathogenic variant in the HBB gene.
Beta-thalassemia trait is often suspected in individuals with reduced mean corpuscular volume (MCV), increased red cell distribution width (RDW), and elevated hemoglobin A2 (HBA2) on electrophoresis, although iron deficiency may mask these findings [3]. Differentiating iron deficiency anemia from thalassemia trait is essential and can be achieved through iron studies, including serum ferritin, transferrin saturation, and total iron-binding capacity. Molecular testing plays a pivotal role in cases with inconclusive hematologic parameters, to estimate reproductive risks. The advent of next-generation sequencing (NGS) has significantly enhanced the ability to detect rare or complex hemoglobinopathies, even in patients with mild phenotypes or atypical presentations [4].
Although genotypic data often correlates with clinical severity, several modifiers may influence the expression of BT. These include co-inherited alpha-thalassemia loss-of-function variants, that alter fetal hemoglobin production (e.g., BCL11A, HBS1L-MYB) [4-6], and other epigenetic factors. In particular, co-inheritance of alpha-globin gene alterations can reduce the α/β globin chain imbalance, resulting in a milder clinical presentation even in individuals carrying variants associated with β-thalassemia major [4-6].
We report on a man with laboratory findings suggestive of a monoallelic variant in HBB, in which sequencing analysis revealed he was an individual with intermedia beta-thalassemia.
Materials and Methods
Following informed consent, the proband underwent a full diagnostic workup including complete blood count, iron studies (serum ferritin, serum iron, transferrin, and transferrin saturation), and haemoglobin electrophoresis. Molecular analyses were performed on peripheral blood samples collected in EDTA tubes.
Genomic DNA was extracted using standard procedures. Targeted sequencing of the HBB, HBA1 and HBA2 genes was carried out using a NGS panel designed for hemoglobinopathies (Devyser Thalassemia kit). Variant classification was performed according to the American College of Medical Genetics and Genomics (ACMG) guidelines [5]. Segregation analysis was conducted by testing both parents of the proband.
Additionally, the most common variants associated with hereditary hemochromatosis type 1 (HFE gene: C282Y, H63D, S65C) were screened using Multiplex PCR techniques (Devyser HFE kit). The partner of the proband underwent molecular testing for HBB, HBA1 genes to assess reproductive risk.
Case Presentation
A 33-year-old Caucasian male was referred to genetic counselling during his partner’s first pregnancy, because of a family history of neurodevelopmental delay and epilepsy.
At the time of evaluation, the proband was clinically asymptomatic. He reported no history of blood transfusions, no evidence of splenomegaly, and normal growth and development. Complete blood count showed a markedly reduced mean corpuscular volume (MCV) of 63.5 fL (reference: 80–100 fL), low mean corpuscular hemoglobin (MCH) of 21.3 pg (reference: 27.2–33.5 pg), and a significantly elevated red cell distribution width (RDW) of 28.9% (reference: 10.0–14.3%), revealing microcytic, hypochromic anemia.
Iron studies revealed hyperferritinemia (ferritin: 866.8 µg/L; reference: 30–400 µg/L), serum iron of 119 µg/dL, transferrin of 150 mg/dL (Figure 1A) [9]. Haemoglobin profile was requested and the patient was characterized by an HbA level of 81.3%, a slightly elevated HBA2 (3.3%), and an atypical fraction (Z11 zone) of 14.2% (Figure 1B). This pattern was consistent with the suspicion of a carrier status for a pathogenetic hemoglobin variant.
The transferrin saturation accounted for 55.87%, enabling us to raise the suspicion of a possible iron-loading disorder. Molecular testing for hemochromatosis type 1 (HFE1, MIM #235200) was performed, yielding negative results for the more common HFE variants (C282Y, H63D, and S65C) (Figure 2A). Targeted genetic testing for hemoglobinopathies was pursued by NGS of the HBB, HBA1 and HBA2 genes, identifying three pathogenic variants in heterozygous state: c.118C>T; p.(Gln40Ter) and c.6259_5720del (Sicilian variant) in the HBB gene (Figure 2B), and c.427T>A; p.(Ter143LysextTer31) in the HBA2 gene.
Segregation analysis revealed that the HBB nonsense variant (c.118C>T) and the HBA2 variant were maternally inherited, while the Sicilian deletion was paternally inherited, confirming the compound heterozygosity in HBB. The proband’s partner underwent the same genetic testing and was found to carry a variant of uncertain significance (VUS) in the HBB gene, c.314G>A (Chr11:5225284 C>T), in heterozygous state, highlighting potential reproductive risk for the couple.
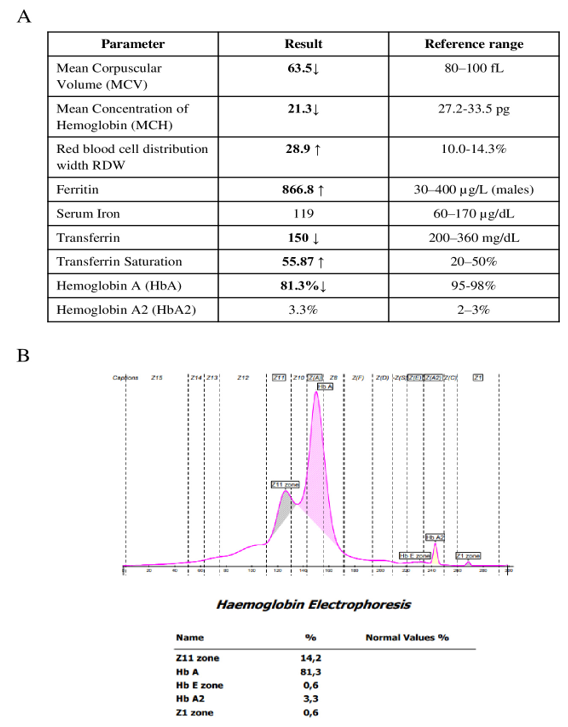
Figure 1: A. Hematological and iron parameters of the proband at the time of genetic evaluation, compared to reference range (Frenzel H, et al. 2011). B. Hemoglobin electrophoresis: the trace shows a predominant HbA fraction (81.3%), with HBA2 at the upper limit of normal (3.3%) and a Z11 zone fraction of 14.2%, consistent with the suspicion of a carrier of a hemoglobin variant.
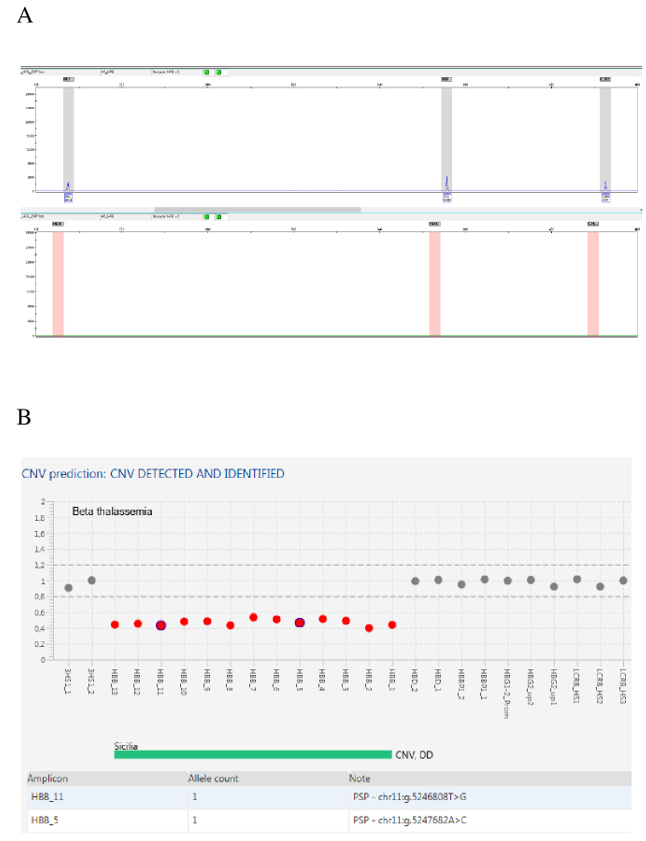
Figure 2: A. The electropherogram shows a wild-type profile at the C282Y, H63D, and S65C polymorphic sites of the HFE gene, with no pathogenic variants detected. B. CNV Amplification Suite Software analysis in the patient carrying the heterozygous c.6259_5720del variant in HBB, also called “Sicilian variant”.
Discussion
Haemoglobin is a tetrameric protein composed of two α- and two β-globin chains. In adults, haemoglobin A (HbA, α2β2) represents ~97% of total haemoglobin, while haemoglobin A2 (α2δ2) and fetal haemoglobin (α2γ2) account for ~2% and <1%, respectively [7].
β-thalassemia major is typically caused by homozygous or compound heterozygous loss-of-function variants in the HBB gene, leading to absent or markedly reduced β-globin synthesis. Affected individuals usually present within the first two years of life with severe microcytic anemia, require lifelong transfusions, and develop iron overload due to both disease and treatment2. In the present case, the proband harboured two pathogenic variants in HBB: c.118C>T p.(Gln40Ter) and c.6259_5720del (Sicilian variant). It is known that the presence of biallelic loss-of-function variants in HBB is classically associated with β-thalassemia major [8], but the patient was clinically asymptomatic, with no need for transfusion and only moderate haematological abnormalities. This phenotypic attenuation is likely explained by the co-inheritance of the HBA2 c.427T>A variant, which may mitigate the α/β-globin imbalance decreasing the alpha/non-alpha globin chain imbalance, as supported by previous findings on phenotypic modifiers in thalassemia [4-6].
Recognizing these modifiers is crucial when genotype-phenotype correlations appear inconsistent, as they inform prognosis, guide therapeutic strategies, and optimize genetic counselling.
The partner of the proband carried the HBB c.314G>A variant, in the 3' untranslated region, that can be classified as a variant of unknown significance according to the ACMG. Combined Annotation Dependent Depletion (CADD) was 5.374. GnomAD version 4.1 allele frequency was 0.00000342 in exomes and not reported in genomes and it is phylogenetically conserved (PhastCons100way: 0.477; PhyloP100way: -0.210). In silico predictors considered the variant “benign moderate”. Although not currently classified as pathogenic, the presence of a VUS in the partner of a known carrier warrants reproductive caution. According to ACMG guidelines, such findings necessitate individualized counselling and may prompt further analysis, including in silico prediction, familial segregation studies, or functional assays [5].
This case illustrates how a genotype consistent with a severe hemoglobinopathy may result in a milder clinical phenotype due to modifying genetic factors.
Finally, this report highlights the need for extended molecular testing in patients with unexplained microcytosis and altered iron parameters. Identifying biallelic pathogenic variants in HBB and co-inherited α-globin alteration not only refines diagnosis and the most appropriate management but also underscores the importance of integrating molecular data into reproductive risk assessments.
Conclusion
The present case underscores the importance of comprehensive molecular testing in the diagnostic evaluation of patients with atypical haematological findings. Despite a genotype typically associated with β-thalassemia major, the proband exhibited a mild phenotype likely modulated by a co-inherited pathogenic HBA2 variant. This discordance highlights the role of phenotypic modifiers in shaping clinical expression. Furthermore, the detection of a HBB variant of uncertain significance in the proband’s partner emphasizes the need for extended carrier screening and careful interpretation of genetic results in reproductive settings. Accurate molecular characterization enabled a more precise assessment of reproductive risk and guided appropriate counselling.
In conclusion, the integration of haematological data, iron metabolism parameters, and targeted molecular analysis is essential for defining the underlying cause of anemia, refining prognosis, and informing reproductive decision-making.
Ethical Approval Statement: This study was conducted in accordance with the ethical standards of the Declaration of Helsinki (1964) and its subsequent revisions. Ethical approval was obtained from the Local Ethics Committee of Gemelli Isola Comitato Etico Territoriale (CET 3). All family members provided also written informed consent for publication.
Acknowledgment: We thank the patient and his family for participating in this study.
Author contributions: S. Mariani: formal analysis, investigation, methodology and writing—original draft; S. Bucossi: formal analysis, investigation, methodology; R. Squitti: formal analysis, methodology, and writing—review and editing; A. Franciulli: formal analysis, investigation, methodology; M. C. A. Rongioletti: methodology, and writing—review and editing; G. Mastromoro: methodology, and writing—review and editing.
Competing Interests and Funding: The authors declare no competing interests and founding.
References
- Weatherall DJ, Clegg JB. The Thalassaemia Syndromes. 4th ed. Blackwell Science, 2001.
- Galanello R, Origa R. Beta-thalassemia. Orphanet J Rare Dis, 2010; 5: 11. doi:10.1186/1750-1172-5-11.
- Muncie HL Jr, Campbell JS. Alpha and beta thalassemia. Am Fam Physician, 2009; 80(4): 339-344.
- Galanello R, Cao A. Gene test review. Beta-thalassemia. Genet Med, 2011; 13(8): 708-717. doi:10.1097/GIM.0b013e3181a1d559.
- Richards S, Aziz N, Bale S, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med, 2015; 17(5): 405-424. doi:10.1038/gim.2015.30.
- Danjou F, Anni F, Perseu L, et al. Genetic modifiers of β-thalassemia and clinical severity as assessed by age at first transfusion. Haematologica, 2012; 97(7): 989-993. doi:10.3324/haematol.2011.053504.
- Schechter AN. Hemoglobin research and the origins of molecular medicine. Blood, 2008; 112(10): 3927-3938. doi:10.1182/blood-2008-07-077412.
- Cao A, Galanello R, Rosatelli MC. Genotype-phenotype correlations in beta-thalassemias. Blood Rev, 1991; 5(1): 1-12.
- Frenzel H, et al. Reference intervals for haemoglobin A2 and F measured by capillary electrophoresis. Clin Chem Lab Med, 2011; 49(2): 313-316.

